Abstract
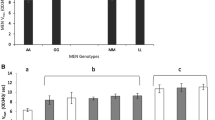
Two Drosophila melanogaster strains, each heterozygous for “fast” and “slow” alleles at the Adh locus, and each having balanced second chromosomes, were found to differ in the apparent thermostability of the slow allozyme. The two strains were crossed, and F1heterozygotes were separated on the basis of the origin of the slow allele. After electrophoresis, the cellulose acetate strips were treated 1 1/2 min at 35 C. The putatively more sensitive allozyme showed a strikingly greater response to heat. These findings further support the conclusion that electrophoretically cryptic allelic differences exist which are expressed in thermostability differences. Further application of this approach has revealed one similar sensitive slow allozyme and three cases of a relatively resistant fast ADH allozyme in wild-caught flies.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bernstein, S. C., Throckmorton, H. L., and Hubby, J. L. (1973). Still more genetic varIability in natural populations. Proc. Natl. Acad. Sci. 703928.
Lewontin, R. C. (1973). Population genetics. Ann. Rev. Genet. 71.
Schwartz, M., Gerace, L., O'Donnell, J., and Sofer, W. (1974). Drosophila alcohol dehydrogenase: Origin of the multiple forms. In Markert, C. L. (ed.), Isozymes, Vol. I: Molecular Structure, Academic Press, New York, pp. 725–751.
Singh, R. S., Hubby, J. L., and Lewontin, R. C. (1974). Molecular heterosis for heat-sensitive enzyme alleles. Proc. Natl. Acad. Sci. 711808.
Singh, R. C., Hubby, J. L., and Throckmorton, L. H. (1975). A study of genic variation by electrophoretic and heat denaturation techniques at the Odh locus in members of the Drosophila virilis group. Genetics 80637.
Thörig, G. E. W., Schoone, A. A., and Scharloo, W. (1975). Variation between electrophoretically identical alleles at the alcohol dehydrogenase locus in Drosophila melanogaster. Biochem. Genet. 13271.
Wright, T. R. F., and MacIntyre, R. J. (1965). Heat-stable and heat-labile esterase-6F enzymes in Drosophila melanogaster produced by different Est 6 Falleles. Jour. Elisha Mitchell Scientific Soc. 8117.
Author information
Authors and Affiliations
Additional information
This work was supported by NIH Grant GM18967-02-03.
Rights and permissions
About this article
Cite this article
Milkman, R. Further evidence of thermostability variation within electrophoretic mobility classes of enzymes. Biochem Genet 14, 383–387 (1976). https://doi.org/10.1007/BF00484776
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00484776