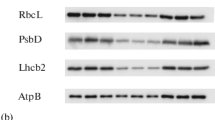
Summary
Carotenoid deficient, maize seedlings ordinarily incur extensive photooxidative damage to their chloroplasts. This damage can be minimized by growing seedlings in very dim light. Cytosolic mRNA encoding a prevalent chloroplast protein, the light-harvesting chlorophyll a/b protein (LHCP), accumulates under low-intensity light, but rapidly disappears when plants are exposed to higher intensity light. Transcription studies were performed in vitro on nuclei isolated from carotenoid deficient seedlings grown first in low-intensity light and then transferred to higher intensity light. The rate of LHCP gene transcription was rapidly reduced in high intensity light. We propose that a signal of chloroplast origin is a necessary component of optimal transcription of LHCP genes and other nuclear genes encoding chloroplast proteins. Our data indicate that the signal originates at an early stage of chloroplast development, is continuously required, for LHCP gene transcription and has a short half-life. Photooxidative damage to the chloroplast destroys its capacity for further synthesis of the signal. A significant diurnal fluctuation in LHCP transcriptional activity was also observed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- LHCP:
-
ligh-harvesting chlorophyll a/b protein
- SSu:
-
small subunit
- PEP:
-
phosphoenolpyruvate
- CPI:
-
photosystem I chlorophyll a protein
References
Anderson IC, Robertson DS (1960) Role of carotenoids in protecting chlorophyll from photodestruction. Plant Physiol 35:531–534
Bachmann MD, Robertson DS, Bowen CC, Anderson IC (1973) Chloroplast ultrastructure in pigment-deficient mutants of Zea mays under reduced light. J Ultrastruct Res 45:384–406
Batschauer A, Mösinger E, Kreuz K, Dorr I, Apel K (1986) The implication of a plastid-derived factor in the transcriptional control of nuclear genes encoding the light-harvesting chlorophyll a/b protein. Eur J Biochem, 154:625–634
Blume DE, McClure JW (1980) Developmental effects of Sandoz 6707 on activities of enzymes of phenolic and general metabolism in barley shoots grown in the dark or under low or high intensity light. Plant Physiol 65:238–244
Börner T, Mendel RR, Schiemann J (1986) Nitrate reductase is not accumulated in chloroplast-ribosome-deficient mutants of higher plants. Planta 169:202–207
Burgess DG, Taylor WC (1987) Chloroplast photooxidation affects the accumulation of cytosolic mRNAs encoding chloroplast proteins in maizer Planta 170:520–527
Feierabend J, Winkelhusener T (1982) Nature of photooxidative events in leaves treated with chlorosis-inducing herbicides. Plant Physiol 70:1277–1282
Fish LE, Kuck U, Bogorad L (1985) Two partially homologous light-inducible maize chloroplast genes encoding polypeptides of the P700 chlorophyll a-protein complex of photosystem I. J Biol Chem 260:1413–1421
Guifoyle TJ, Dietrich MA (1987) In: Bruening G, Harada J, Kosuge T, Hollander A (eds) Trailoring genes for crop improvement. Plenum, Press, New York, pp 87–100
Harpster MH, Taylor WC (1986) Maize phosphoenolpyruvate carboxylase: Cloning and characterization of mRNAs encoding isozymic forms. J Biol Chem 261:6132–6136
Harpster MH, Mayfield SP, Taylor WC (1984) Effects of pigmentdeficient mutants on the accumulation of photosynthetic proteins in maize. Plant Mol Biol. 3:59–71
Kelley PM, Tolan DR (1986) The complete amino acid sequence for the anaerobically induced aldolase from maize derived from cDNA clones. Plant Physiol 82:1076–1080
Luthe DS, Quatrano ES (1980) Transcription in isolated wheat nuclei: I. Isolation of nuclei and elimination of endogenous ribonuclease activity. Plant Physiol 65:305–308
Marks MD, Lindell JS, Larkins BA (1985) Nucleotide sequence analysis of zein mRNAs from maize endosperm. J Biol Chem 260:16451–16459
Martineau B, Taylor WC (1985) Photosynthetic gene expression and cellular differentiation in developing maize leaves. Plant Physiol 78:399–404
Mascia P (1978) An analysis of precursors accumulated by several chlorophyll biosynthetic mutants of maize. Mol Gen Genet 161:237–244
Mayfield SP, Taylor WC (1984) Carotenoid-deficient, maize seedlings fail to accumulate light-harvesting chlorophyll a/b binding protein (LHCP) mRNA. Eur J Biochem 144:79–84
Mayfield SP, Taylor WC (1987) Chloroplast photooxidation inhibits the expression of a set of nuclear genes. Mol Gen Genet 208:309–314
Mayfield SP, Nelson T, Taylor WC (1986a) The fate of chloroplast proteins during photooxidation in carotenoid-deficient maize leaves Plant Physiol 82:760–764
Mayfield SP, Nelson T, Taylor WC, Malkin R (1986b) Carotenoid synthesis and pleiotropic effects in carotenoid-deficient seedlings of maize. Planta 169:23–32
Oelmüller R, Mohr R (1986) Photooxidative destruction of chloroplasts and its consequences for expression of nuclear genes. Planta 167:106–113
Oelmüller R, Levitan I, Bergfeld R, Rajasekhar BK, Mohr H (1986) Expression of nuclear genes as affected by treatments acting on the plastids. Planta 168:482–492
Piechulla B, Pichersky E, Cashmore AR, Gruissem W (1986) Expression of nuclear and plastid genes for photosynthesis-specific proteins during tomato fruit development and ripening. Plant Mol Biol 7:367–376
Rajasekhar VK, Mohr H (1986) Appearance of nitrite reductase in cotyledons of the mustard (Sinapis alba L.) seedlings as affected by nitrate, phytochrome and photooxidative damage of plastids. Planta 168:369–376
Reiss T, Bergfeld R, Link G, Thien W, Mohr H (1983) Photooxidative destruction of chloroplasts and its consequences for cytosolic enzyme levels and plant development. Planta 159:518–528
Simpson J, van Montague M, Herrera-Estrella L (1986) Photosynthesis-associated gene families: Differences in response to tissue-specific and environmental factors. Science 233:34–38
Taylor WC, Mayfield SP (1985) In: Steinback KL, Arntzen CJ, Bogorad L, Bonitz S (eds) Molecular biology of the photosynthetic apparatus. Cold Spring Harbor Laboratory. Cold Spring Harbor, New York, pp 413–416
Taylor WC, Burgess DG, Mayfield SP (1986) The use of carotenoid deficiencies to study nuclear-chloroplast regulatory interactions. In: Randall DD, Miles CD, Nelson CJ, Blevins DG, Miernyk JA (eds) Current topics in plant biochemistry and physiology, vol 5. Univ Missouri Press, Columbia, pp 117–127
Author information
Authors and Affiliations
Additional information
Communicated by R.B. Goldberg
Rights and permissions
About this article
Cite this article
Burgess, D.G., Taylor, W.C. The chloroplast affects the transcription of a nuclear gene family. Mol Gen Genet 214, 89–96 (1988). https://doi.org/10.1007/BF00340185
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00340185