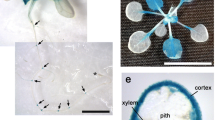
Abstract
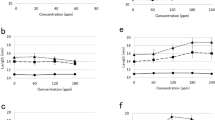
Ethylene treatment (approx. 20 μl ·1-1 in air for 2 d) of tobacco (Nicotiana tabacum L. cv. Havana 425) plants markedly increases the endo-β-1,3-glucanase (EC 3.2.1.39) content of leaves. The antigenic form of the enzyme induced is the same one whose production is blocked by treating cultured cells with combinations of auxin (1.1 · 10-5 M α-naphthaleneacetic acid) and cytokinin (1.4 · 10-6 M kinetin). Evidence is presented that cultured tobacco cells require ethylene for β-1,3-glucanase accumulation: i) ethylene treatment increased the accumulation of \-1,3-glucanase in callus tissues >10 d after subculturing and in cell-suspension cultures; ii) callus tissues can produce ethylene; iii) conditions known to inhibit ethylene production (1 mM CoCl2; 33° C treatment) or ethylene action (approx. 1.6 mmol · 1-1 norbornadiene in air) inhibited β-1,3-glucanase accumulation by callus tissues treated for 4 d following subculturing; and, these inhibitory effects were prevented by exogenous ethylene. Combinations of auxin and cytokinin blocked ethylene-induced accumulation of β-1,3-glucanase by cell-suspension cultures. The results favor a model in which ethylene induces results favor a model in which ethylene induces β 1,3-glucanase accumulation, and auxin and cytokinin inhibit this induction process.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- NAA:
-
α-naphthaleneacetic acid
- NDE:
-
norbornadiene
References
Abeles, F.B., Forrence, L.E. (1970) Temporal and hormonal control of β-1,3-glucanase in Phaseolus vulgaris L. Planta 45, 395–400
Ballance, G.M., Manners, D.J. (1978) Partial purification and properties of an endo-1,3-β-D-glucanase from germinated rye. Phytochemistry. 17, 1539–1543
Bartnicki-Garcia, S. (1968) Cell wall chemistry, morphogenesis, and taxonomy of fungi. Annu. Rev. Microbiol. 22, 87–108
Binns, A., Meins, F., Jr. (1973) Habituation of tobacco pith cells for factors promoting cell division is heritable and potentially reversible. Proc. Natl. Acad. Sci. USA 70, 2660–2662
Boller, T. (1985) Induction of hydrolases as a defense reaction against pathogens. UCLA Symp. Mol. Cell. Biol. N.S. 22, 247–262
Boller, T., Gehri, A., Mauch, F., Voegeli, U. (1983) Chitinase in bean leaves: induction by ethylene, purification, properties, and possible function. Planta 157, 22–31
Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254
Broglie, K., Durance-Tardif, M., Gaynor, J.J., Broglie, R. (1985) Ethylene regulated gene expression: molecular cloning and in vivo expression of chitinase from bean and tomato. In: Abstr. 1st Int. Congr. Plant Molec. Biol., p. 141, Galau, G.A., ed. University of Georgia, Athens, USA
Broglie, K.E., Gaynor, J.J., Broglie, R.M. (1986) Ethylene-regulated gene expression: Molecular cloning of the genes encoding an endochitinase from Phaseolus vulgaris. Proc. Natl. Acad. Sci. USA 83, 6820–6824
Davies, E., Maclachlan, G.A. (1968) Effects of indoleacetic acid on intracellular distribution of β-1,3-glucanase activities in the pea epicotyl. Arch. Biochem. Biophys. 128, 595–600
Eichholz, R., Harper, J., Felix, G., Meins, F., Jr. (1983) Evidence for an abundant 33,000-dalton polypeptide regulated by cytokinins in cultured tobacco tissues. Planta 158, 410–415
Felix, G. (1985) Hormonal regulation of abundant polypeptides with β-1,3-glucanase and chitinase activity in tobacco. Ph.D. Diss., University of Basel, Switzerland
Felix, G., Meins, F., Jr. (1985) Purification, immunoassay and characterization of an abundant, cytokinin-regulated polypeptide in cultured tobacco tissues. Evidence the protein is a β-1,3-glucanase. Planta 164, 423–428
Felix, G., Meins, F., Jr. (1986) Developmental and hormonal regulation of β-1,3-glucanase in tobacco. Planta 167, 206–211
Huxter, T.J., Reid, D.M., Thorpe, T.A. (1979) Ethylene production by tobacco Nicotiana tabacum callus. Physiol. Plant. 46, 374–380
Lau, O.L., Yang, S.F. (1976) Inhibition of ethylene production by cobaltous ion. Plant Physiol. 58, 114–117
Laurell, C.B., McKay, E.J. (1981) Electroimmunoassay. Methods Enzymol. 73, 339–369
Letham, D.S. (1978) Cytokinins. In: Phytohormones and related compounds — A comprehensive treatise, vol. 1, pp. 205–263, Letham, D.S., Goodwin, P.B., Higgins, T.J.V., eds. Elsevier/North-Holland, Amsterdam
Linsmaier, E.M., Skoog, F. (1965) Organic growth factor requirements of tobacco tissue cultures. Physiol. Plant. 18, 100–127
Mauch, F., Hadwiger, L.A., Boller, T. (1984) Ethylene: Symptom, not signal for the induction of, chitinase and β-1,3-glucanase in pea pods by pathogens and elicitors. Plant Physiol. 76, 607–611
Meins, F., Jr. (1975) Temperature-sensitive expression of auxin-autotrophy by crown-gall teratoma cells of tobacco. Planta 122, 1–9
Meins, F., Jr., Lutz, J. (1980) The induction of cytokinin habituation in primary pith explants of tobacco. Planta 149, 402–407
Miller, A.R., Pengelly, W.L. (1984) Ethylene production by shoot-forming and unorganized crown-gall tumor tissues of Nicotiana and Lycopersicon cultured in vitro. Planta 161, 418–424
Mohnen, D., Shinshi, H., Felix, G., Meins, F., Jr. (1985) Hormonal regulation of β-1,3-glucanase messenger RNA levels in cultured tobacco tissues. EMBO J 4, 1631–1635
Moore, A.E., Stone, B.A. (1972) Effect of senescence and hormone treatment on the activity of a β-1,3-glucan hydrolase in Nicotiana glutinosa leaves. Planta 104, 93–109
Pengelly, W.L., Meins, F., Jr. (1983) Growth, auxin requirement, and indole-3-acetic acid content of cultured crown-gall and habituated tissues of tobacco. Differentiation 25, 101–105
Pegg, G.F. (1976) The response of ethylene-treated tomato plants to infection by Verticillium albo-atrum. Physiol. Plant Pathol. 9, 215–226
Pegg, G.F. (1977) Glucanohydrolases of higher plants: A possible defence mechanism against parasitic fungi. In: Cell wall biochemistry related to specificity in host-plant pathogen interactions, pp. 305–345, Solheim, B., Raa, J., eds. Universitetsforlaget, Oslo
Shinshi, H., Mohnen, D., Meins, F., Jr. (1987) Regulation of a plant pathogenesis-related enzyme: Inhibition of chitinase and chitinase mRNA accumulation in cultured tobacco tissues by auxin and cytokinin. Proc. Natl. Acad. Sci. USA 84, 89–93
Sisler, E.C., Yang, S.F. (1984) Anti-ethylene effect of cis-2-butene and cyclic olefins. Phytochemistry 23, 2765–2768
Yu, Y.B., Adams, D.O., Yang, S.F. (1980) Inhibition of ethylene production by 2,4-dinitrophenol and high temperature. Plant Physiol. 66, 268–290
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Felix, G., Meins, F. Ethylene regulation of β-1,3-glucanase in tobacco. Planta 172, 386–392 (1987). https://doi.org/10.1007/BF00398668
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00398668