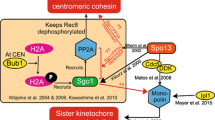
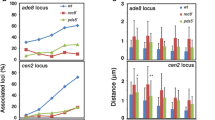
Abstract
Kinetochores and chromatid cores of meiotic chromosomes of the grasshopper species Arcyptera fusca and Eyprepocnemis plorans were differentially silver stained to analyse the possible involvement of both structures in chromatid cohesiveness and meiotic chromosome segregation. Special attention was paid to the behaviour of these structures in the univalent sex chromosome, and in B univalents with different orientations during the first meiotic division. It was observed that while sister chromatid of univalents are associated at metaphase I, chromatid cores are individualised independently of their orientation. We think that cohesive proteins on the inner surface of sister chromatids, and not the chromatid cores, are involved in the chromatid cohesiveness that maintains associated sister chromatids of bivalents and univalents until anaphase I. At anaphase I sister chromatids of amphitelically oriented B univalents or spontaneous autosomal univalents separate but do not reach the poles because they remain connected at the centromere by a long strand which can be visualized by silver staining, that joins stretched sister kinetochores. This strand is normally observed between sister kinetochores of half-bivalents at metaphase II and early anaphase II. We suggest that certain centromere proteins that form the silver-stainable strand assure chromosome integrity until metaphase II. These cohesive centromere proteins would be released or modified during anaphase II to allow normal chromatid segregation. Failure of this process during the first meiotic division could lead to the lagging of amphitelically oriented univalents. Based on our results we propose a model of meiotic chromosome segregation. During mitosis the cohesive proteins located at the centromere and chromosome arms are released during the same cellular division. During meiosis those proteins must be sequentially inactivated, i.e. those situated on the inner surface of the chromatids must be eliminated during the first meiotic division while those located at the centromere must be released during the second meiotic division.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adachi Y, Luke M, Laemmli UK (1991) Chromosome assembly in vitro: topoisomerase II is required for condensation. Cell 64: 137–148
Brinkley BR (1990) Centromeres and kinetochores: integrated domains on eukayotic chromosomes. Curr Opin Cell Biol 2: 446–452
Carlson JD (1938) Mitotic behavior of induced chromosomal fragments lacking spindle attachments in the neuroblasts of the grasshopper. Proc Natl Acad Sci USA 24: 500–507
Cooke CA, Heck MMS, Earnshaw WC (1987) The inner centromere protein (INCENP) antigens: movement from inner centromere to midbody during mitosis. J Cell Biol 105: 2053–2067
Earnshaw WC (1991 a) When is a centromere not a kinetochore? J Cell Sci 99: 1–4
Earnshaw WC (1991 b) Large scale chromosome structure and organization. Curr Opin Struct Biol 1: 237–244
Earnshaw WC, Cooke CA (1989) Proteins of the inner and outer centromere of mitotic chromosomes. Genome 31: 541–552
Earnshaw WC, Cooke CA (1989) Analysis of the distribution of the INCENPs throughout mitosis reveals the existence of a pathway of structural changes in the chromosomes during metaphase and early events in cleavage furrow formation. J Cell Sci 98: 443–461
Earnshaw WC, Rattner JB (1989) A map of the centromere (primary constriction) in vertebrate chromosomes at metaphase. In: Vig B, Resnick M (eds) Aneuploidy: Mechanisms of origin. Liss, New York, pp 33–42
Esponda P (1978) Cytochemistry of kinetochores under electron microscopy, Exp Cell Res 114: 247–252
Esponda P, Krimer DB (1979) Development of the synaptonemal complex and polycomplex formation in three species of grasshoppers. Chromosomal 73: 237–245
Maguire MP (1990) Sister chromatid cohesiveness: vital function, obscure mechanism. Biochem Cell Biol 68: 1231–1242
Moens PB, Church K (1979) The distribution of synaptonemal complex material in metaphase I bivalents of Locusta and Chloealtis (Orthoptera: Acrididae). Chromosoma 73: 247–254
Nicklas RB (1988) Chromosomes and kinetochores do more in mitosis than previously thought. In: Gustason JP, Appels R (eds) Chromosome structure and function: The impact of new concepts. Plenum Press, New York, pp 53–74
Pluta AF, Cooke CA, Earnshaw WC (1990) Structure of the human centromere at metaphase. Trends Biochem Sci 15: 181–185
Rattner JB (1991) The structure of the mammalian centromere. BioEssays 13: 51–56
Rattner JB, Kingwell BG, Fritzler MJ (1988) Detection of distinct structural domains within the primary constriction using autoantibodies. Chromosoma 96: 360–367
Rose D, Thomas W, Holm C (1990) Segregation of recombined chromosomes inmeiosis I requires DNA topoisomerase II. Cell 60: 1009–1017
Rufas JS, Gimenez-Abian J, Suja JA, Garcia de la Vega C (1987) Chromosome organization in meiosis revealed by light microscope analysis of silver-stained cores. Genome 29: 706–712
Rufas JS, Mazzella C, Sujade la Vega C (1989) Kinetochore structures are duplicate prior to the first meiotic metaphase. A model of meiotic behaviour of kinetochores in grasshoppers. Eur J Cell Biol 48: 220–226
Suja JA, Garcia de la Vega C, Rufas JS (1989) Mechanisms promoting the appearance of abnormal spermatids in B-carrier individuals of Eyprepocnemis plorans (Orthoptera). Genome 32: 64–71
Suja JA, de laTorre J, Gimenez-Abian JF, Garcia de la Vega C, Rufas JS (1991) Meiotic chromosome structure. Kinetochores and chromatid cores in standard and B-chromosomes of Arcyptera fusca (Orthoptera) revealed by silver staining. Genome 34: 19–27
Wood ER, Earnshaw WC (1990) Mitotic chromatin condensation in vitro using somatic cell extracts and nuclei with variable levels of endogenous topoisomerase II. J Cell Biol 111: 2839–2850
Author information
Authors and Affiliations
Additional information
by D.P. Bazett-Jones
Rights and permissions
About this article
Cite this article
Suja, J.A., Antonio, C. & Rufas, J.S. Involvement of chromatid cohesiveness at the centromere and chromosome arms in meiotic chromosome segregation: A cytological approach. Chromosoma 101, 493–501 (1992). https://doi.org/10.1007/BF00352472
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00352472