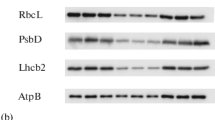
Abstract
In a preceding paper (Oelmüller and Mohr 1986, Planta 167, 106–113) it was shown that in the cotyledons of the mustard (Sinapis alba L.) seedling the integrity of the plastid is a necessary prerequisite for phytochrome-controlled appearance of translatable mRNA for the nuclear-encoded small subunit (SSU) of ribulose-1,5-bisphosphate carboxylase and the light-harvesting chlorophyll a/b-binding protein of photosystem II (LHCP). It was concluded that a signal from the plastid is essential for the expression of nuclear genes involved in plastidogenesis. The present study was undertaken to characterize this postulated signal. Chloramphenicol, an inhibitor of intraplastidic protein synthesis and Norflurazon, an inhibitor of carotenoid synthesis (to bring about photooxidative sensitivity of the plastids) were applied. We obtained the following major results. (i) After a brief period of photooxidative damage a rapid decrease of the above translatable mRNAs was observed. Conclusion: the signal is short-lived and thus required continually. (ii) Once the plastids became damaged by photooxidation, no recovery with regard to nuclear gene expression was observed after a transfer to non-damaging light conditions. Conclusion: even a brief period of damage suffices to prevent production of the signal. (iii) Chloramphenicol inhibited nuclear gene expression (SSU, LHCP) and plastidic development when applied during the early stages of plastidogenesis. Once a certain stage had been reached (between 36–48 h after sowing at 25° C) nuclear gene expression became remarkably insensitive toward inhibition of intraplastidic translation. Conclusion: a certain developmental stage of the plastid must be reached before the signal is released by the plastid. (iv) Under the growth conditions we adopted in our experiments the plastids in the mesophyll cells of mustard cotyledons developed essentially between 36 and 120 (-144) h after sowing. Only during this period could translatable mRNAs for SSU and LHCP be detected. Conclusion: the signal is released by the plastids only during this time span.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CAP:
-
Chloramphenicol (D-threo)
- cFR:
-
continuous far-red light
- FR:
-
far-red light (3.5 W·m-2)
- GPD:
-
glyceraldehyde-3-phosphate dehydrogenase
- LHCP:
-
light-harvesting chlorophyll a/b-binding protein of photosystem II
- LSU:
-
large subunit of RuBPCase
- MDH:
-
malate dehydrogenase
- NF:
-
Norflurazon
- NIR:
-
nitrite reductase
- Pfr:
-
physiologically active form of phytochrome
- R:
-
red light (6.8 W·m-2)
- RG9-light:
-
long-wavelength far-red light (10 W·m-2)
- RuBPCase:
-
ribulose-1,5-bisphosphate carboxylase
- SSU:
-
small subunit of RuBPCase
- WLs :
-
strong white light (28 W·m-2)
- \(\varphi _\lambda = \frac{{[Pfr]\lambda }}{{[Ptot]}}\) :
-
photoequilibrium of phytochrome at wavelength λ
References
Allsop, D., Atkinson, Y.E., Bradbeer, J.W. (1981) The effect of a high non-permissive temperature on chloroplast development in barley. In: Photosynthesis V. Chloroplast development, pp 787–795 Akoyunoglou, G., ed. Balaban. Int. Sci. Services, Philadelphia
Berry-Lowe, S., Meagher, R. (1985) Transciptional regulation of a gene encoding the small subunit of ribulose-1.5-bisphosphate carboxylase in soybean tissue is linked to the phytochrome response. Mol. Cell. Biol. 5, 1910–1917
Biekmann, S., Feierabend, J. (1985) Synthesis and degradation of unassembled polypeptides of the coupling factor of photophosphorylation CF1 in 70 S ribosome-deficient rye leaves. Eur. J. Biochem. 152, 529–535
Börner, T., Metzlaff, M., Koch-Siemenroth, A., Steiner, K., Hagemann, R. (1985) Chloroplast control of the accumulation of nuclear-DNA encoded proteins. 1st Int. Congr. Plant Mol. Biol. Savannah, USA
Bradbeer, W. (1981) Chloroplast development: A perspective. In: Photosynthesis V. Chloroplast development, pp. 745–754, Akoyunoglou, G., ed. Balaban Int. Sci. Services, Philadelphia
Bradbeer, W., Atkinson, Y., Börner, T., Hagemann, R. (1979) Cytoplasmic synthesis of plastid polypeptides may be controlled by plastid-synthesized RNA. Nature 279, 816–817
Britsch, L., Grisebach, H. (1985) Improved preparation and assay of chalcone synthase. Phytochemistry 24, 1975–76
Brödenfeldt, R., Mohr, H. (1986) Use of immunotitration to demonstrate phytochrome-mediated synthesis de novo of chalcone synthase and phenylalanine ammonia lyase in mustard seedling cotyledons. Z. Naturforsch. 41 c, 61–68
Douce, R., Joyard, J. (1984) The regulatory role of the plastid envelope during development. In: Chloroplast Biogenesis, pp. 71–132, Baker, N.R., Barber, J. eds. Elsevier, Amsterdam
Eckes, P., Schell, J., Willmitzer, L. (1985) Organ-specific expression of three leaf/stem specific cDNAs from potato is regulated by light and correlated with chloroplast development. Mol. Gen. Genet. 199, 216–224
Feierabend, J. (1979) Role of cytoplasmic protein synthesis and its coordination within the plastidic protein synthesis in the biogenesis of chloroplasts. Ber. Dtsch. Bot. Ges. 92, 553–574
Frosch, S., Bergfeld, R., Mohr, H. (1976) Light control of plastogenesis and ribulosebisphosphate carboxylase levels in mustard seedling cotyledons. Planta 133, 53–56
Frosch, S., Jabben, M., Bergfeld, R., Keinig, H., Mohr, H. (1979) Inhibition of carotenoid biosynthesis by the herbicide SAN 9789 and its consequences for the action of phytochrome on plastogenesis. Planta 145, 497–505
Gottmann, K., Schäfer, E. (1982) In vitro synthesis of phytochrome apoprotein directed by mRNA from light and dark grown Avena seedlings. Photochem. Photobiol. 35, 521–525
Hagemann, R., Börner, T. (1978) Plastid ribosome-deficient mutants of higher plants as a tool in studying chloroplast biogenesis. In: Chloroplast development, pp. 709–719, Akoyunoglou, G., Argyroudi-Akoyunoglou, J.H. eds. Elsevier, Amsterdam
Harpster, M.H., Mayfield, S.P., Taylor, W.C. (1984) Effects of pigment-deficient mutants on the accumulation of photosynthetic proteins in maize. Plant Mol. Biol. 3, 59–71
Hock, B. (1973) Isoenzyme der Malat-Dehydrogenase aus Wassermelonenkeimlingen: Mikroheterogenität und deren Aufhebung bei der Samenkeimung. Planta 110, 329–344
Link, G., (1982) Phytochrome control of plastid mRNA in mustard (Sinapis alba L.). Planta 154, 81–86
Löhr, G.W., Waller, H.D. (1974) Glucose-6-phosphate-Dehydrogenase. In: Methoden der enzymatischen Analyse, pp. 673–681, Bergmeyer, H.U., ed. Verlag Chemie, Weinheim
Mohr, H. (1966) Untersuchungen zur phytochrominduzierten Photomorphogenese des Senfkeimlings (Sinapis alba L.) Z. Pflanzenphysiol. 54, 63–85
Mohr, H. (1972) Lectures on photomorphogenesis. Springer, Berlin Heidelberg New York
Mohr, H. (1984) Phytochrome and chloroplast development. In: Chloroplast biogenesis. pp. 305–347, Baker, N.R., Barber, J. eds. Elsevier, Amsterdam
Mösinger, E., Batschauer, A., Schäfer, E., Apel, K. (1985) Phytochrome control of in vitro transcription of specific genes in isolated nuclei from barley (Hordeum vulgare). Eur. J. Biochem. 147, 137–142
Oelmüller, R., Mohr, H. (1984) Specific action of blue light on phytochrome-mediated enzyme syntheses in the shoot of milo (Sorghum vulgare Pers.). Plant Cell Environ. 8, 27–31
Oelmüller, R., Mohr, H. (1986) Photooxidative destruction of chloroplasts and its consequences for expression of nuclear genes. Planta 167, 106–113
Rajasekhar, V.K., Mohr, H. (1986) Appearance of nitrite reductase in cotyledons of the mustard (Sinapis alba L.) seedling as affected by nitrate, phytochrome and photo-oxidative damage of plastids. Planta, in press
Reiß, T., Bergfeld, R., Link, G., Thien, W. Mohr, H. (1983) Photooxidative destruction of chloroplasts and its consequences of cytosolic enzyme levels and plant development. Planta 159, 518–528
Sautter, C., Hock, B. (1982) Fluorescence immunohistochemical localisation of malate dehydrogenase isoenzymes in watermelon cotyledons. Plant Physiol. 70, 1162–1168
Sharma, R., Schopfer, P. (1982) Sequential control of phytochrome-mediated synthesis de novo of β-amylase in the cotyledons of mustard (Sinapis alba L.) seedlings. Planta 155, 183–189
Silverthorne, J., Tobin, E.M. (1984) Demonstration of transcriptional regulation of specific genes by phytochrome action. Proc. Natl. Acad. Sci. USA 81, 1112–1118
Strzalka, K., Kwiatowska, M. (1979) Transport of proteins from cytoplasma into plastids in chloramaphenicol-treated bean leaf discs. Autoradiographic Evidence. Planta 146, 393–398
Suissa, M. (1983) Spectrophotometric quantitation of silver grains eluted from autoradiograms. Anal. Biochem. 133, 511–514
Taylor, W.C., Mayfield, S.P., Martineau, B. (1984) The role of chloroplast development in nuclear gene expression. In: Molecular biology of development, pp. 601–610, Davidson, E.H., Firtel, R.A., eds. New York
Tobin, E., Silverthorne, J. (1985) Light regulation of gene expression in higher plants. Annu. Rev. Plant Physiol. 36, 569–593
Vernet, T., Fleck, J., Durr, A., Fritsch, C., Pinck, M., Hirth, L. (1982) Expression of the gene coding for the small subunit of ribulosebisphosphate carboxylase during differentiation of tobacco plant protoplasts. Eur. J. Biochem. 126, 489–494
Wagner, E., Bienger, I., Mohr, H. (1967) Die Steigerung der durch Phytochrom bewirkten Anthocyansynthese des Senfkeimlings (Sinapis alba L.) durch Chloramphenicol. Planta 75, 1–9
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oelmüller, R., Levitan, I., Bergfeld, R. et al. Expression of nuclear genes as affected by treatments acting on the plastids. Planta 168, 482–492 (1986). https://doi.org/10.1007/BF00392267
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00392267