Abstract
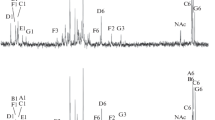
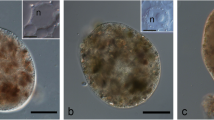
Cell walls were prepared from freeze-dried samples of 7 strains of Methanobacterium by mechanical disintegration of the cells followed by incubation with trypsin. Electron microscopy revealed the presence of sacculi exhibiting the shape of the original cells, on which no surface structure could be detected. Ultrathin sections of the isolated sacculi showed a homogenously electron dense layer of about 10–15 nm in width. The ash content varied between 8 and 18% of dry weight. The sacculi of all the strains contained Lys: Ala: Glu: GlcNAc or GalNAc in a molar ratio of about 1:1.2:2:1. In one strain (M. ruminantium M 1) alanine is replaced by threonine, however. Neutral sugars and-in some strains-additional amounts of the amino sugars were present in variable amounts, and could be removed by formamide extraction or HF treatment without destroying the sacculi. No muramic acid or d-amino acids typical of peptidoglycan were found. Therefore, the sacculi of the methanobacteria consist of a different polymer containing a set of three l-amino acids and one N-acetylated amino sugar. From cells of Methanospirillum hungatii no sacculi, but tube-like sheaths could be isolated, which tend to fracture perpendicularly to the long axis of the sheath along the fibrills seen on the surface. The sheaths consist of protein containing 18 amino acids and small amounts of neutral sugars. They are resistent to the proteinases tested and are not disintegrated by boiling in 2% sodium dodecylsulfate for 30 min.
The three Gram-negative strains Black Sea isolate JR-1, Cariaco isolate JR-1 and Methanobacterium mobile do not contain a rigid sacculus, but merely a SDS-sensitive surface layer composed of regularly arranged protein subunits. This evidence indicates that, within the methanogens, different cell wall polymers characteristic of particular groups of organisms may have evolved during evolution, and supports the hypothesis that the evolution of the methanogens was separated from that of the peptidoglycan-containing procaryotic organisms at a very early stage.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- SDS:
-
sodium dodecylsulfate
- EDTA:
-
ethylenediaminetetra acetic acid
- DNP:
-
dinitrophenyl
References
Albersheim, P., Nevins, D. J., English, P. D., Karr, A.: A method for the analysis of sugars in plant cell-wall polysaccharides by gas liquid chromatography. Carboh. Res. 5, 340–345 (1967)
Baddiley, J., Davison, A. L.: The occurence and location of teichoic acid in lactobacilli. J. Gen. Microbiol. 24, 295–299 (1961)
Bailey, J. L.: Techniques in protein chemistry. Amsterdam-London-New York, Elsevier 1962
Balch, W. E., Magrum, L. J., Fox, G. E., Wolfe, R. S., Woese, C. R.: An ancient divergence among the bacteria. J. Mol. Evol. 9, 305–311 (1977)
Bergmeyer, H. U.: Methoden der enzymatischen Analyse. Weinheim: Verlag Chemie 1974
Blumenkrantz, N., Asboe-Hansen, G.: New method for quantitative determination of uronic acids. Analyt. Biochem. 54, 484–489 (1973)
Bryant, M. P., McBride, B. C., Wolfe, R. S.: Hydrogen-oxidizing methane bacteria. I. Cultivation and methanogenesis. J. Bacteriol. 95, 1118–1123 (1968)
Chen, P. S., Toribara, T. Y., Warner, H.: Microdetermination of phosphorus. Analyt. Chem. 28, 1756–1758 (1956)
Dodgston, K. S., Price, R. G.: A note on the determination of the ester sulphate content of sulphated polysaccharides. Biochem. J. 84, 106–110 (1962)
Fox, G. F., Magrum, L. J., Balch, W. E., Wolfe, R. S., Woese, C. R.: Classification of methanogenic bacteria by 16S ribosomal RNA characterization. Proc. Natl. Acad. Sci. U.S.A. 74, 4537–4541 (1977)
Ghuysen, J. M., Tipper, D. I., Strominger, J. L.: Enzymes that degrade bacterial cell walls. In: Methods in enzymology, Vol. VIII (S. P. Colowick, N. O. Kaplan, eds.), pp. 685–699. LOndon-New York: Academic Press 1966
Hestrin, S.: The reaction of acetylcholine and other carboxylic acid derivatives with hydroxylamine and its analytical application. J. Biol. Chem. 180, 249–261 (1949)
Jones, J. B., Bowers, B., Stadtman, T. C.: Methanococcus vannielii: ultrastructure and sensitivity to detergents and antibiotics. J. Bacteriol. 130, 1357–1363 (1977)
Kandler, O., Hippe, H.: Lack of peptidoglycan in the cell walls of Methanosarcina barkeri. Arch. Microbiol. 113, 57–60 (1977)
Kandler, O., König, H.: Chemie der peptidoglycanfreien Zellwände der Methanbakterien. Hoppe-Seyler's Z. Physiol. Chem. 359, 282–283 (1978)
Kato, K., Strominger, J. L.: Structure of the cell wall of Staphylococcus aureus. IX. Mechanism of hydrolysis of the L11 enzyme. Biochem. 7, 2754–2761 (1968)
Larson, D. M., Setsinger, D. C., Waibel, P. E.: Procedure for determination of d-amino acids. Anal. Biochem. 39, 395–401 (1971)
Merck: “Biochemical” Enzyme. Darmstadt: Merck 1974
Mort, A. J., Lamport, D. T. A.: Anhydrous hydrogen fluoride deglycosylates glycoproteins. Analyt. Biochem. 82, 289–309 (1977)
Rao, K. R., Sober, H. A.: Preparation and properties of 2,4-dinitrophenyl-l-amino acids. J. Am. Chem. Soc. 76, 1328–1331 (1954)
Romesser, J. A., Mayer, F., Spiess, E., Wolfe, R. S.: Methanococcus malmarii, sp. n., and Methanococcus cariacii, sp. n. Isolation and characterization of two marine methanogens. Int. J. Syst. Bacteriol. (in press, 1978)
Ryter, A., Kellenberger, E., Birch-Andersen, A., Maaløe, O.: Etude au microscope électronique de plasmas contenant de l'acide désoxyribonucléique. I. Les nucléoides des bactéries en croissance active. Z. Naturforsch. 13b, 597–605 (1958)
Schleifer, K. H., Kandler, O.: Peptidoglycan types of bacterial cell walls and their taxonomic implications. Bacteriol. Rev. 36, 407–477 (1972)
Schleifer, K. H., Plapp, R., Kandler, O.: Die Aminosäuresequenz des Mureins von Microbacterium lacticum. Biochim. Biophys. Acta 154, 573–582 (1958)
Sentandreu, R., Northcote, D. H.: The structure of a glycopeptide isolated from the yeast cell wall. Biochem. J. 109, 419–432 (1968)
Spurr, A. R.: A low-viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res. 26, 31–43 (1969)
Steber, J., Schleifer, K. H.: Halococcus morrhuae: A sulfated heteropolysaccharide as the structural component of the bacterial cell wall. Arch. Microbiol. 105, 173–177 (1975)
Takebe, I.: Extent of crosslinkage in the murein sacculus of Escherichia coli B cell wall. Biochim. Biophys. Acta 101, 124–126 (1965)
Taylor, C. D., Wolfe, R. S.: A simplified assay for coenzyme M. J. Biol. Chem. 249, 4886–4890 (1974)
Trevelyan, W. E., Procter, D. D., Harrison, J. S.: Detection of sugars on paper chromatograms. Nature 166, 444–445 (1950)
Westphal, O., Jann, K.: Bacterial Lipopolysaccharides. In: Methods in carbohydrate chemistry, Vol. V (R. L. Whistler, J. N. BeMiller, M. L. Wolfrom, eds.), pp. 83–91. New York-London: Academic Press 1965
Witzerbin-Falszpan, J., Das, B. C., Petit, J.-F., Lederer, E.: The amino acids of the cell wall of Myobacterium tuberculosis var. bovis, strain BCG. Eur. J. Biochem. 32, 525–532 (1973)
Zeikus, J. G.: The biology of methanogenic bacteria. Bacteriol. Rev. 41, 514–541 (1977)
Zeikus, J. G., Bowen, V. G.: Comparative ultrastructure of methanogenic bacteria. Can. J. Microbiol. 21, 121–129 (1975)
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. Dr. Adolf Butenandt on the occasion of his 75th birthday
Rights and permissions
About this article
Cite this article
Kandler, O., König, H. Chemical composition of the peptidoglycan-free cell walls of methanogenic bacteria. Arch. Microbiol. 118, 141–152 (1978). https://doi.org/10.1007/BF00415722
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00415722