Abstract
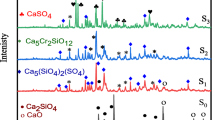
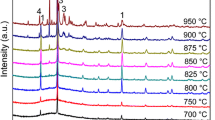
The effect of Cr2O3 on some anorthite-diopside glass-ceramics has been investigated up to amounts of 5 mol%. The solubility in the glassy compositions analysed is total for the oxide, but for amounts higher than 0.5 mol%, an insoluble spinel form, MgCr2O4, precipitates. Ultraviolet-visible spectroscopy has proved to be the most sensitive technique to the presence of Cr(III) in a crystalline spinel site, followed by X-ray diffraction and scanning electron microscopy observations. Electron spin resonance and X-ray photoelectron spectroscopy techniques excluded any oxidation state, other than Cr3+. The influence of the transition cation on glass nucleation is that of an increasing bulk effect with chromium, and thus chromium-spinel, content. The magnesium content affects spinel formation, while heat treatments up to 1100 °C do not. The spinel formation influences the anorthite-diopside ratio in the glass-ceramic, with a large favour towards the pyroxene.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. B. Volf, “Chemical Approach to Glass”, “Glass Science and Technology”, Vol. 7 (Elsevier, Amsterdam, 1984) pp. 334–9.
G. H. Beall and D. A. Duke, in “Glass-Ceramic Technology”, “Glass: Science and Technology”, Vol. 1, edited by D. R. Uhlmann and N. J. Kreidl (Academic Press, New York, 1983) pp. 886–99.
P. F. James, in “Volume nucleation in silicate glasses”, “Glasses and Glass-ceramics”, edited by M. H. Lewis (Chapman and Hall, London, 1989) pp. 61–105.
F. Durville, B. Champagnon, E. Duval, F. Gaume, A. F. Wright and A. N. Fitch, Phys. Chem. Glasses 25 (1984) 126.
H. S. Kim, R. D. Rawlings and P. S. Rogers, J. Mater. Sci. 24 (1989) 1025, and references quoted therein.
C. Leonelli, T. Manfredini, M. Paganelli, P. Pozzi and G. C. Pellacani, ibid. 26 (1991) 5041.
L. Barbieri, C. Leonelli, T. Manfredini, M. Paganelli and G. C. Pellacani, J. Thermal Anal. 38 (1992) 2639.
S. Dingkun and L. A. Orlova, J. Non-Cryst. Solids 112 (1989) 207.
A. A. Omar, A. W. A. El-Shennawi and G. A. Khater, Br. Ceram. Trans. J. 90 (1991) 179, and references quoted therein.
M. J. Robson and M. W. Davies, BISRA-C/64/69 (1971).
M. P. Seah and G. C. Smith, in “Practical Surface Analysis”, edited by D. Briggs and M. P. Seah, Vol. 1, 2nd Edn, Appendix 1 (Wiley, Chichester, 1990) pp. 543–4.
A. A. Omar, S. M. Salman and M. Y. Mahmoud, Ceram. 15 (1985) 57, and references quoted therein.
K. Matusita, S. Sakka and Y. Matsui, J. Mater. Sci. 10 (1975) 961.
K. Metusita and S. Sakkor, J. Non-Cryst. Solids 38–39 (1980) 741.
X. J. Xu, C. S. Ray and D. F. Day, J. Am. Ceram. Soc. 74 (1991) 909.
F. Durville, B. Champagnon, E. Duval and G. Boulon, J. Phys. Chem. Solids 46 (1985) 701.
R. Reisfeld, A. Kisilev, E. Greenberg, A. Buch and M. Ish-Shalom, Chem. Phys. Lett. 104 (1984) 153.
Idem, ibid. 105 (1984) 405.
P. C. Schultz, J. Am. Ceram. Soc. 57 (1974) 178.
D. E. O'Reilly and D. S. Mac Iver, J. Phys. Chem. 66 (1962) 276.
K. Tanaka and K. Kimiya, J. Mater. Sci. Lett. 10 (1991) 1095.
D. L. Griscom, J. Non-Cryst. Solids 40 (1980) 211.
C. D. Wagner, W. M. Riggs, L. E. Davis, J. S. Moulder and G. E. Muilnberd, in “Handbook of X-ray Photoelectron Spectroscopy”, edited by C. D. Wagner (Perkin Elmer, Eden Prairie, USA, 1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barbieri, L., Leonelli, C., Manfredini, T. et al. Solubility, reactivity and nucleation effect of Cr2O3 in the CaO-MgO-Al2O3-SiO2 glassy system. JOURNAL OF MATERIALS SCIENCE 29, 6273–6280 (1994). https://doi.org/10.1007/BF00354571
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00354571