Summary
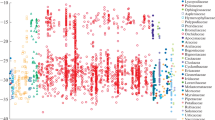
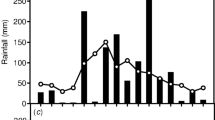
The occurrence of Crassulacean acid metabolism (CAM), as judged from δ13C values, was investigated in epiphytes and some related plant species at a series of sites covering the approximate altitudinal range of epiphytes in Papua New Guinea. Comprehensive collections were made at each site and the occurrence of water storage tissue and blade thickness was also determined. Some 26% of epiphytic orchids from a lowland rainforest (2–300 m.a.s.l) showed δ13C values typical of obligate CAM and possessed leaves thicker than 1 mm. A second group of orchids, mostly with succulent leaves, possessed intermediate δ13C values between -23 and -26% and accounted for 25% of the total species number. Some species of this group may exhibit weak CAM or be facultative CAM plants. The remainder of the lowland rainforest species appeared to be C3 plants with δ13C values between -28 and -35%. and generally possessed thin leaves. Obligate CAM species of orchids from a lower montane rainforest (1175 m.a.s.l) comprised 26% of the species total and mostly possessed thick leaves. The remainder of the species were generally thin-leaved with δ13C values between -26 and -35%. largely indicative of C3 photosynthesis. Orchids with intermediate δ13C values were not found in the lower montane rainforest. Obligate CAM appeared to be lacking in highland epiphytes from an upper montane rainforest and subalpine rainforest (2600–3600 m.a.s.l). However the fern, Microsorium cromwellii had a δ13C value of -21.28%. suggesting some measure of CAM activity. Other highland ferns and orchids showed more negative °13C values, up to-33%., typical of C3 photosynthesis. The highland epiphytic orchids possessed a greater mean leaf thickness than their lowland C3 counterparts due to the frequent occurrence of water storage tissue located on the adaxial side of the leaf. It is suggested that low daytime temperatures in the highland microhabitats is a major factor in explaining the absence of CAM. The increased frequency of water storage tissue in highland epiphytes may be an adaptation to periodic water stress events in the dry season and/or an adaptation to increased levels of UV light in the tropicalpine environment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Avadhani PN, Goh CJ, Rao AN, Arditti J (1982) Carbon fixation in orchids. In: Arditti J (ed) Orchid biology, reviews and perspectives, II. Comstok/ Cornell University Press Ithaca London, pp 173–193
Barcikowski W, Nobel PS (1984) Water relations of cacti during desiccation: distribution of water in tissues. Bot Gaz 145:110–115
Caldwell MM (1981) Plant response to solar ultraviolet radiation: In: Lange OL, Nobel PS, Osmond CB, Ziegler H (eds) Physiological plant ecology. Encycl Plant Physiol, New Series, Vol 12A. Springer, Berlin Heidelberg New York, pp 169–197
Coutinho LM (1969) Novas observacoes a ocortenica do “efeito de de Sassure” e suas relacoes com a suculencia, a temperatura folhear e os movimentos estomaticos. Botanica 24:97–102
Farquhar GD, Ball MC, Caemmerer S von, Roksandic Z (1982a) Effect of salinity and humidity on °13C value of halophytes — evidence for diffusional isotope fractionation determined by the ratio of intercellular/atmospheric partial pressure of CO2 under different environmental conditions. Oecologia (Berlin) 52:121–124
Farquhar GD, O'Leary MH, Berry JA (1982b) On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Aust J Plant Physiol 9:121–137
Fu CF, Hew CS (1982) Crassulacean acid metabolism in orchids under water stress. Bot Gaz 143:294–297
Gerwick BC, Williams GJ (1978) Temperature and water regulation of gas exchange of Opuntia polyacantha. Oecologia (Berlin) 35:149–159
Goh CJ, Avadhani PN, Loh CS, Hanegraaf C, Arditti J (1977) Diurnal stomatal and acidity rhythms in orchid leaves. New Phytol 78:365–372
Griffiths H, Smith JAC (1983) Photosynthetic pathways in the Bromeliaceae of Trinidad: relations between life-forms, habitat preference and the occurrence of CAM. oecologia (Berlin) 60:176–184
Hew CS (1984) Drymoglossum under water stress. Amer Fern J 74:37–39
Hnatiuk RJ, Smith JMB, McVean DN (1976) The climate of Mt. Wilhelm. Mt. Wilhelm Studies 2, ANU, BG/4, Canberra
Holttum RE (1968) Flora of Malaya Vol. II Ferns of Malaya. Government Printing Office, Singapore
Hope GS (1980) New Guinea mountain vegetation descriptions. In: van Royen P, The alpine flora of New Guinea, Vol 1. J Cramer, Vaduz, pp 153–122
Humphreys GS (1984) The environment and soils of Chimbu Province, Rapua New Guinea, with particular reference to soil erosion. Research Bull. 35, DPI, Port Moresby
Johns RJ, Stevens PF (1971) Mount Wilhelm flora: a checklist of the species. Div Bot, Dept Forests, Lae, Bulletin 6:1–60
Kluge M, Ting IP (1978) Crassulacean acid metabolism. Ecological studies, vol 16. Springer, Berlin Heidelberg New York
Lerman JC (1975) How to interpret variations in the carbon isotope ratio of plants: biologic and environmental effects. In: Marcelle R (ed) Environmental and biological control of photosynthesis. Dr W Junk, The Hague, pp 323–335
McAlpine JR, Keig G, Short K (1975) Climatic tables for Papua New Guinea. CSIRO, Melbourne
McWilliams EL (1970) Comparative rates of dark CO2 uptake and acidification in the Bromeliaceae, Orchidaceae and Euphorbiaceae. Bot Gaz 131:285–290
Medina E (1974) Dark CO2 fixation, habitat preference and evolution within the Bromeliaceae. Evolution 28:677–686
Medina E, Delgado M (1976) Photosynthesis and night CO2 fixation in Echeveria columbiana v. Poellnitz. Photosynthetica 10:155–163
Medina E, Minchin P (1980) Stratification of °13C values in Amazonian rain forests. Oecologia (Berlin) 45:377–378
Medina E, Osmond CB (1981) Temperature dependence of dark CO2 fixation and acid accumulation in Kalanchoë daigremontiana. Aust J Plant Physiol 8:641–649
Medina E, Troughton JH (1974) Dark CO2 fixation and the carbon isotope ratio in Bromeliaceae. Plant Sci Lett 2:357–362
Medina E, Delgado M, Troughton JH, Medina JD (1977) Physiological ecology of CO2 fixation in Bromeliaceae. Flora 166:137–152
Milburn TR, Pearson DJ, Ndegwe NA (1968) Crassulacean acid metabolism under natural tropical conditions. New Phytol 67:883–879
Neales TF, Hew CS (1975) Two types of carbon fixation in tropical orchids. Planta 123:303–306
Osmond CB (1978) Crassulacean acid metabolism: a curiosity in context. Ann Rev Plant Physiol 29:379–414
Osmond CB, Ziegler H, Stichler W, Trimborn P (1975) Carbon isotope discrimination in alpine succulent plants supposed to be capable of crassulacean acid metabolism. Oecologia (Berlin) 18:209–217
Richards PW (1952) The tropical rainforest. Camb Univ Press, London New York Melbourne
Schmidt JE, Kaiser WM (1987) Response of the succulent leaves of Peperomia magnoliaefolia to dehydration. Plant Physiol 83:190–194
Sinclair R (1984) Water relations of tropical epiphytes. III. Evidence for Crassulacean acid metabolism. J Exp Bot 35:1–7
Smith JAC, Griffiths H, Bassett M, Griffiths NM (1985) Day-night changes in the leaf water relations of epiphytic bromeliads in the rain forests of Trinidad. Oecologia (Berlin) 67:475–485
Smith JAC, Griffiths H, Lüttge U (1986) Comparative ecophysiology of CAM and C3 bromeliads. I. The ecology of the Bromeliaceae in Trinidad. Plant, Cell Environ 9:359–376
Teeri JA, Tonsor SJ, Turner M (1981) Leaf thickness and carbon isotope composition in the Crassulaceae. Oecologia (Berlin) 50:367–369
Tieszen LL, Senyimba MM, Imbamba SK, Troughton JH (1979) The distribution of C3 and C4 grasses and carbon isotope discrimination along an altitudinal and moisture gradient in Kenya. Oecologia (Berlin) 37:337–350
Troughton JH (1979) δ13C as an indicator of carboxylation reactions. In: Gibbs M, Latzko E (eds) Photosynthesis II: Photosynthetic carbon metabolism. Encycl Plant Physiol, New Series, Vol 6. Springer, Berlin Heidelberg New York, pp 140–147
Wade LK, McVean DN (1969) Mt. Wilhelm Studies I. The alpine and subalpine vegetation. ANU, BG/1, Canberra
Wagner J, Larcher W (1981) Dependence of CO2 gas exchange and acid metabolism of the alpine CAM plant Sempervivum montanum on temperature and light. Oecologia (Berlin) 50:88–93
Winter K (1985) Crassulacean acid metabolism. In: Barber J, Baker NR (eds) Photosynthetic mechanisms and the environment Elsevier, pp 329–387
Winter K, Tenhunen JD (1982) Light-stimulated burst of carbon dioxide uptake following nocturnal acidification in the Crassulacean acid metabolism plant Kalanchoë daigremontiana. Plant Physiol 70:1718–1722
Winter K, Wallace BJ, Stocker GC, Roksandic Z (1983) Crassulacean acid metabolism in Australian vascular epiphytes and some related species. Oecologia (Berlin) 57:129–141
Winter K, Osmond CB, Hubick KT (1986) Crassulacean acid metabolism in the shade. Studies on an epiphytic fern, Pyrrosia longifolia, and other rainforest species from Australia. Oecologia (Berlin) 68:224–230
Wong SC, Hew CS (1976) Diffusive resistance, titratable acidity, and CO2 fixation in two tropical epiphyte ferns. Amer Fern J 66:121–124
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Earnshaw, M.J., Winter, K., Ziegler, H. et al. Altitudinal changes in the incidence of crassulacean acid metabolism in vascular epiphytes and related life forms in Papua New Guinea. Oecologia 73, 566–572 (1987). https://doi.org/10.1007/BF00379417
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00379417