Abstract
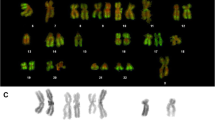
Unfixed metaphase chromosome preparations from human lymphocyte cultures were immunofluorescently labelled using antibodies to defined histone epitopes. Both mouse monoclonal antibody HBC-7, raised against the N-terminal region of H2B, and rabbit serum R5/12, which recognizes H4 acetylated at Lys-12, gave non-uniform labelling patterns, whereas control antibodies against total histone fractions H4 and H1 produced homogeneous fluorescence. HBC-7 bound approximately uniformly to the bulk of the chromosomes, but the major heterochromatic domains of chromosomes 1, 9, 15, 16 and the Y showed significantly brighter fluorescence. Serum R5/12 indicated an overall reduction in acetylation of H4 in metaphase chromosomes compared with interphase nuclei, although some specific chromosomal locations had considerably elevated acetylation levels. Acetylation levels in the major heterochromatic domains appeared extremely low. To investigate further the differences noted in heterochromatin labelling, metaphases from cultures grown in the presence of various agents known to induce undercondensation of the major heterochromatic domains were similarly immunolabelled. Decondensed heterochromatin no longer exhibited higher than normal immunofluorescence levels with HBC-7. The higher resolution afforded by “stretching” the centromeric heterochromatin of chromosomes 1, 9 and 16 confirmed the low level of H4 acetylation in these domains. We consider the implications of these observations in relation to chromatin conformation and activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bird AP (1986) CpG-rich islands and the function of DNA methylation. Nature 321: 209–213
Csordas A (1990) On the biological role of histone acetylation. Biochem J 265: 23–38
D'Anna JA, Gurely LR, Tobey RA (1983) Extent of histone modifications and H1° content during cell cycle progression in the presence of butyrate. Exp Cell Res 147: 407–417
Donlon TA, Magenis RE (1983) Methyl green is a substitute for distamycin A in the formation of distamycin A/DAPI C-bands. Hum Genet 65: 144–146
Felsenfeld G (1978) Chromatin. Nature 271: 115–122
Gatti M, Smith DA, Baker BS (1983) A gene controlling condensation of heterochromatin in Drosophila melanogaster. Science 221: 83–85
Gooderham K, Jeppesen P (1983) Chinese hamster metaphase chromosomes isolated under physiological conditions: a partial characterization of associated non-histone proteins and protein cores. Exp Cell Res 144: 1–14
Gosden JR, Mitchell AR, Buckland RA, Clayton RP, Evans HJ (1975) The location of four human satellite DNAs on human chromosomes. Exp Cell Res 92: 148–158
Haaf T, Dominguez-Steglich M, Schmid M (1990) Immunocytogenetics: IV. Human autoantibodies to heterochromatin-associated proteins. Cytogenet Cell Genet 53: 40–51
Hilwig I, Gropp A (1973) Decondensation of constitutive heterochromatin in L cell chromosomes by a benzimidazole compound (“33258 Hoechst”). Exp Cell Res 81: 474–477
James TC, Elgin SCR (1986) Identification of a nonhistone chromosomal protein associated with heterochromatin in Drosophila melanogaster and its gene. Mol Cell Biol 6: 3862–3872
Jeppesen P, Nicol L (1986) Non-kinetochore directed autoantibodies in scleroderma/CREST: identification of an activity recognizing a metaphase chromosome core non-histone protein. Mol Biol Med 3: 369–384
Jeppesen PGN, Bankier AT, Sanders L (1978) Non-histone proteins and the structure of metaphase chromosomes. Exp Cell Res 115: 293–302
Jones PA, Taylor SM (1980) Cellular differentiation, cytidine analogs and DNA methylation. Cell 20: 85–93
Matthews HR (1988) Histone modifications and chromatin structure. In: Adolph K (ed) Chromosomes and chromatin. 1. CRC Press, Boca Raton, Florida, pp 3–32
Miller OJ, Schnedl W, Allen J, Erlanger BF (1974) 5-Methylcytosine localized in mammalian constitutive heterochromatin. Nature 251: 636–637
Moroi Y, Peebles C, Fritzler MJ, Steigerwald J, Tan EM (1980) Autoantibody to centromere (kinetochore) in scleroderma sera. Proc Natl Acad Sci USA 77: 1627–1631
Musich PR, Brown FL, Maio JJ (1977) Subunit structure of chromatin and the organization of eukaryotic highly repetitive DNA: nucleosomal proteins associated with a highly repetitive mammalian DNA. Proc Natl Acad Sci USA 74: 3297–3301
Omori A, Igo-Kemenes T, Zachau HG (1980) Different repeat lengths in rat satellite I DNA containing chromatin and bulk chromatin. Nucleic Acids Res 8: 5363–5375
Perry PE, Thomson EJ (1986) Immunogold labelling of metaphase cells. Cytogenet Cell Genet 41: 121–125
Prosser J, Frommer M, Paul C, Vincent PC (1986) Sequence relationships of three human satellite DNAs. J Mol Biol 187: 145–155
Reuter G, Giarre M, Farah J, Gausz J, Spierer A, Spierer P (1990) Dependence of position-effect variegation in Drosophila on dose of a gene encoding an unusual zinc-finger protein. Nature 344: 219–223
Richmond TJ, Finch JT, Rushton B, Rhodes D, Klug A (1984) Structure of the nucleosome core particle at 7 Ao resolution. Nature 311: 532–537
Rocchi A, DiCastro M, Prantera G (1979) Effects of DAPI on human leukocytes in vitro. Cytogenet Cell Genet 23: 250–254
Schmid M, Grunert D, Haaf T, Engel W (1983) A direct demonstration of somatically paired heterochromatin of human chromosomes. Cytogenet Cell Genet 36: 554–561
Simpson RT (1978) Structure of the chromatosome, a chromatin particle containing 160 base pairs of DNA and all the histones. Biochemistry 17: 5524–5531
Stollar BD, Ward M (1970) Rabbit antibodies to histone fractions as specific reagents for preparative and comparative studies. J Biol Chem 245: 1261–1266
Strauss F, Varshavsky A (1984) A protein binds to a satellite DNA repeat at three specific sites that would be brought into mutual proximity by DNA folding in the nucleosome. Cell 37: 889–901
Thoma F, Koller T, Klug A (1979) Involvement of histone H1 in the organization of the nucleosome and of the salt-dependent superstructures of chromatin. J Cell Biol 83: 403–427
Turner BM (1982) Immunofluorescent staining of human metaphase chromosomes with monoclonal antibody to histone H2B. Chromosoma 87: 345–357
Turner BM (1989) Acetylation and deacetylation of histone H4 continue through metaphase with depletion of more-acetylated isoforms and altered site usage. Exp Cell Res 182: 206–214
Turner BM, Fellows G (1989) Specific antibodies reveal ordered and cell-cycle-related use of histone-H4 acetylation sites in mammalian cells. Eur J Biochem 179: 131–139
Turner BM, O'Neill LP, Allan IM (1989) Histone H4 acetylation in human cells. Frequency of acetylation at different sites defined by immunolabelling with site-specific antibodies. FEBS Lett 253: 141–145
Weintraub H, vanLente F (1974) Dissection of chromosome structure with trypsin and nucleases. Proc Natl Acad Sci USA 71: 4249–4253
Whitfield GF, Fellows G, Turner BM (1986) Characterization of monoclonal antibodies to histone 2B. Localization of epitopes and analysis of binding to chromatin. Eur J Biochem 157: 513–521
Wu RS, Panusz HT, Hatch CL, Bonner WM (1986) Histones and their modifications. CRC Crit Rev Biochem 20: 201–263
Zhang XY, Horz W (1982) Analysis of highly purified satellite DNA containing chromatin from the mouse. Nucleic Acids Res 10: 1481–1494
Author information
Authors and Affiliations
Additional information
by W.C. Earnshaw
Rights and permissions
About this article
Cite this article
Jeppesen, P., Mitchell, A., Turner, B. et al. Antibodies to defined histone epitopes reveal variations in chromatin conformation and underacetylation of centric heterochromatin in human metaphase chromosomes. Chromosoma 101, 322–332 (1992). https://doi.org/10.1007/BF00346011
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00346011