Abstract
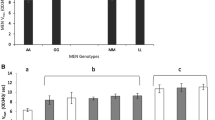
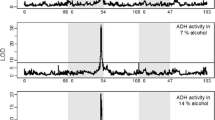
Adult Drosophila melanogaster from naturally occurring populations in the Eastern United States were examined by gel electrophoresis for their alcohol dehydrogenase (ADH) phenotype. The ADH enzymes were partially purified and characterized. Frequencies of the controlling alleles, Adh 4 and Adh 6, were discovered to vary in a clinal pattern. Adh 6 reaches a maximum frequency of about 0.90 in the South and minimum of about 0.50 in the North. Partially purified enzymes from the three Adh genotypes varied according to specific activity, substrate specificity, and heat stability. A differential influence of pH was indicated. There was little variation in K m values for ethanol and DPN+ among the enzymes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dunn, G. R., Wilson, T. G., and Jacobson, K. B. (1969). Age dependent changes in alcohol dehydrogenase in Drosophila. J. Expt. Zool. 171185.
Gibson, J. (1970). Enzyme flexibility in Drosophila melanogaster. Nature 227959.
Grell, E. H., Jacobson, K. B., and Murphy, J. B. (1968). Alterations of genetic material for analysis of alcohol dehydrogenase isozymes of Drosophila melanogaster. Ann. N.Y. Acad. Sci. 151441.
Jacobson, K. B. (1968). Alcohol dehydrogenase of Drosophila: Interconversion of isoenzymes. Science 159324.
Jacobson, K. B., Murphy, J. B., and Hartman, F. C. (1970). Isoenzymes of Drosophila alcohol dehydrogenase. I. Isolation and interconversion of different forms. J. Biol. Chem. 2451075.
Jacobson, K. B., Murphy, J. B., Knopp, J. A., and Ortiz, J. R. (1972). Multiple forms of Drosophila alcohol dehydrogenase. III. Conversion of one form to another by nicotinamide adenine dinucleotide or acetone. Arch. Biochem. Biophys. 14922.
Johnson, F. M., and Denniston, C. (1964). Genetic variation of alcohol dehydrogenase in Drosophila melanogaster. Nature, 204906.
Johnson, F. M., Schaffer, H. E., Gillaspy, J. E., and Rockwood, E. S. (1969). Isozyme genotype-environment relationships in natural populations of the harvester ant, Pogonomyrmex barbatus, from Texas. Biochem. Genet. 3429.
Kimura, M., and Ohta, T. (1971). Protein polymorphism as a phase of molecular evolution. Nature 229467.
Knopp, J. A., and Jacobson, K. B. (1972). Multiple forms of Drosophila alcohol dehydrogenase. IV. Protein fluorescence studies. Arch. Biochem. Biophys. 14936.
Koehn, R. K. (1969). Esterase heterogeneity: Dynamics of polymorphism. Science 163943.
Koehn, R. K., and Rasmussen, D. I. (1967). Polymorphic and monomorphic serum esterase heterogeneity in castomid fish populations. Biochem. Genet. 1131.
Lowry, O. H., Rosenbrough, M. J., Farr, A. L., and Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193265.
Merritt, R. B. (1972) Geographic distribution and enzymatic properties of lactate dehydrogenase allozymes in the fathead minnow, Pimephales promelas. Am. Naturalist 106173.
Mukai, T., Mettler, L. E., and Chigusa, S. I. (1971). Linkage disequilibrium in a local population of Drosophila melanogaster. Proc. Natl. Acad. Sci. 681065.
Prakash, S., Lewontin, R. C., and Hubby, J. L. (1969). A molecular approach to the study of genic heterozygosity in natural populations. IV. Patterns of genic variation in central, marginal and isolated populations of Drosophila pseudoobscura. Genetics 61841.
Rasmuson, B., Nilson, L. R., Rasmuson, M., and Zeppezauer, E. (1966). Effects of heterozygosity on alcohol dehydrogenase (ADH) activity in Drosophila melanogaster. Hereditas 56313.
Richmond, R. C. (1972). Enzyme variability in the Drosophila willistoni group. III. Amounts of variability in the superspecies D. paulistorum. Genetics 7087.
Selander, R. K., Yang, S. Y., and Hunt, W. G. (1969). Polymorphism in esterases and hemoglobin in wild populations of the house mouse (Mus musculus). Univ. Texas Publ. 6918271.
Sofer, W., and Ursprung, H. (1968). Drosophila alcohol dehydrogenase: Purification and partial characterization. J. Biol. Chem. 2433110.
Stone, W. S., Johnson, F. M., Kojima, K., and Wheeler, M. R. (1968). Isozyme variation in island populations of Drosophila. I. An analysis of a species of the nasuta complex in Samoa and Fiji. Univ. Texas Publ. 6818157.
Ursprung, H., Sofer, W. H., and Burroughs, N. (1970). Ontogeny and tissue distribution of alcohol dehydrogenase in Drosophila melanogaster. Wilhelm Roux' Arch. 164201.
Yamazaki, T. (1971). Measurement of fitness at the esterase-5 locus in Drosophila pseudoobscura. Genetics 67579.
Author information
Authors and Affiliations
Additional information
This work was supported by AEC Contract No. AT-(40-1)-3980 and by PHS Research Grant No. GM 11546.
Paper No. 3880 of the Journal Series of the North Carolina Experiment Station, Raleigh, North Carolina. This work incorporates, in part, the thesis research of C. L. Vigue to be submitted in partial fulfillment of the Ph.D. requirements in Genetics.
Rights and permissions
About this article
Cite this article
Vigue, C.L., Johnson, F.M. Isozyme variability in species of the genus Drosophila. VI. Frequency-property-environment relationships of allelic alcohol dehydrogenases in D. melanogaster . Biochem Genet 9, 213–227 (1973). https://doi.org/10.1007/BF00485735
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00485735