Summary
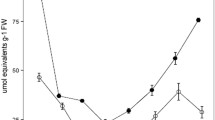
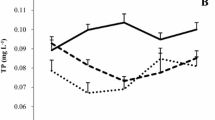
Isoetes bolanderi dominates the littoral flora of Siesta (elevation 2,440 m) and Ellery (2,905 m) lakes in the Sierra Nevada Range of California, USA. Both lakes are sparesly vegetated and I. bolanderi maintained aboveground oven dry weight of 30–50 m−22 through most of the 1981 summer growing season. Plants at the higher elevation Ellery Lake were half as large as plants at Siesta Lake and had substantially more biomass in corms. Titratable acidity levels in Isoetes leaves showed a diurnal fluctuation <50 μeq g1 fresh weight early in the season at the highest elevation site but this increased to ∼300 μeq g1 FW by mid-summer; starch and chlorophyll levels likewise increased in the leaves over this time. Throughout the season the magnitude of the diurnal acid change was comparable inIsoetes from both lakes but the dynamics of daytime deacidification were not. Averaged over the season, total daytime deacidification at Ellery Lake was 65% complete by noon whereas at Siesta Lake it was only 22% complete by noon. It is suggested that this may be related to the fact that Siesta Lake was more acidic and thus more carbon was in the form of free CO2. In both lakes water chemistry showed no consistent diurnal fluctuation in pH or free CO2 though total inorganic carbon levels were at the extreme low end for aquatic habitats. The studies reported here suggest that under extremely low inorganic carbon levels there may be selection for nighttime CO2 assimilation. Consistent with this hypothesis is the observation that emergent I. bolanderi plants, resulting from fluctuating water levels, initiated leaves with stomata(unlike adjacent submerged plants) and, although these leaves had substantially higher chlorophyll levels, they showed an order of magnitude less acid fluctuation than submerged leaves.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adams MS, Guilizzoni P, Adams S (1978) Relationship of dissolved inorganic carbon to macrophyte photosynthesis in some Italian lakes. Limnol Oceanogr 23:912–919
APHA (1976) Standard methods for the examination of water and wastewater. 14th ed American Public Health Association, Washington, D.C.
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoxidase in Beta vulgaris. Plant Physiol 24:1–15
Boylen CW, Sheldon RB (1976) Submergent macrophytes: Growth under winter ice cover. Science 182:841–2
Boston HL, Adams MS Evidence of crassulacean acid metabolism in two North American Isoetids. Aquat Bot (in press)
Clark DD, Burk JH (1980) Resource allocation patterns of two California Sonoran desert ephemerals. Oecologia (Berlin) 46:86–91
Gutmann I, Wahlefeld AW (1974) L-Malate: Determination with malate dehydrogenase and NAD. In: HU Bergmeyer (ed), Methods of enzymatic analysis, 4th Vol. Academic Press, NY p 1585–1589
Hudson GJ, John PMV, Bailey BJ, Southgate DAT (1976) The automated determination of carbohydrate. Development of a method for available carbohydrates and its application to foodstuffs. J Sci Food Agric 27:681–687
Keeley JE (1981a) Isoetes howellii: A submerged aquatic CAM plant? Am J Bot 68:420–424
Keeley JE (1981b) Diurnal acid metabolism in vernal pool Isoetes. Madroño 28:167–171
Keeley JE (1982) Distribution of diurnal acid metabolism in the genus Isoetes. Am J Bot 69:254–257
Keeley JE, Bowes G (1982) Gas exchange characteristics of the submerged aquatic crassulacean acid metabolism plant, Isoetes howellii. Plnat Physiol 70:1455–1458
Keeley JE, Morton BA (1982) Distribution of diurnal acid metabolism in submerged aquatic plants outside the genus Isoetes. Photosynthetica 16 (in press)
Keeley JE (1983a) Crassulacean acid metabolism in the seasonally submerged aquatic Isoetes howellii. Oecologia (Berlin) 58:57–62
Keeley JE (1983b) Lack of diurnal acid metabolism in terrestrial Isoetes (Isoetaceae). Photosynthetica 17 (in press)
Kluge M, Ting IP (1978) Crassulacean acid metabolism. Springer-Verlag, Berlin
Lee L, Belknap F (1970) Hard water as a limiting factor in the distribution of Isoetes echinospora. Am Fern J 60:134–136
Osmond CB (1978) Crassulacean acid metabolism: A curiosity in context. Ann Rev Plant Physiol 29:379–414
Pfeiffer NE (1922) Monograph on the Isoetaceae. Ann Mo Bot Gard 9:79–103
Prins HBA, Helder RJ (1981) The microlimate of submerged leaves in relation to CO2 and HCO −23 assimilation. XIII International Botanical Congress, Sydney Australia, 21–28 August 1981, Abstracts
Prins HBA, Snel JFH, Helder RJ, Zanstra PE (1980) Photosynthetic HCO −23 utilization and OH−2 excretion in aquatic angiosperms. Plant Physiol 66:818–822
Seddon B (1965) Occurence of Isoetes echinospora in eutrophic lakes in Wales. Ecology 46:747–748
Sestak Z, Catsky J, Jarvis PG (eds) (1971) Plant photosynthetic production. Manual of methods. W. Junk, The Hague
Schulthorpe CD (1967) The biology of aquatic vascular plants. E Arnold, London
Søndergaard M, Sand-Jensen K (1979) Carbon uptake by leaves and roots of Littorella uniflora (L.) Aschers. Aquat Bot 6:1–12
Westlake DF (1967) Some effects of low velocity currents on the metabolism of aquatic macrophytes. J Exp Bot 18:187–205
Wium-Anderson S (1971) Photosynthetic uptake of free CO2 by the roots of Lobellia dortmanna. Physiol Plant 25:245–248
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Keeley, J.E., Walker, C.M. & Mathews, R.P. Crassulacean acid metabolism in Isoetes bolanderi in high elevation oligotrophic lakes. Oecologia 58, 63–69 (1983). https://doi.org/10.1007/BF00384543
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00384543