Abstract
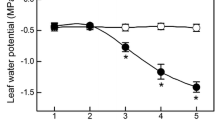
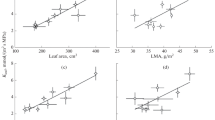
The expansion growth of plant organs is inhibited at low water potentials (Ψ w), but the inhibition has not been compared in different organs of the same plant. Therefore, we determined elongation rates of the roots, stems, leaves, and styles (silks) of maize (Zea mays L.) as soil water was depleted. The Ψ w was measured in the region of cell expansion of each organ. The complicating effects of transpiration were avoided by making measurements at the end of the dark period when the air had been saturated with water vapor for 10 h and transpiration was less than 1% of the rate in the light. Growth was inhibited as the Ψ w in the region of cell expansion decreased in each organ. The Ψ w required to stop growth was-0.50,-0.75, and-1.00 MPa, in this order, in the stem, silks, and leaves. However, the roots grew at these Ψ w and ceased only when Ψ w was lower than-1.4 MPa. The osmotic potential decreased in each region of cell expansion and, in leaves, roots and stems, the decrease was sufficient to maintain turgor fully. In the silks, the decrease was less and turgor fell. In the mature tissue, the Ψ w of the stem, leaves and roots was similar to that of the soil when adequate water was supplied. This indicated that an equilibrium existed between these tissues, the vascular system, and the soil. At the same time, the Ψ w was lower in the expanding regions than in the mature tissues, indicating that there was a Ψ w disequilibrium between the growing tissue and the vascular system. The disequilibrium was interpreted as a Ψ w gradient for supplying water to the enlarging cells. When water was withheld, this gradient disappeared in the leaf because Ψ w decreased more in the xylem than in the soil, indicating that a high flow resistance had developed in the xylem. In the roots, the gradient did not decrease because vascular Ψ w changed about the same amount as the soil Ψ w. Therefore, the gradient in Ψ w favored water uptake by roots but not leaves at low Ψ w. The data show that expansion growth responds to low Ψ w differently in different growing regions of the plant. Because growth depends on the maintenance of turgor for extending the cell walls and the presence of Ψ w gradients for supplying water to the expanding cells, several factors could have been responsible for these differences. The decrease of turgor in the silks and the loss of the Ψ w gradient in the leaves probably contributed to the high sensitivity of these organs. In the leaves, the gradient loss was so complete that it would have prevented growth regardless of other changes. In the roots, the maintenance of turgor and Ψ w gradients probably allowed growth to continue. This difference in turgor and gradient maintenance could contribute to the increase in root/shoot ratios generally observed in water-limited conditions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Ψ s :
-
osmotic potential
- Ψ w :
-
water potential
References
Acevedo, E., Fereres, E., Hsiao, T.C., Henderson, D.W. (1979) Diurnal growth trends, water potential, and osmotic adjustment of maize and sorghum leaves in the field. Plant Physiol. 64, 476–480
Acevedo, E., Hsiao, T.C., Henderson, D.W. (1971) Immediate and subsequent growth responses of maize leaves to changes in water status. Plant Physiol. 48, 631–636
Ayers, P.G. (1981) Root growth and solute accumulation in pea in response to water stress and powdery mildew. Physiol. Plant Pathol 19, 169–180
Barrs, H.D. (1965) Comparison of water potentials in leaves as measured by two types of thermocouple psychrometers. Aust. J. Biol. Sci. 18, 36–52
Blizzard, W.E., Boyer, J.S. (1980) Comparative resistance of the soil and the plant to water transport. Plant Physiol. 66, 809–814
Boyer, J.S. (1967) Leaf water potentials measured with a pressure chamber. Plant Physiol. 42, 133–137
Boyer, J.S. (1968) Relationship of water potential to growth of leaves. Plant Physiol. 43, 1056–1062
Boyer, J.S. (1970) Leaf enlargement and metabolic rates in corn, soybeans, and sunflower at various water potentials. Plant Physiol 46, 233–235
Boyer, J.S. (1971) Recovery of photosynthesis in sunflower after a period of low leaf water potential. Plant Physiol. 47, 816–820
Boyer, J.S. (1974) Water transport in plants: mechanism of apparent changes in resistance during absorption. Planta 117, 187–207
Boyer, J.S., Knipling, E.B. (1965) Isopiestic technique for measuring leaf water potentials in leaves with a thermocouple psychrometer. Proc. Natl. Acad. Sci. USA 54, 1049–1051
Boyer, J.S., Wu, G. (1978) Auxin increases the hydraulic conductivity of auxin-sensitive hypocotyl tissue. Planta 139, 227–237
Bunce, J.A. (1977) Leaf elongation in relation to leaf water potential in soybeans. J. Exp. Bot. 28, 156–161
Cavalieri, A.J., Boyer, J.S. (1982) Water potentials induced by growth in soybean hypocotyls. Plant Physiol. 69, 492–496
Chu, A.C.P., McPherson, H.G. (1977) Sensitivity to desiccation of leaf extension in prairie grass. Aust. J. Plant Physiol. 4, 381–387
Cosgrove, D.J. (1981) Analysis of the dynamic and steady-state responses of growth rate and turgor pressure to changes in cell parameters. Plant Physiol. 68, 1439–1446
Cosgrove, D.L., Cleland, R.E. (1983) Solutes in the free space of growing stem tissue. Plant Physiol. 72, 326–331
Cosgrove, D.L., Van Volkenburgh, E., Cleland, R.E. (1984) Stress relaxation of cell walls and the yield threshold for growth. Planta 162, 46–54
Cutler, J.M., Rains, D.W. (1977) Effect of irrigation history on responses of cotton to subsequent water stress. Crop Sci. 17, 329–335
Cutler, J.M., Rains, D.W., Loomis, R.S. (1977) Role of changes in solute concentration in maintaining favorable water balance in field-grown cotton. Agron. J. 69, 773–779
Cutler, J.M., Shannon, K.W., Steponkus, P.L. (1980) Influence of water deficits and osmotic adjustment on leaf elongation in rice. Crop Sci. 20, 314–318
El Nadi, A.H., Brouwer, R., Locker, J.Th. (1969) Some responses of the root and the shoot of Vicia faba plants to water deficits. Neth. J. Agric. Sci. 17, 133–142
Gates, C.T. (1955) The response of the young tomato plant to a brief period of water shortage. Aust. J. Biol. Sci. 8, 196–214
Greacen, E.L., Oh, J.S. (1972) Physics of root growth. Nature New Biol. 235, 24–25
Herrero, M.P., Johnson, R.R. (1981) Drought stress and its effect on maize reproductive systems. Crop Sci. 21, 105–110
Hoagland, D.R., Arnon, D.I. (1950) The water culture method for growing plants without soil. Calif. Agric. Exp. Stat. Circ. 347
Hoffman, G.T., Rawlins, S.L., Garber, M.J., Cullen, E.M. (1971) Water relations and growth of cotton as influenced by salinity and relative humidity. Agron. J. 63, 822–826
Hsiao, T.C., Acevedo, E. (1974) Plant responses to water deficits, water-use efficiency, and drought resistance. Agric. Meteorol. 14, 59–84
Hsiao, T.C., Acevedo, E., Fereres, E., Henderson, D.W. (1976) Water stress, growth, and osmotic adjustment. Phil. Trans. R. Soc. London B 273, 479–500
Jones, M.M., Turner, N.C. (1978) Osmotic adjustment in leaves of sorghum in response to water deficits. Plant Physiol. 61, 122–126
Kiesselback, T.A. (1949) The structure and reproduction of corn. Univ. of Nebraska Coll. Agric., Agr. Exp. Stat. Res. Bull. No. 161
Lockhart, J.A. (1965) An analysis of irreversible cell extension. J. Theor. Biol. 8, 264–275
Matsuda, K., Riazi, A. (1981) Stress-induced osmotic adjustment in growing regions of barley leaves. Plant Physiol. 68, 571–576
Meyer, R.F., Boyer, J.S. (1972) Sensitivity of cell division and cell elongation to low water potentials in soybean hypocotyls. Planta 108, 77–87
Meyer, R.F., Boyer, J.S. (1981) Osmoregulation, solute distribution, and growth in soybean seedlings having low water potentials. Planta 151, 482–489
Michelena, V.A., Boyer, J.S. (1982) Complete turgor maintenance at low water potentials in the elongation region of maize leaves. Plant Physiol. 69, 1145–1149
Milburn, J.A. (1966) The conduction of sap. I. Water conduction and cavitation in water stressed leaves. Planta 69, 34–42
Molz, F.L., Boyer, J.S. (1978) Growth-induced water potentials in plant cells and tissues. Plant Physiol. 62, 423–429
Munns, R., Brady, C.J., Barlow, E.W.R. (1979) Solute accumulation in the apex and leaves of wheat during water stress. Aust. J. Plant Physiol. 6, 379–389
Munns, R., Wier, R. (1981) Contribution of sugars to osmotic adjustment in elongating and expanded zones of wheat leaves during moderate water deficits at two light levels. Aust. J. Plant Physiol. 8, 93–105
Ray, P.M., Green, P.B., Cleland, R. (1972) Role of turgor in plant cell growth. Nature 239, 163–164
Read, D.J., Bartlett, E.M. (1972) The physiology of drought resistance in soybean plants (Glycine max). J. Appl. Ecol. 9, 481–499
Sharp, R.E., Davies, W.J. (1979) Solute regulation and growth by roots and shoots of water stressed maize plants. Planta 147, 43–49
Takami, S., Turner, N.C., Rawson, H.M. (1981) Leaf expansion of four sunflower (Helianthus annuus L.) cultivars in relation to water deficits. 1. Pattern during plant development. Plant Cell Environ. 4, 399–407
Westgate, M.E., Boyer, J.S. (1984) Transpiration-and growth-induced water potentials in maize. Plant Physiol. 74, 882–889
Westgate, M.E., Boyer, J.S. (1985) Carbohydrate reserves and reproductive development at low leaf water potentials in maize. Crop Science, in press
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Westgate, M.E., Boyer, J.S. Osmotic adjustment and the inhibition of leaf, root, stem and silk growth at low water potentials in maize. Planta 164, 540–549 (1985). https://doi.org/10.1007/BF00395973
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00395973