Abstract
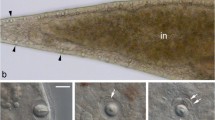
We have studied the fate of electron dense microbodies in nematode-trapping organs (traps) of the fungus A. oligospora during the initial hours following nematode capture. The interaction studies were performed with isolated traps which had captured a nematode under conditions where the fungal cells had no access to external energy sources. Video enhanced contrast microscopy showed that under these conditions the number of dense bodies present in the trap cell that formed the penetration tube, rapidly decreased. During subsequent penetration and development of the infection bulb this decrease continued while at this time common cell organelles such as mitochondria and vacuoles were formed. This was confirmed by electron microscopy which also revealed that the dense bodies were degraded by means of an autophagic process. The organelles were degraded individually and finally turned into compartments which, based on ultrastructural criteria, were considered vacuoles. Fusion of such vacuoles into larger organelles frequently occurred. The degradation process was initiated early in the interaction since initial stages were already evident within 15 min after capture. Generally it took 1–2 h before the infection bulb had fully developed and trophic hyphae formation started. During this time the original trap cell, characterized by numerous dense bodies, was transformed into an active vegetative hyphal cell containing typical cell organelles such as nuclei, mitochondria, a strongly proliferated endoplasmic reticulum, vacuoles and “normal” microbodies but lacked dense bodies. This disappearance of dense bodies was confined to the cell that penetrated the nematode and—less frequently—its two neighbouring cells in the hyphal loop. In the other cells, constituting the trap, the dense bodies remained unaffected. As will be discussed, the present results support our current view that traps of A. oligospora contribute to the survival of the organism in its natural environment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Friman E, Olsson S & Nordbring-Hertz B (1985) Heavy trap formation by Arthrobotrys oligospora in liquid culture. FEMS Microbiol. Ecol. 31: 17–21
Jansson B & Nordbring-Hertz B (1981) Trap and conidiophore formation in Arthrobotrys superba. Trans. Br. Mycool. Soc. 77(1): 205–207
Nordbring-Hertz B (1972) Scanning electron microscopy of the nematode trapping organs in Arthrobotrys oligospora. Physiol. Plant 26: 279–284
Nordbring-Hertz B (1973) Peptide-induced morphogenesis in the nematode-trapping fungus Arthrobotrys oligospora. Physiol. Plant. 29: 223–23
Nordbring-Hertz B (1977) Nematode-induced morphogenesis in the predacious fungus Arthrobotrys oligospora. Nematologica 23: 443–451
Nordbring-Hertz B (1984) Mycelial development and lectin carboohydrate interactions in nematode-trapping fungi. In: Jennings DH & Rayner ADM (Eds) The Ecology and Physiology of the Fungal Mycelium (pp 419–432) Cambridge University Press, Cambridge
Nordbring-Hertz B & Stalhammar-Carlemalm M (1978) Capture of mematodes by Arthrobotrys oligospora; an electron microscope study. Can. J. Bot. 56: 1297–1307
Nordbring-Hertz B, Veenhuis M & Harder W (1984) Dialysis membrane technique for ultrastructural studies of microbial interactions. Appl. Environm. Microbiol. 47: 195–197
Nordbring-Hertz B, Wyss U, Zuncke U & Veenhuis M (1986) Trap formation and capture of nematodes by Arthrobotrys oligospora. Institut für den Wissenschaftlichen Film, series 19, number 12, Göttingen
Nordbring-Hertz B, Fehne-Persson K, De Vries OMH & Veenhuis M (1989) Protoplast formation from adhesive network traps of Arthrobotrys oligospora. Trans. Br. Mycol. Soc., 92(1): 50–54
Toth R, Toth D & Nordbring-Hertz B (1980) Cytochemical localization of acid phosphatase in the nematode-trapping fungus Arthrobotrys oligospora. Mycologia 72: 813–817
Van Dijken JP, Veenhuis M, Vermeulen CA & Harder W (1976) Cytochemical localization of catalase activity in methanol-grown Hansenula polymorpha. Arch. Microbiol. 105: 261–267
Veenhuis M, Van Dijken JP & Harder W (1980) A new method for the cytochemical demonstration of phosphotase activities in yeasts based on the use of cerous ions. FEMS Microbiol. Lett. 9: 285–291
Veenhuis M, Douma A, Harder W & Osumi M (1983) Degradation and turnover of peroxisomes in the yeast Hansenula polymorpha induced by selective inactivation of peroxisomal enzymes. Arch. Microbiol. 134: 193–203
Veenhuis M, Nordbring-Hertz B & Harder W (1984) Occurrence, characterization and development of two different types of microbodies in the nematophagous fungus Arthrobotrys oligospora. FEMS Microbiol. Lett. 24: 31–38
Veenhuis M, Nordbring-Hertz B & Harder W (1985a) An electron microscopial analysis of capture and initial stages of penetration of nematodes by Arthrobotrys oligospora. Antonie van Leeuwenhoel 51: 385–398
Veenhuis M, Nordbring-Hertz B & Harder W (1985b) Development and fate of electron-dense microbodies in trap cells of the nematophagous fungus Arthrobotrys oligospora. Antonie van Leeuwenhoek 51: 399–407
Veenhuis M, Sjollema K, Nordbring-Hertz B & Harder W (1989a) An improved method for light-and electron microscopical studies on nematode/fungal interactions. Antonie van Leeuwenhoek, 55: 361–368
Veenhuis M, Harder W & Nordbring-Hertz B (1989b) Occurrence and metabolic significance of microbodies in trophic hyphae of the nematophagous fungus Arthrobotrys oligospora. Antonie van Leeuwenhoek, 56: 241–249 (this issue)
Wyss U & Zuncke U (1986) The potential of high resolution video-enhanced contrast microscopy in nematological research. Revue Nematol. 9(1): 91–94
Zwart KB (1983) Metabolic significance of microbodies in the yeasts Candida utilis and Hansenula polymorpha. PhD Thesis, University of Groningen
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Veenhuis, M., Van Wijk, C., Wyss, U. et al. Significance of electron dense microbodies in trap cells of the nematophagous fungus Arthrobotrys oligospora . Antonie van Leeuwenhoek 56, 251–261 (1989). https://doi.org/10.1007/BF00418937
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00418937