Abstract
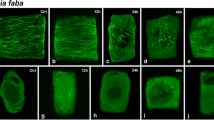
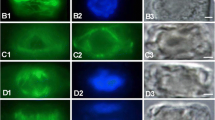
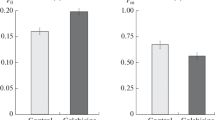
The effects of oryzalin, a dinitroaniline herbicide, on chromosome behavior and on cellular microtubules (MTs) were examined by light microscopy and immunogold staining, respectively, in endosperm cells from Haemanthus katherinae Bak. Brief treatments with 1.0·10-8 M oryzalin reduced markedly the migration rate of anaphase chromosomes and 1.0·10-7 M oryzalin stopped migration abruptly. Oryzalin (1.0·10-7 M) depolymerized MTs and prevented the polymerization of new MTs at all stages of the mitotic cycle. The chromosome condensation cycle was unaffected by oryzalin. Endothelial cells from the heart of Xenopus leavis showed no chromosomal or microtubular rearrangements after oryzalin treatment. The inhibition by oryzalin of the polymerization of tubulin isolated from cultured cells of Rosa sp. cv. Paul's scarlet was examined in vitro by turbidimetry, electron microscopy and polymer sedimentation analysis. Oryzalin inhibited the rapid phase of taxol-induced polymerization of rose MTs at 24°C with an apparent inhibition constant (K i ) of 2.59·106 M. Shorter and fewer MTs were formed with increasing oryzalin concentrations, and maximum inhibition of taxol-induced polymerization occurred at approx. 1:1 molar ratios of oryzalin and tubulin. Oryzalin partially depolymerized taxol-stabilized rose MTs. Ligand-binding experiments with [14C]oryzalin demonstrated the formation of a tubulin-oryzalin complex that was time- and pH-dependent. The tubulin-oryzalin interaction (24°C, pH 7.1) had an apparent affinity constant (K app) of 1.19·105 M-1. Oryzalin did not inhibit taxol-induced polymerization of bovinebrain MTs and no appreciable binding of oryzalin to brain tubulin or other proteins was detected. The results demonstrate pharmacological differences between plant and animal tubulins and indicate that the most sensitive mode of action of the dinitroaniline herbicides is the direct poisoning of MT dynamics in cells of higher plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- MT:
-
microtubule
- SIB:
-
sucrose isolation buffer
- TO:
-
tubulin-oryzalin complex
References
Ashton, F.M., Crafts, A.S. (1981) Mode of action of herbicides. wiley Interscience, New York
Ashton, F.M., de Villiers, O.T., Glenn, R.K., Duke, W.B. (1977) Localization of metabolic sites of action of herbicides. Pestic. Biochem. Physiol. 7, 122–141
bajer, A.S., Cypher, C., Molè-Bajer, J., Howard, H.M. (1982) Taxol-induced anaphase reversal: Evidence that elongating microtubules can exert a pushing force in living cells. Proc. Natl. Acad. Sci. USA 79, 6569–6573
Bajer, A.S., Molè-Bajer, J. (1986) Drugs with colchicine-like effects that specifically disassemble plant but not animal microtubules. N.Y. Acad. Sci. 466, 767–784
Bane, S., Puett, D., Macdonald, T.L., Williams, R.C., Jr. (1984) Binding to tubulin of the colchicine analog 2-methoxy-5-(2′, 3′,4′,-trimethoxyphenyl)tropone. J. Biol. Chem. 259, 7391–7398
Bartels, P.G., Hilton, J.L. (1973) Comparison of trifluralin, oryzalin, pronamide, propham, and colchicine treatments on microtubules. Pestic. Biochem. Physiol. 3, 462–472
Bayer, D.E., Foy, C.L., Mallory, T.E., Cutter, E.G. (1967) Morphological and histological effects of trifluralin on root development. Am J. Bot. 54, 945–952
Borisy, G.G. (1972) A rapid method for quantitative determination of microtubule protein using DEAE-cellulose filters. Anal. Biochem. 50, 373–385
De Mey, J., Lambert, A.M., Bajer, A.S., Moeremans, M., De-Brabander, M. (1982) Visualization of microtubules in interphase and mitotic plant cells of Haemanthus endosperm with the immuno-gold staining method. Proc. Natl. Acad. Sci. USA 79, 1898–1902
Dustin, P. (1984) Microtubules. Springer Verlag, Berlin
Fedtke, C. (1982) Biochemistry and physiology of herbicide action. Springer Verlag, Berlin
Fitzgerald, T.J. (1976) Molecular features of colchicine associated with antimitotic activity and inhibition of tubulin polymerization. Biochem. Pharmacol. 25, 1383–1387
Gaskin, F., Cantor, C.R., Shelanski, M.L. (1974) Turbidometric studies of the in vitro assembly and disassembly of porcine neurotubules. J. Mol. Biol. 80, 737–758
Gunning, B.E.S., Hardham, A.R. (1982) Microtubules. Annu. Rev. Plant Physiol. 33, 651–698
Hertel, C., Marmé, D. (1983) Herbicides and fungicides inhibit Ca2+ uptake by plant mitochondria: a possible mechanism of action. Pestic. Biochem. Physiol. 19, 282–290
Hertel, C., Quader, H., Robinson, D.G., Marmé, D. (1980) Anti-microtubular herbicides and fungicides affect Ca2+ transport in plant mitochondria. Planta 149, 336–340
Hess, F.D. (1979) The influence of the herbicide trifluralin on flagellar regeneration in Chlamydomonas. Exp. Cell Res. 119, 99–109
Hess, F.D., Bayer, D.E. (1974) The effect of trifluralin on the ultrastructure of dividing cells of the root meristem of cotton (Gossypium hirsutum L. Acala 4-42′) J. Cell Sci. 15, 429–441
Hess, F.D., Bayer, D.E. (1977) Binding of the herbicide trifluralin to Chlamydomonas flagellar tubulin. J. Cell Sci. 24, 351–360
Jackson, W.T., Stetler, D.A. (1973) Regulation of mitosis. IV. An in vitro and ultrastructural study of effects of trifluralin. Can. J. Bot. 51, 1513–1518
Johnson, K.A., Borisy, G.G. (1975) The equilibrium assembly of microtubules in vitro. In: Molecules and cell movement, pp. 119–141, Inoue, S., Stephens, R.E., eds., Raven Press, New York
Karr, T.L., Purich, D.L. (1979) A microtubule assembly/disassembly model based on drug effects and depolymerization kinetics after rapid dilution. J. Biol. Chem. 254, 10885–10888
Klotz, I.M. (1982) Number of receptor sites from Scatchard graphs: facts and fantacies. Science 217, 1247–1249
Kumar, N. (1981) Taxol-induced polymerization of purified tubulin. J. Biol. Chem. 256, 4156–4160
Lee, J.C., frigon, R.P., Timasheff, S.N. (1973) The chemical characterization of calf brain microtubule protein subunits. J. Biol. Chem. 248, 7253–7262
Margolis, R.L., Wilson, L. (1977) Addition of colchicine-tubulin complex to microtubule ends: The mechanism of substoichiometric colchicine poisoning. Proc. Natl. Acad. Sci. USA 74, 3466–3470
Molè-Bajer, J. (1958) Cine-micrographic analysis of c-mitosis in endosperm. Chromosoma 9, 332–358
Molè-Bajer, J., Bajer, A. (1968) Studies of selected endosperm cells with the light and electron microscope. Cellule 67, 257–265
Morejohn, L.C., Bureau, T.E., Fosket, D.E. (1985) Inhibition of plant cell proteolytic activities that degrade tubulin. Cell Biol. Int. Rep. 9, 849–857
Morejohn, L.C., Bureau, T.E., Tocchi, L.P., Fosket, D.E. (1984) Tubulins from different higher plant species are immunologically nonidentical and bind colchicine differentially. Proc. Natl. Acad. Sci. USA 81, 1440–1444
Morejohn, L.C., Bureau, T.E., Tocchi, L.P., Fosket, D.E. (1987) Resistance of Rosa microtubule polymerization to colchicine results from a low-affinity interaction of colchicine and tubulin. Planta 170, 230–241
Morejohn, L.C., Fosket, D.E. (1982) Higher plant tubulin identified by self-assembly into microtubules in vitro. Nature 297, 426–428
Morejohn, L.C., Fosket, D.E. (1984a) Taxol-induced rose microtubule polymerization in vitro and its inhibition by colchicine. J. Cell Biol. 99, 141–147
Morejohn, L.C., Fosket, D.E. (1984b) Inhibition of plant microtubule polymerization in vitro by the phosphoric amide herbicide amiprophos-methy. Science 224, 874–876
Morejohn, L.C., Fosket, D.E. (1986) Tubulins from plants, fungi and protists: a review. In: Cell and molecular biology of the cytoskeleton, pp. 257–329, Shay, J.W., ed. Plenum Publishing Corp. New York
Moreland, D.E., Farmer, F.S., Hussey, G.G. (1972a) Inhibition of photosynthesis and respiration by substituted 2,6-dinitroaniline herbicides. I. Effects on chloroplast and mitochondrial activities. Pestic. Biochem. Physiol. 2, 342–353
Moreland, D.E., Farmer, F.S., Hussey, G.G. (1972b) Inhibition of photosynthesis and respiration by substituted 2,6-dinitroaniline herbicides. II. Effects on responses in excised plant tissues and treated seedlings. Pestic. Biochem. Physiol. 2, 354–363
Mudge, L.C., Gossett, B.J., Murphy, T.R. (1984) Resistance of goosegrass (Eleusine indica) to dinitroaniline herbicides. Weed Sci. 32, 591–594
Olmsted, J.B., Borisy, G.G. (1973) Characterization of microtubule assembly in porcine brain extracts by viscometry. Biochemistry 12, 4282–4289
Ray, K., Bhattacharyya, B., Biswas, B.B. (1981) Role of B-ring of colchicine in its binding to tubulin. J. Biol. Chem. 256, 6241–6244
Rieder, C.L., Bajer, A.S. (1978) Effect of elevated temperatures on spindle microtubules and chromosome movements in cultured newt lung cells. Cytobios 18, 201–234
Robinson, D.G., Herzog, W. (1977) Structure, synthesis and orientation of microfibrils. III. A survey of the action of microtubule inhibitors on microtubules and microfibril orientation in Oocystis solitaria. Cytobiologie 15, 463–474
Robinson, D.G., Quader, H. (1982) The microtubule-microfibril syndrome. In: The cytoskeleton in plant growth and development, pp. 109–126, Lloyd, C. ed. Academic Press, London
Scatchard, G. (1949) The interactions of proteins for small molecules and ions. Ann. N.Y. Acad. Sci. 51, 660–672
Schiff, P.B., Horwitz, S.B. (1980) Taxol stabilizes microtubules in mouse fibroblast cells. Proc. Natl. Acad. Sci. USA 77, 1561–1565
Schiff, P.B., Horwitz, S.B. (1981) Taxol assembles tubulin in the absence of exogenous guanosine 5′-triphosphate or microtubule associated proteins. Biochemistry 20, 3247–3252
Shelanski, M.L., Gaskin, F., Cantor, C.A. (1973) Microtubule assembly in the absence of added nucleotides. Proc. Natl. Acad. Sci. USA 70, 765–768
Sternlicht, H., Ringel, I. (1970) Colchicine inhibition of microtubule assembly via copolymer formation. J. Biol. Chem. 254, 10540–10550
Strachen, S.D., Hess, F.D. (1982) Dinitroaniline herbicides adsorb to glass. J. Agric. Food Chem. 30, 389–391
Strachen, S.D., Hess, F.D. (1983) The biochemical mechanism of action of the dinitroaniline berbicide oryzalin. Pestic. Biochem. Physiol. 20, 141–150
Upadhyaya, M.K., Noodén, L.D. (1980) Mode of dinitroaniline action. II. Characterization of [14C]oryzalin uptake and binding. Plant Physiol. 66, 1048–1052
Vaughn, K.C. (1986a) Cytological studies of dinitroaniline-resistant Eleusine. Pestic. Biochem. Physiol. 26, 66–74
Vaughn, K.C. (1986b) Dinitroaniline resistance in goosegrass (Eleusine indica (L.) Gaertner.) is due to an altered tubulin. (Abstr.) Weed Sci. Soc. Am. 26, a 77
Vaughn, K.C., Marks, M.D., Weeks, D.P. (1987) A dinitroaniline-resistant mutant of Eleusine indica exhibits cross-resistance and super-sensitivity to antimicrotubule herbicides and drugs. Plant Physiol. 83, 956–964
Wilson, L., Anderson, K., Chin, D. (1976) Nonstoichiometric poisoning of microtubule polymerization: A model for the mechansim of action of the vinca alkaloids, podophyllotoxin and colchicine. In: Cold Spring Harbor Conf. on Cell Proliferation (Book C), pp. 1051–1064, Goldman, R., Pollard, T., Rosenbaum, J., eds. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y., USA
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Morejohn, L.C., Bureau, T.E., Molè-Bajer, J. et al. Oryzalin, a dinitroaniline herbicide, binds to plant tubulin and inhibits microtubule polymerization in vitro. Planta 172, 252–264 (1987). https://doi.org/10.1007/BF00394595
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00394595