Abstract
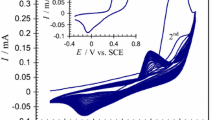
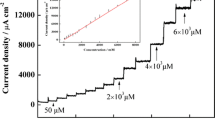
Several types of amperometric biosensors comprised of immobilized glucose oxidase in chemically-modified ceramic-carbon matrices are compared. The electrodes are comprised of several building blocks each performing a specific function. Glucose oxidase is used to catalyze the bio-oxidation of glucose; carbon powder imparts conductivity and favorable electrochemical characteristics; the Ormosil network provides rigidity and porosity; and the organic modification of the Ormosil imparts controlled surface polarity. Additionally, hydrophilic chemical modifiers are incorporated in order to control the size of the wetted, electroactive layer; high dispersion of inert metal catalysts is used to enhance hydrogen peroxide oxidation and redox mediators may be co-immobilized when oxygen independent response is desirable. The electrodes can be prepared either in the form of thick supported film, useful for disposable electrodes or as bulk-modified, disk shape electrodes, which can be used as renewable surface electrodes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S.Braun, S.Rappoport, R.Zusman, D.Avnir, and M.Ottolenghi, Mat. Lett. 10, 1 (1990).
D.Avnir, S.Braun, O.Lev, and M.Ottolenghi, Chem. Mater. 6, 1605 (1994).
L.M.Ellerby, C.R.Nishida, F.Nishida, S.A.Yamanaka, B.Dunn, J.S.Valentine, and J.I.Zink, Science 225, 1113 (1992).
B.C. Dave, B. Dunn, J.S. Valentine, and J.I. Zink, Anal. Chem. 66, 1120A (1994).
J.Brinker and G.Scherer Sol-Gel Science (Academic Press, San-Diego, 1989).
G.Philipp and H.Schmidt, J. Non-Cryst. Solids 63, 283 (1984).
Better Ceramics Through Chemistry VI, edited by C. Sanchez, M.L. Mecartney, C.J. Brinker, and A.K. Cheetham (Mater. Res. Soc. Symp. Proc., 1994), earlier volumes in this series.
P.Audebert, C.Demaille, and C.Sanchez, Chem. Mater. 5, 911 (1993).
Y. Tatsu, K. Yamashita, M. Yamaguchi, S. Yamamura, H. Yamamoto, and S. Yoshikawa, Chem. Lett. 1619 (1992).
U.Narang, P.N.Prasad, F.V.Bright, K.Ramanathan, N.D.Kumar, B.D.Malhotra, M.N.Kamalasanan, and S.Chandra, Anal. Chem. 66, 3139 (1994).
Y.Kurokawa, H.Ohta, M.Okubo, and M.Takahashi, Carbohydr. Polym. 23, 1 (1994).
V.Glezer and O.Lev, J. Am. Chem. Soc. 115, 2533 (1993).
I.Pankratov and O.Lev, J. Electroanal. Chem. 393, 35 (1995).
M.Tsionsky, G.Gun, V.Glezer, and O.Lev, Anal. Chem. 66, 1747 (1994).
R.Wilson and A.P.F.Turner, Biosensors and Bioelectronics 7, 165 (1992).
P.D.Hale, L.I.Boguslavsky, T.Inagaki, H.I.Karan, H.S.Lee, and T.A.Skotheim, Anal. Chem. 63, 677 (1991).
S.A.Yamanaka, F.Nishida, L.M.Ellerby, C.R.Nishida, B.Dunn, J.S.Valentine, and J.I.Zink, Chem. Mater. 4, 485 (1992).
G. Gun, M. Tsionsky, and O. Lev, in Better Ceramics Through Chemistry, VI, edited by C. Sanchez, M.L. Mecartney, C.J. Brinker, and A.K. Cheetham (Mater. Res. Soc. Symp. Proc. 1994), pp. 1011–1016.
M.Tsionsky and O.Lev, Anal. Chem. 67, 2409 (1995).
J.Gun, M.Tsionsky, L.Rabinovich, Y.Golan, I.Rubinstein, and O.Lev, J. Electroanal. Chem. 395, 57 (1995).
G.Gun, M.Tsionsky, and O.Lev, Anal. Chim. Acta 294, 261 (1994).
W.J.Albery, P.N.Bartlett, and D.H.Craston, J. Electroanal. Chem. 194, 223 (1985).
A.Heller, Acc. Chem. Res. 23, 128 (1988).
S. Sampath and O. Lev, in preparation.
J.Zagal, R.K.Sen, and E.Yeager, J. Electroanal. Chem. 83, 207 (1977).
M.Tsionsky and O.Lev, J. Electrochem. Soc. 142, 2154 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sampath, S., Pankratov, I., Gun, J. et al. Sol-gel derived ceramic-carbon enzyme electrodes: Glucose oxidase as a test case. J Sol-Gel Sci Technol 7, 123–128 (1996). https://doi.org/10.1007/BF00401892
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00401892