Abstract
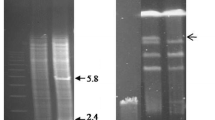
1) Rhodobacter capsulatus (formerly Rhodopseudomonas capsulata) strain 37b4 was subjected to transposon Tn5 mutagenesis. 2) Kanamycin-resistant transconjugants were screened for their inability to reduce trimethylamine-N-oxide (TMAO) as judged by the lack of alkali production during anaerobic growth on plates containing glucose as carbon source and cresol red as pH indicator. 3) Of 6 mutants examined, all were found to have considerably decreased levels of methylviologen-dependent TMAO reductase activity and dimethylsulphoxide (DMSO) reductase activity. 4) Periplasmic fractions of one of these mutants (DK9) and of the parent strain were subjected to sodium dodecylsulphate polyacrylamide gel electrophoresis. The gels were stained for TMAO-reductase and DMSO-reductase. With the wild-type strain, only a single polypeptide band, Mr=46,000, stained for TMAO and DMSO reductase activity. In mutant DK9 this band was not detectable. 5) In contrast to the parent strain, harvested washed cells of mutant DK9 were unable to generate a cytoplasmic membrane potential in the presence of TMAO or DMSO under dark anaerobic conditions. 6) In contrast to the parent strain, DK9 was unable to grow in dark anaerobic culture with fructose as the carbon source and TMAO as oxidant.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- TMAO:
-
trimethylamine-N-oxide
- DMSO:
-
dimethylsulphoxide
- PMS:
-
phenazine methosulphate
- Δψ:
-
cytoplasmic membrane potential
References
Clark AJ, Cotton NPJ, Jackson JB (1983) The influence of the ionic conductance on the relation between electron transport and protonmotive force in intact cells of Rhodopseudomonas capsulata. Eur J Biochem 130:575–580
Clayton RK (1963) Towards the isolation of a photosynthetic reaction centre in Rhodopseudomonas capsulata. Biochim Biophys Acta 73:312–323
Cotton NPJ, Jackson JB (1982) Kinetics of carotenoid absorption changes in intact cells of photosynthetic bacteria. Biochim Biophys Acta 679:138–145
Cox JC, Madigan MT, Faringer JF, Gest H (1980) Redox mechanisms in “oxidant-dependent” hexose fermentation by Rhodopseudomonas capsulata. Arch Biochem Biophys 204:10–17
Datta N, Hedges RW, Shaw EJ, Sykes RB, Richmond MH (1971) Properties of an R factor from Pseudomonas aeruginosa. J Bacteriol 108:1244–1249
Davidson AE, Fukimoto HE, Jackson CE, Barrett EL, Chang GW (1979) Mutants of Salmonella typhimurium defective in the reduction of trimethylamine oxide. FEMS Microbiol Lett 6: 417–420
Drews G, Oelze J (1981) Organisation and differentiation of membranes of phototrophic bacteria. Adv Microbial Physiol 22:1–92
Ferguson SJ, Jackson JB, McEwan AG (1987) Anaerobic respiration in the Rhodospirillacae: characterisation of pathways and evaluation of roles in redox balancing during photosynthesis. FEMS Microbiol Rev 46:117–143
Kaufmann N, Hudig H, Drews G (1984) Transposon Tn5 mutagenesis of genes for the photosynthetic apparatus in Rhodopseudomonas capsulata. Mol Gen Genet 198:153–158
Kleckner N, Roth J, Botstein D (1977) Genetic engineering in vivo using translocatable drug resistance elements. J Mol Biol 116:125–159
Kwan HS, Barrett EL (1983) Roles for menaquinone and the two trimethylamine oxide (TMAO) reductases in TMAO respiration in Salmonella typhimurium: Mad (Apr lac) insertion mutations in men and tor. J Bacteriol 115:1147–1155
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the lead of bacteriophage T4. Nature (Lond.) 227:680–685
Lowry OH, Rosebrough HJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Madigan MR, Gest H (1978) Growth of photosynthetic bacteria anaerobically in darkness, supported by “oxidant-dependent” sugar fermentation. Arch Microbiol 117:119–122
McEwan AG, George CL, Ferguson SJ, Jackson JG (1982) A nitrate reductase activiy in Rhodopseudomonas capsulata linked to electron transfer and generation of a membrane potential. FEBS Lett 150:277–280
McEwan AG, Ferguson SJ, Jackson JB (1983) Electron flow to dimethylsulphoxide or trimethylamine-N-oxide generates a membrane potential in Rhodopseudomonas capsulata. Arch Micrbiol 136:300–305
McEwan AG, Jackson JB, Ferguson SJ (1984) Rationalisation of properties of nitrate reductases in Rhodopseudomonas capsulata. Arch Microbiol 137:344–349
McEwan AG, Greenfield AJ, Wetzstein HG, Jackson JB, Ferguson SJ (1985a) Nitrous oxide reduction by members of the family Rhodospirillaceae and the nitrous oxide reductase of Rhodopseudomonas capsulata. J Bacteriol 164:823–830
McEwan AG, Wetzstein HG, Ferguson SJ, Jackson JB (1985b) Periplasmic location of the terminal reductase in trimethylamine-N-oxide and dimethylsulphoxide respiration in the photosynthetic bacterium Rhodopseudomonas capsulata. Biochim Biophys Acta 806:410–417
McEwan AG, Cotton NPJ, Ferguson SJ, Jackson JB (1985c) The role of auxiliary oxidants in the maintencance of a balanced redox poise for photosynthesis in bacteria. Biochim Biophys Acta 810:140–147
McEwan AG, Wetzstein HG, Meyer O, Jackson JB, Ferguson SJ (1987) The periplasmic nitrate reductase of Rhodobacter capsulatus; purification, characterisation and distinction from a single reductase for trimethylamine-N-oxide, dimethylsulph-oxide and chlorate. Arch Microbiol 147:340–345
Pfennig N (1977) Phototrophic green and purple bacteria: a comparative systematic survey. Microbiology 31:275–290
Richardson DJ, Kelly DJ, Jackson JB, Ferguson SJ, Alef K (1986) Inhibitory effects of myxothiazol and 2-n-heptyl-4-hydroxquinoline-N-oxide on the auxiliary electron transport pathways of Rhodobacter capsulatus. Arch Microbiol 146:159–165
Richardson DJ, King GF, Kelly DJ, McEwan AG, Ferguson SJ, Jackson JB (1988) The role of auxiliary oxidants in maintaining redox balance during phototrophic growth of Rhodobacter capsulatus on reduced carbon substrates. Arch Microbiol 150:131–137
Schulz JE, Weaver PF (1982) Fermentation and anaerobic respiration by Rhodospirillum rubrum and Rhodopseudomonas capsulata. J Bacteriol 149:181–190
Shimokawa B, Ishimoto I (1979) Purification and some properties of inducible tertiary amine-N-oxide reductase from E. coli. J Biochem 86:1709–1717
Simon R, Priefer V, Peuhler A (1983) Vector plasmids for in vivo and in vitro, manipulations of Gram-negative bacteria. In: Peuhler A (ed) Molecular biology of bacteria plant interaction. Springer, Berlin Heidelberg New York p 99
Styrvold OB, Strom AR (1984) Dimethyl sulphoxide and trimethylamine-N-oxide respiration of Proteus vulgaris. Arch Microbiol 140:74–78
Weaver PF, Wall JD, Gest H (1975) Characterisation of Rhodopseudomonas capsulata. Arch Microbiol 105:207–216
Yamamoto J, Okibo N, Ishimoto M (1986) Further characterisation of trimethylamine-N-oxide reductase from Escherichia coli, a molybdoprotein. J Biochem (Tokyo) 99:1773–1779
Yen H-C, Marrs BL (1977) Growth of Rhodopseudomonas capsulata under anaerobic dark conditions with dimethylsulphoxide. Arch Biochem Biophys 181:411–418
Yu PL, Cullum J, Drews G (1981) Conjugational transfer systems of Rhodopseudomonas capsulata mediated by R plasmids. Arch Microbiol 128:390–393
Zumft WG, Dohler K, Korner H (1985) Isolation and characterisation of transposon Tn5-induced mutants of Pseudomonas perfectomarina defective in nitrous oxide respiration. J Bact 613:918–924
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kelly, D.J., Richardson, D.J., Ferguson, S.J. et al. Isolation of transposon Tn5 insertion mutants of Rhodobacter capsulatus unable to reduce trimethylamine-N-oxide and dimethylsulphoxide. Arch. Microbiol. 150, 138–144 (1988). https://doi.org/10.1007/BF00425153
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00425153