Abstract
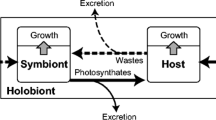
Stimulation of photosynthate excretion from zooxanthellae and free-living algae by tissue homogenate of several bivalves was studied. Mantle tissue homogenate of Tridacna derasa enhanced 10-to 15-fold excretion of photosynthetically fixed carbon from freshly isolated zooxanthellae within 2 h incubation. Maximum carbon excretion was 35 to 45% of the total carbon fixed. This excretion stimulating activity was detected in the homogenates of the mantle, adductor muscle, gill, and kidney. However, no excretion stimulating activity was detected in the haemolymph. The excretion stimulation activity of mantle homogenate, directed against freshly isolated zooxanthellae from T. derasa, was higher in bivalves belonging to the Tridacnidae (T. derasa, T. crocea, T. maxima, T. squamosa, Hippopus hippopus) than in the Cardiidae (Fragum fragum, F. mundum, F. unedo), non-symbiotic bivalves (Mytilus edulis, Meretrix lusoria, Ruditapes philippinarum) or gastropods (Umbonium giganteum, Turbo argyrostoma). The mantle homogenate of T. derasa enhanced photosynthate excretion by free-living algae belonging to the Dinophyceae (Prorocentrum micans, Amphidinium carterae, and Heterocapsa triquetra) but did not enhance its excretion by free-living algae belonging to the Chlorophyceae, Cyanophyceae, Rhodophyceae, Prasinophyceae, and Haptophyceae. T. derase used in this study originated from Belau (Palau). T. crocea, T. squamosa, T. maxima, H. hippopus and F. unedo were collected at Ishigaki Island in Okinawa in 1992. F. mundum and F. fragm were collected at Okinawa Island in 1992.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abe, T., Tsuzuki, M., Miyachi, S. (1987). Transport and fixation of inorganic carbon during photosynthesis of Anabaena grown under ordinary air. I. Active species of inorganic carbon utilized for photosynthesis. Pl. Cell Physiol. 28: 273–281
Blank, R. J., Trench, R. K. (1985). Speciation and symbiotic dinoflagellates. Science, N. Y. 229: 656–658
Borowitzka, M. A., Larkum, A. W. D. (1976). Calcification in the green alga Halimeda. II. The exchange of Ca2+ and the occurrence of age gradients in calcification and photosynthesis. J. exp. Bot. 27: 864–878
Bradford, M. M. (1976). A rapid sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analyl. Biochem. 72: 248–254
Cernichiari, L., Muscatine, L., Smith, D. C. (1969). Maltose excretion by the symbiotic algae of Hydra viridis. Proc. R. Soc. Lond. (Ser. B) 173: 557–576
Cook, C. B. (1983). Metabolic interchange in algae-invertebrate symbiosis. Int. Rev. Cytol. 14: 117–210
Fankboner, P. V. (1971). Intracellular digestion of symbiotic zooxanthellae by host amoebocytes in giant clams (Bivalvia: Tridacnidae), with a note on the nutritional role of the hypertrophied siphonal epidermis. Biol. Bull. mar. biol. Lab., Woods Hole 141: 222–234
Goreau, T. F., Goreau, N. I., Yonge, C. M. (1973). On the utilization of photosynthetic products from zooxanthellae and of a dissolved amino acid in Tridacna maxima f. elongata (Mollusca: Bivalvia). J. Zool., Lond. 169: 417–454
Guillard, R. R. L., Ryther, J. H. (1962). Studies on marine planktonic diatoms. I. Cyclotella nana Hustedt and Detonula confervacea (Cleve) Gran. Can. J. Microbiol. 8: 229–239
Hinde, R. (1988). Factors produced by symbiotic marine invertebrates with affect translocation between the symbionts. In: Scannerini S. et al. (ed.) NATO ASI Series, Vol. H17. Cell to cell signals in plant, animal and microbial symbiosis. Springer-Verlag, Berlin, p. 311–324
Jeffrey, S. W., Humphrey, G. F. (1975). New spectrophotometric equations for determining chlorophylls a, b, c 1, and c 2 in higher plants, algae and natural phytoplankton. Biochem. Physiol. Pfl. 167: 191–194
Kawaguti, S. (1950). Observations on the heart shell, Corculum cardissa (L.) and its associated zooxanthellae. Pacif. Sci. 4: 43–49
Kawaguti, S. (1966). Electron microscopy on the mantle of the giant clam with special reference to zooxanthellae and iridophores. Biol. J. Okayama Univ. 12: 81–92
Kawaguti, S. (1968). Electron microscopy on zooxanthellae in the mantle nad gill of the heart shell. Biol. J. Okayama Univ. 14: 1–12
Kawaguti, S. (1983). The third record of association between bivalve molluses and zooxanthellae. Proc. Japan Acad. 59 (Ser. B): 17–20
Keller, M. D., Selvin, R. C., Claus, W., Guillard, R. R. L. (1987). Media for the culture of oceanic ultraplankton. J. Phycol. 23: 633–638
Koch, W. (1953). Untersuchungen an bakterienfreien Massenkulturen der einzelligen Rotalge Porphyridium cruentum Naegeli. Archs Microbiol. 18: 232–241
Mansour, K. (1946). Communication between the dorsal edge of the mantle and the stomach of Tridacna. Nature, Lond. 157: 844
Muscatine, L. (1965). Symbiosis of hydra and algae. III Extracellular products of the algae. Comp. Biochem. Physiol. 16: 77–92
Muscatine, L. (1967). Glycerol excretion by symbiotic algae from corals and Tridacna and its control by the host. Science, N. Y. 156: 516–519
Muscatine, L. (1990). The role of symbiotic algae in carbon and energy flux in reef corals. In: Dubinsky Z. (ed.) Coral reefs. Elsevier Science Publishers B. V., Amsterdam, p. 75–87
Muscatine, L., Cernichiari, E (1969). Assimilation of photosynthetic products of zooxanthellae by a reef coral. Biol. Bull. mar. biol. Lab., Woods Hole 137: 506–523
Muscatine, L., Pool, R. R., Cernichiari, E. (1972). Some factors influencing selective release of soluble organic material by zooxanthellae from reef corals. Mar. Biol. 13: 298–308
Norton, J. H., Shepherd M. A., Long H. M., Fitt, W. K. (1992). The zooxanthellal tubular system in the giant clam. Biol. Bull. mar. biol. Lab., Woods Hole 183: 503–506
Okaichi, T., Nishio, S., Imatomi, Y. (1982). Collection and mass culture. In: Jap. Fish. Soc. (ed.) Toxic phytoplankton-occurrence, mode of action, and toxins. Koseisha-Koseikaku, Tokyo, p. 23–34 (in Japanese)
Streamer, M., Griffiths, D. J., Luong-van Thinh (1988). The products of photosynthesis by zooxanthellae (Symbiodinium microadriaticum) of Tridacna gigas and their transfer to the host. Symbiosis 6: 237–252
Sutton, D. C., Hoegh-Guldberg, O. (1990). Host-zooxanthellae interactions in four temperate marine invertebrate symbioses: assessment of effect of host extracts on symbionts. Biol. Bull. mar biol. Lab., Woods Hole 178: 175–186
Trench, R. K. (1971). The physiology and biochemistry of zooxanthellae symbiotic with marine coelenterates. III. The effect of homogenates of host tissues on the excretion of photosynthetic products in vitro by zooxanthellae from two marine coelenterates. Proc. R. Soc. Lond. (Ser. B) 177: 251–264
Trench, R. K., Wethey, Porter, D. S. (1981). Observations on the symbiosis with zooxanthellae among the Tridacnidae (Mollusca, Bivalvia). Biol. Bull. mar. biol. Lab., Woods Hole 161: 180–198
Umeshita, H., Yamasu, T. (1985). On the morphology of a species of strawberry cockle Fragum sp. Biol. Mag. Okinawa no 23: 50 (in Japanese)
Wafar, M. V., Qasim, S. Z. (1975). Carbon fixation and excretion in symbiotic algae (zooxanthellae) in the presence of host homogenates. Indian J. mar. Sci. 4: 43–46
Waterbury, J. B., Stanier, R. Y. (1981). Isolation and growth of cyanobacteria from marine and hypersaline environments. In: Starr, M. P., Stolp, H., Trüper, H. G. (eds.) The orokaryotes, Vol. 1. Springer-Verlag, Berlin, p. 221–223
Author information
Authors and Affiliations
Additional information
Communicated by T. Ikeda, Nagasaki
Rights and permissions
About this article
Cite this article
Masuda, K., Miyachi, S. & Maruyama, T. Sensitivity of zooxanthellae and non-symbiotic microalgae to stimulation of photosynthate excretion by giant clam tissue homogenate. Marine Biology 118, 687–693 (1994). https://doi.org/10.1007/BF00347517
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00347517