Abstract
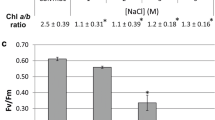
The responses of the filamentous cyanobacterium Spirulina platensis to increased NaCl concentrations (0.25–1.0 M) in addition to the concentration of sodium in the growth medium were studied. A two stage response to the salt stress was observed. This consisted of a relatively short shock stage, followed by adaptation process. It was shown that upon exposure to high salt concentrations of 0.5 M and above, immediate inhibition of photosynthesis and respiration, and complete cessation of growth occurred. After a time lag, the energy-yielding processes exhibited restored activity. At 0.5 and 1.0M NaCl photosynthesis reached 80% and 50% that of the control, while respiration was enhanced by 140 and 200%, respectively. The time lags were longer when the cells were exposed to higher NaCl concentrations. The resumption of growth and the establishment of new steady state growth rates were found to be correlated to the recovery in respiration. The relationship between the growth rates after adaptation and the increased NaCl concentrations was found to be inversely linear. The cellular sodium content was maintained at a constant low level, regardless of the external NaCl concentration, while potassium content declined linearly vs. the external NaCl concentration. The carbohydrate content of the cells rose exponentially with the increase in NaCl concentration.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Bennet A, Bogorad L (1973) Complementary chromatic adaptation in a filamentous blue-green alga. J Cell Biol 58:419–435
Blumwald E, Tel-Or E (1982) Osmoregulation and cell composition in salt adaptation of Nostoc muscorum. Arch Microbiol 132:168–172
Blumwald E, Mehlhorn RJ, Packer L (1984a) Salt adaptation mechanisms in the cyanobacterium Synechococcus 6311. In: Sybesma S (ed) Advances in photosynthesis research. Martinus Nijhoff/ Dr W Junk Publ, The Hague, pp 627–630
Blumwald E, Wolosin JM, Packer L (1984b) Na+/H+ exchange in the cyanobacterium Synechococcus 6311. Biochem Biophys Res Commun 122:452–459
Ciferri O (1983) Spirulina, the edible microorganism. Microbiol Rev 47:551–578
Ehrenfeld J, Cousin JL (1984) Ionic regulation of the unicellular green alga Dunaliella teritolecta: response to hypertonic shock. J Membr Biol 77:45–55
Fry IV, Huflejt M, Erber WWA, Peschek GA, Packer L (1986) The role of respiration during adaptation of the freshwater cyanobacterium Synechococcus 6311 to salinity. Arch Biochem Biophys 244:686–691
Hagemann M, Erdmann N, Wittenburg E (1987) Synthesis of glucosylglycerol in salt-stressed cells of the cyanobycterium Microcystis firma. Arch Microbiol 148:275–279
Hassid WZ, Abraham S (1957) Chemical procedures for analysis of polysaccharides. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, vol 3. Academic Press, New York, pp 34–50
Katz A, Avron M (1985) Determination of intracellular osmotic volume and sodium concentration in Dunaliella. Plant Physiol 78:817–820
Krulwich TA (1986) Bioenergetics of alkalophilic bacteria. J Membr Biol 89:113–125
Mackay MM, Norton RS, Borowitzka LJ (1984) Organic osmoregulatory solutes in cyanobacteria. J Microbiol 130:2177–2191
Molitor V, Erber W, Peschek GA (1986) Increased levels of cytochrome oxidase and sodium-proton antiporter in the plasma membrane of Anacystis nidulans after growth in sodium enriched media. FEBS Lett 204:251–256
Reed RH, Richardson DL, Stewart WDP (1985) Na+ uptake and extrusion in the cyanobacterium Synechocystis PCC 6714 in response to hyper-saline treatment. Evidence for transient changes in plasmalema Na+ permeability. Biochim Biophys Acta 814:347–355
Reed RH (1986) Halotolerant and halophilic microbes. In: Herbert RA, Codd GA (eds) Microbes in extreme environment. Academic Press, London, pp 55–82
Richmond A (1988) Spirulina. In: Borowitzka A, Borowitzka L (eds) Micro-algal biotechnology. Cambridge University Press, Cambridge, pp 85–121
Vonshak A (1986) Laboratory techniques for the culturing of microalgae. In: Richmond A (ed) Handbook for microalgal mass culture. CRC Press, Boca Raton Florida, pp 117–145
Vonshak A (1987) Biological limitation in developing the biotechnology for algal mass cultivation. Science De L'eau 6:99–103
Vonshak A, Richmond A (1981) Photosynthetic and respiratory activity in Anacystis nidulans adapted to osmotic stress. Plant Physiol 68:504–505
Vonshak A, Richmond A (1988) Mass production of Spirulina: an overview. Biomass (in press)
Warr SRC, Reed RH, Chudek JA, Foster R, Stewart WDP (1985) Osmotic adjustment in Spirulina platensis. Planta 163:424–429
Wyn-Jones RC, Gorham J (1983) Osmoregulation. In: Lange OL, Nobel PS, Osmond CB, Ziegler H (eds) Encyclopedia of plant physiology, vol 12C, Physiological plant ecology, III. Response to the chemical and biological environment. Springer, Berlin Heidelberg New York, pp 35–58
Author information
Authors and Affiliations
Additional information
Publication No. 34 from the Micro-Algal Biotechnology Lab.
Rights and permissions
About this article
Cite this article
Vonshak, A., Guy, R. & Guy, M. The response of the filamentous cyanobacterium Spirulina platensis to salt stress. Arch. Microbiol. 150, 417–420 (1988). https://doi.org/10.1007/BF00422279
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00422279