Abstract
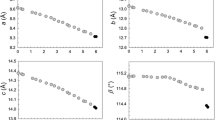
CaSO4 has been investigated at room temperature over the pressure range of 0 to 160 kbar. A reversible, sluggish transition that occurs at about 20 kbar with a volume change of about 4% has been confirmed. The x-ray powder data collected at pressure can be indexed on the monoclinic monazite structure cell (P21/n) which is considered to be the most likely model structure for the new HP CaSO4 phase. The transition is largely distortional and involves rotation and displacement, but essentially preserves the chains of edge shared CaO8(9) and SO4 polyhedra found in the orthorhombic anhydrite (Bbmm) structure.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Stephens, D. R.: The hydrostatic compression of eight rocks. J. Geophys. Res. 69, 2967–2978 (1964)
Kahn, J. S.: High pressure phase transformation of CaSO4 during a nuclear explosion, Lawrence Livermore Laboratory Report UCRL-70142 (1966)
Pistorius, C. W. F. T., Boeyens, C. A., Clark, J. B.: Phase diagrams of NaBF4 and NaClO4 to 40 kbar and the crystal-chemical relationship between the structures of CaSO4, AgMnO4, BaSO4 and High-NaClO4. High Temp. High Pressures 1, 41–52 (1969)
Jamieson, J. C., Lawson, A. W.: X-ray diffraction studies in the 100 kilobar pressure range. J. Appl. Phys. 33, 776–780 (1962)
Borg, I. Y., Smith, D. K.: X-ray diffraction studies on CdTe at high pressure. J. Phys. Chem. Solids 28, 49–54 (1967)
Schock, R. N., Olinger, B., Duba, A.: Additional data on the compression of olivine to 140 kilobars. J. Geophys. Res. 77, 382–384 (1972)
Christian, R. H.: Equation of state of alkali halides at high pressure. Lawrence Livermore Laboratory Report UCRL-4900 (1957)
Bridgman, P. W.: The compression of twenty-one halogen compounds and eleven other simple substances to 100,000 kg/cm3. Proc. Am. Acad. Arts Sci. 76, 5 (1945)
Clark, C. M., Smith, D. K., Johnson, G. G.: A Fortran IV program for calculating x-ray powder patterns — version V. College of Earth and Mineral Science Bulletin, Penn. State Univ., 152 pp. (1973)
Finney, J. J., Rao, N. N.: The crystal structure of cheralite. Am. Mineralogist 52, 13–19 (1967)
Mooney, R. C. L.: Crystal structure of a series of rare earth phosphates. J. Chem. Phys. 16, 1003 (1948)
Pistorius, C. W. F. T., Pistorius, M. C.: Lattice constants and thermal expansion properties of the chromates and selenates of lead, strontium, and barium. Z. Krist. 117, 259–271 (1962)
Gobel, J. B., Wilson, A. S.: INDEX, a computer program for indexing x-ray diffraction patterns. BNWL-22 Battelle Northwest Laboratory Report (1965)
Shannon, R. D., Prewitt, C. T.: Effective ionic radii in oxides and fluorides. Acta Cryst. B 25, 925–946 (1969)
Kokkoros, P. A., Rentzeperis, P. J.: X-ray investigation of the anhydrous cadmium and mercuric sulfates. Acta Cryst. 14, 329–330 (1961)
Snyman, H. C., Pistorius, C. W. F. T.: Some crystallographic properties of CaSeO4 and its hydrates. Z. Krist. 119, 151–154 (1963)
Muller, O., Roy, R.: Phase transitions among the ABX4 compounds. Z. Krist. 138, 237–253 (1973)
Wolten, G. M.: Diffusionless phase transformations in zirconia and hafnia. J. Am. Ceram. Soc. 46, 418–422 (1963)
Author information
Authors and Affiliations
Additional information
Work performed under the auspices of the U.S. Atomic Energy Commission.
Rights and permissions
About this article
Cite this article
Borg, I.Y., Smith, D.K. A high pressure polymorph of CaSO4 . Contr. Mineral. and Petrol. 50, 127–133 (1975). https://doi.org/10.1007/BF00373332
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00373332