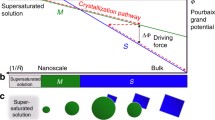
Abstract
Chemical routes to synthesize inorganics can start with solutions where multiple elements remain well mixed during liquid evaporation, precursor decomposition and crystallization. Because crystallization generally occurs at temperatures where diffusion is very limited, a single phase can crystallize with a greatly extended solid-solution that does not achieve its equilibrium phase assemblage until much higher temperatures where diffusion is no longer constrained. Partitioning at these higher temperatures can lead to unique microstructures such as the nano-composite illustrated here for a metastable Zr(1−x)Al x O2−x/2 (x≤0.57) phase that partitions into a composite containing t-ZrO2 grains and α-Al2O3 plates.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lange, F.F., Marshall, D.B., and Porter, J.R., “Controlling Microstructures Through Phase Partitioning From Metastable Precursors: The ZrO2-Y2O3 System,” p. 519–32 in Ultrastructure Processing of Advanced Ceramics. Edited by J.D. Mackenzie and D.R. Ulrich, John Wiley and Son, N.Y. (1988).
Leung, D.K., Chan, C.J., Ruhle, M., and Lange, F.F., “Metastable Crystallization, Phase Partitioning, and Grain Growth of ZrO2-Gd2O3 Materials Processed from Liquid Precursors,” J. Am. Ceram. Soc., 74 [11] 2786–92 (1991).
Balmer, M.L., Lange, F.F., and Levi, C.G., “Metastable Phase Selection and Partitioning in ZrO2-MgO Processed from Liquid Precursors,” J. Am. Cer. Soc., 75 [4] 946–52 (1992).
Mari Lou Balmer, Lange, Fred F., and Levi, Carlos, G., “Phase Selection and Extended Solubility in ZrO2-Al2O3 Processed from Liquid Precursors,” to be published.
Wilson, S.J. and McConnell, J.D.C., “A Kinetic Study of the System γ-AlOOH/Al2O3,” J. Solid State Chem., 34, 315–22 (1980).
Jayaram, V., Levi, C.G., and Mehrabian, R., “Characterization of Al2O3-ZrO2 Powders Produced by Electrohydrodynamic Atomization,” Materials Science and Engineering, A124, 65–81 (1990).
Kalonji, G., McKittrick, J., and Hobbs, L.W., “Applications of Rapid Solidification Theory and Practice to Al2O3-ZrO2 Ceramics,”; pp. 816–825 in Advances in Ceramics, Vol. 12, Science and Technology of Zirconia II, Edited by N. Claussen, M. Ruhle, and A.H., Heuer., American Ceramic Society, Columbus, OH, 1981.
Ando T. and Shiohara, Y., “Metastable Alumina Structures in Melt Extracted Alumina-25 wt% Zirconia and Alumina-42 wt% Zirconia Ceramics,” J. Am. Cer. Soc., 74[2] 410–17 (1991).
Alper, A.M. and Stewart, G.H., (ed.), Science of Ceramics, Vol. 3, Academic Press, London, 1967, p. 339.
Yamaguchi, O., Shirai, M., and Yoshinaka, M., “Formation and Transformation of Cubiz ZrO2 Solid Solutions in the System ZrO2-Al2O3,” J. Am. Cer. Soc., 71[12] C-510–C-512 (1988).
Balmer, Mari, Lou, Lange Fred F. and Levi, Carlos, G. “Development of Nano-Composite Microstructures in ZrO2-Al2O3 Via the Solution Precursor Method” Submitted to The Journal of the American Ceramic Society.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lange, F.F., Balmer, M.L. & Levi, C.G. Diffusion limited crystallization and phase partitioning in ZrO2-metal oxide binary systems. J Sol-Gel Sci Technol 2, 317–321 (1994). https://doi.org/10.1007/BF00486263
Issue Date:
DOI: https://doi.org/10.1007/BF00486263