Abstract
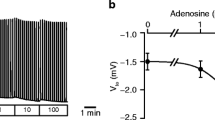
The pancreatic duct has been regarded as a typical cAMP-regulated epithelium, and our knowledge about its Ca2+ homeostasis is limited. Hence, we studied the regulation of intracellular calcium, [Ca2+]i, in perfused rat pancreatic ducts using the Ca2+-sensitive probe fura-2. In some experiments we also measured the basolateral membrane voltage, V bl, of individual cells. The resting basal [Ca2+]i was relatively high, corresponding to 263±28 nmol/l, and it decreased rapidly to 106±28 nmol/l after removal of Ca2+ from the bathing medium (n=31). Carbachol increased [Ca2+]i in a concentration-dependent manner. At 10 μmol/l the fura-2 fluorescence ratio increased by 0.49±0.06 (n=24), corresponding to an increase in [Ca2+]i by 111±15 nmol/l (n=17). ATP, added to the basolateral side at 0.1 mmol/l and 1 mmol/l, increased the fluorescence ratio by 0.67±0.06 and 1.01±14 (n=46; 12), corresponding to a [Ca2+]i increase of 136±22 nmol/l and 294±73 nmol/l respectively (n= 15; 10). Microelectrode measurements showed that ATP (0.1 mmol/l) hyperpolarized V bl from −62±3 mV to-70±3 mV, an effect which was in some cases only transient (n=7). This effect of ATP was different from that of carbachol, which depolarized Vbl. Applied together with secretin, ATP delayed the secretin-induced depolarization and prolonged the initial hyperpolarization of V bl (n=4). Several other putative agonists of pancreatic HCO −3 secretion were also tested for their effects on [Ca2+]i. Bombesin (10 nmol/l) increased the fura-2 fluorescence ratio by 0.24±0.04 (n=8), neurotensin (10 nmol/l) by 0.25±0.04 (n=6), substance P (0.1 μmol/l) by 0.22±0.06 (n=6), and cholecystokinin (10 nmol/l) by 0.14±0.03 (n=7). Taken together, our studies show that Ca2+ homeostasis plays a role in pancreatic ducts. The most important finding is that carbachol and ATP markedly increase [Ca2+]i, but their different electrophysiological responses indicate that intracellular signalling pathways may differ.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Argent BE, Arkle S, Cullen MJ, Green R (1986) Morphological, biochemical and secretory studies on rat pancreatic ducts maintained in tissue culture. Q J Exp Physiol 71: 633–648
Ashton N, Argent BE, Green R (1990) Effect of vasoactive intestinal peptide, bombesin and substance P on fluid secretion by isolated rat pancreatic ducts. J Physiol (Lond) 427: 471–482
Dissing S, Nauntofte B, Sten-Knudsen O (1990) Spatial distribution of intracellular, free Ca2+ in isolated rat parotid acini. Pflügers Arch 417: 1–12
Docray GJ (1973) Vasoactive intestinal peptide: secretin-like action on the avian pancreas. Experientia 29: 1510–1511
Evans RL, Ashton N, Elliot AC (1990) Intracellular [Ca2+]i in rat pancreatic ducts is increased by acetylcholine but not by bombesin or substance P. J Physiol (Lond) 429: 90P
Evans RL, Ashton N, Elliot AC, Green R (1992) Effect of acetylcholine on intracellular calcium and fluid secretion in isolated rat pancreatic ducts. J Physiol (Lond) 446: 352P
Fölsch UR, Fischer H, Sölling HD, Creutzfeldt W (1980) Effects of gastrointestinal hormones and carbamylcholine on cAMP accumulation in isolated pancreatic duct fragments from the rat. Digestion 20: 277–292
Gallacher DV, Hanley MR, Petersen OH, Roberts ML, SquirePollard LG, Yule DI (1990) Substance P and bombesin elevate cytosolic Ca2+ by different molecular mechanisms in a rat pancreatic acinar cell line. J Physiol (Lond) 426: 193–207
Gray MA, Greenwell JR, Argent BE (1988) Secretin-regulated chloride channel on the apical plasma membrane of pancreatic duct cells. J Membr Biol 105: 131–142
Gray MA, Greenwell JR, Garton AJ, Argent BE (1990) Regulation of maxi-K+ channels on pancreatic duct cells by cyclic AMP-dependent phosphorylation. J Membr Biol 115:203–215
Grynkiewicz G, Poenie M, Tsien RY (1985) A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem 260: 3440–3450
Habara Y, Kanno T (1991) Dose-dependency in spatial dynamics of [Ca2+]c in pancreatic acinar cells. Cell Calcium 12:533–542
Kasai H, Augustine GJ (1990) Cytosolic Ca2+ gradients triggering unidirectional fluid secretion from exocrine pancreas. Nature 348: 735–738
Klär B, Leipziger J, Nitschke R, Greger R (1993) Ca2+ as a second messenger in CFPAC-1 cells. Cell Physiol Biochem 3: 17–27
Konturek SJ, Domschke S, Domschke W, Wünsch E, Demling L (1977) Comparison of pancreatic responses to portal and systemic secretin and VIP in cats. Am J Physiol 232: E156-E158
Kunzelmann K, Kubitz R, Grolik M, Warth R, Greger R (1992) Small-conductance Cl− channels in HT29 cells: activation by Ca2+, hypotonic swelling and 8-Br-cGMP. Pflügers Arch 421: 238–246
Lingard JM, A1-Nakkash L, Argent BE (1991) Effect of gut hormones and adenosine compounds on 125I efflux from human pancreatic adenocarcinoma cells (BxPC-3). Proc Aust Physiol Pharmacol Soc 22: 159P
Madden ME, Sarras MP Jr (1989) The pancreatic ductal system of the rat: cell diversity, ultrastructure, and innervation. Pancreas 4: 472–485
Nitschke R, Fröbe U, Greger R (1991) Antidiuretic hormone acts via V1 receptors on intracellular calcium in the isolated perfused rabbit cortical thick ascending limb. Pflügers Arch 417: 622–632
Novak I (1988) Pancreatic bicarbonate secretion. In: Häussinger D (ed) pH homeostasis — mechanism and control. Academic Press, London, pp 447–470
Novak I, Greger R (1988) Properties of the luminal membrane of isolated perfused rat pancreatic ducts: effect of cyclic AMP and blockers of chloride transport. Pflügers Arch 411: 546–553
Novak I, Greger R (1988) Electrophysiological study of transport systems in isolated perfused pancreatic ducts: properties of the basolateral membrane. Pflügers Arch 411: 58–68
Novak I, Greger R (1991) Effect of bicarbonate on potassium conductance of isolated perfused rat pancreatic ducts. Pflügers Arch 419: 76–83
Novak I, Pahl C (1993) Patch clamp experiments on isolated pancreatic ducts. Pflügers Arch 422: R71
Novak I, Pahl C (1993) Effect of secretin and inhibitors of HCO −3 /H+ transport on membrane voltage of rat pancreatic duct cells. Pflügers Arch 425:272–279
Pahl C, Novak I (1993) Effect of vasoactive intestinal peptide, carbachol and other agonists on cell membrane voltage of pancreatic duct cells. Pflügers Arch 424: 315–320
Palmer Smith J, Yelamarty RV, Kramer ST, Cheung JY (1993) Effect of cholecystokinin on cytosolic calcium in pancreatic duct segments and ductal cells. Am J Physiol 264: G1177-G1183
Petersen OH (1992) Stimulus-secretion coupling: cytoplasmic calcium signals and the control of ion channels in exocrine acinar cells. J Physiol (Lond) 448: 1–51
Said SI, Mutt V (1972) Isolation from porcine-intestinal wall of a vasoactive octacosapeptide related to secretin and to glucagon. Eur J Biochem 28: 199–204
Sasaki T, Gallacher DV (1990) Extracellular ATP activates receptor-operated cation channels in mouse lacrimal acinar to promote calcium influx in the absence of phosphoinositide metabolism. FEBS Lett 264: 130–134
Sasaki T, Gallacher DV (1992) The ATP-induced inward current in mouse lacrimal acinar cells is potentiated by isoprenaline and GTP. J Physiol (Lond) 447: 103–118
Schulz I (1980) Bicarbonate transport in the exocrine pancreas. Ann NY Acad Sci 341: 191–209
Sewell WA, Young JA (1975) Secretion of electrolytes by the pancreas of the anaesthetized rat. J Physiol (Lond) 252: 379–396
Soltoff SP, McMillan MK, Lechleiter JD, Cantley LC, Talamo BR (1990) Elevation of [Ca2+]i and the activation of ion channels and fluxes by extracellular ATP and phospholipase C-linked agonists in rat parotid acinar cells. Ann NY Acad Sci 603: 76–92
Stuenkel EL, Hootman SR (1990) Secretagogue effect on intracellular calcium in pancreatic duct cells. Pflügers Arch 416:652–658
Stutts MJ, Chinet TC, Mason SJ, Fullton JM, Clarke LL, Boucher RC (1992) Regulation of Cl− channels in normal and cystic fibrosis airway epithelial cells by extracellular ATP. Proc Natl Acad Sci USA 89: 1621–1625
Toescu EC, Lawrie AM, Petersen OH, Gallacher DV (1992) Spatial and temporal distribution of agonist-evoked cytoplasmic Ca2+ signals in exocrine acinar cells analysed by digital image microscopy. EMBO J 11: 1623–1629
Toescu EC, O'Neill SC, Petersen OH, Eisner DA (1992) Caffeine inhibits the agonist-evoked cytosolic Ca2+ signal in mouse pancreatic acinar cells by blocking inositol trisphosphate production. J Biol Chem 267: 23 467–23 470
Trimble ER, Bruzzone R, Biden TJ, Meehan CJ, Andreu D, Merrifield RB (1987) Secretin stimulates cyclic AMP and inositol triphosphate production in rat pancreatic acinar tissue by two fully independent mechanisms. Proc Natl Acad Sci USA 84: 3146–3150
Warth R, Greger R (1993) The ion conductances of CFPAC-1 cells. Cell Physiol Biochem 3: 2–16
Yamagishi F, Homma N, Haruta K, Iwatsuki K, Chiba S (1986) Effects of three purine-related compounds on pancreatic exocrine secretion in the dog. Clin Exp Pharmacol Physiol 13: 425–432
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hug, M., Pahl, C. & Novak, I. Effect of ATP, carbachol and other agonists on intracellular calcium activity and membrane voltage of pancreatic ducts. Pflugers Arch. 426, 412–418 (1994). https://doi.org/10.1007/BF00388304
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00388304