Summary
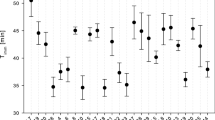
Laboratory respiration rate experiments using three electrophoretically identified clones of the fresh water, planktonic cladoceran, Daphnia pulex, from an eutrophic farm pond, indicated that clones acclimated to both low and high oxygen levels, regulated oxygen consumption across a wide range of oxygen concentrations (1.0–9.0 mg· liter-1). A “threshold” oxygen level of 0.5–1.0 mg·liter-1 was reached, where animals succumbed to oxygen stress, regardless of hemoglobin content. No significant clonal differences in respiration rates were found. These data suggest that members of this Daphnia population are able to regulate oxygen metabolism across a wide range of ambient oxygen concentrations, and indicate a well-adapted respiratory system.
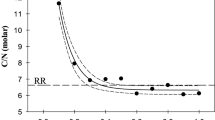
Low-oxygen tolerance experiments and hemoglobin measurements indicated further that physiological differences indeed exist between clones; one clone produced the lowest amount of hemoglobin and was least tolerant of low oxygen levels. These data imply that spatial and temporal changes in dissolved oxygen concentration may be an important selective force influencing the clonal (genotypic) composition of natural cladoceran populations.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Belman BW, Childress JJ (1976) Circulatory adaptations to the oxygen minimum layer in the bathypelagic mysid Gnathophausia ingens. Biol Bull 150:15–37
Carvalho GR (1984) Haemoglobin synthesis in Daphnia magna Straus (Crustacea: Cladocera): ecological differentiation between neighboring populations. Freshwat Biol 14:501–506
Chandler A (1954) Causes of variation in the hemoglobin content of Daphnia (Crustacea: Cladocera) in nature. Proc Zool Soc Lond 124:625–630
Childress JJ (1968) Oxygen minimum layer, vertical distribution and respiration of the mysid Gnathophausia ingens. Science 160:1242–1243
Childress JJ (1971) Respiratory adaptations to the oxygen minimum layer in the bathypelagic mysid Gnathophausia ingens. Biol Bull 141:109–121
Childress JJ (1975) The respiratory rates of midwater crustaceans as a function of depth of occurrence and in relation to the oxygen minimum off southern California. Comp Biochem Physiol 50:787–799
Childress JJ (1976) Physiological approaches to the biology of midwater crustaceans. In: Anderson N (ed) Predictions of Sonic Scattering Layer, Plenum, New York, pp 37–80
Davis JC (1975) Minimal dissolved oxygen requirements of aquatic life with emphasis on Canadian species: a review. J Fish Res Bd Can 32:2295–2332
Fox HM (1945) The oxygen affinities of certain invertebrate hemoglobins. J Exp Biol 21:161–165
Fox HM (1948) The haemoglobin of Daphnia. Proc R Soc Lond B Biol Sci 135:195–212
Fox HM (1954) Oxygen and haem in invertebrates. Nature 174:355
Green J (1956) Variation in the haemoglobin content of Daphnia. Proc R Soc Lond B Biol Sci 145:214–232
Hebert PDN, Crease TJ (1980) Clonal coexistence in Daphnia pulex (Leydig): Another planktonic paradox. Science 207:1363–1365
Heisey D, Porter K (1977) The filtering and respiration rates of Daphnia magna and Daphnia galeata. LImnol Oceanogr 22:839–845
Herbert MR (1954) The tolerance of oxygen deficiency in the water by certain cladocera. Mem Ist Ital Idrobiol 8:97–107
Hutchinson GE (1967) A Treatise on Limnology, Vol 2. Wiley, New York
Kobayashi M (1981) The haemoglobin concentration in relation to the body size in Daphnia magna. Sci Rep Niigata Univ Ser D (Biol) 18:15–19
Kobayashi M (1982a) Influence of body size on haemoglobin concentration and resistance to oxygen deficiency in Daphnia magna. Comp Biochem Physiol 72A:599–602
Kobayashi M (1982b) Relationship between oxygen consumption and resistance to oxygen deficiency of Daphnia magna. Comp Biochem Physiol 73A:239–241
Kobayashi M, Hoshi T (1984) Analysis of respiratory role of haemoglobin in Daphnia magna. Zool Science 1:523–532
Lampert W (1984) The measurement of respiration. In: Downing JA, Rigler FH (eds) A manual on methods for the assessment of secondary productivity in fresh waters, 2nd ed, Blackwell, Oxford, pp 413–468
Landon MS, Stasiak RH (1983) Daphnia haemoglobin concentration as a function of depth and oxygen availability in Arco Lake, Minnesota. Limnol Oceanogr 28:731–737
Lehninger AL (1975) Biochemistry, 2nd ed, Worth, New York
Lynch M (1983) Ecological genetics of Daphnia. Evolution 37:358–374
Lynch M, Weider LJ, Lampert W (1985) Measuring the carbon balance in Daphnia. Limnol Oceanogr (in press)
Obreshkove V, Banta AM (1930) A study of the rate of oxygen consumption in different Cladocera clones derived from the same mother. Physiol Zool 3:1–8
Ray AA (1982) SAS User's Guide, Statistics 1982. SAS Inst Cary, North Carolina
Sokal RR, Rohlf FJ (1981) Biometry, 2nd ed, WH Freeman and Co., San Francisco
Terwilliger RC (1980) Structures of invertebrate hemoglobins. Amer Zool 20:53–67
Vos J, Bernaerts F, Gabriels I, Decleir W (1979) Aerobic and anaerobic respiration of adult Artemia salina L., acclimated to different oxygen concentrations. Comp Biochem Physiol 62A:545–548
Weber RE (1980) Functions of invertebrate hemoglobins with special reference to adaptations to environmental hypoxia. Amer Zool 20:79–101
Weider LJ (1984a) Spatial heterogeneity of Daphnia genotypes: Vertical migration and habitat partitioning. Limnol Oceanogr 29:225–235
Weider LJ (1984b) Spatial and Temporal Genetic Heterogeneity in a Natural Daphnia Population: Ecological and Physiological Differences Between Genotypes, unpubl Ph D dissertation, Univ of Illinois, Urbana-Champaign
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weider, L.J., Lampert, W. Differential response of Daphnia genotypes to oxygen stress: respiration rates, hemoglobin content and low-oxygen tolerance. Oecologia 65, 487–491 (1985). https://doi.org/10.1007/BF00379661
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00379661