Summary
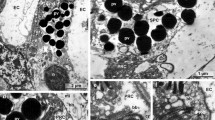
The Limulus ommatidium consists of 4 to 20 retinula cells surrounding the dendrite of the eccentric cell. Adjoining membranes are differentiated into the microvillous rhabdome in the central area of the ommatidium. Three types of pigment cells envelop the sensory cells. The distal pigment cells cover the periphery of the distal half of the ommatidium; proximal pigment cells (beneath the base of the ommatidium) and intraommatidial pigment cells provide glial wrapping for the sensory cells, the partitions between them, and the peripheral loose framework. Processes of the overlying cone cells penetrate into the ommatidium and lie at the edges of the rhabdomal fins. Numerous neurosecretory axons terminate at all levels of the ommatidium on pigment cells, conveyed there either by enveloping pigment cells or by separate neuroglial cells. Tight junctions in the ommatidium are confined to the contacts between rhabdomal miorovilli. The periphery of the rhabdome is surrounded by continuous adhering junctions except at the tip and exit of the eccentric cell dendrite. The discussion centers on possible correlations between known neurophysiological characteristics of ommatidial cells and significant morphological aspects of the ommatidium, such as distribution of supporting cells, extracellular space, and junctional specializations.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barr, L., M. M. Dewey, and W. Berger: Propagation of action potentials and the structure of the nexus in cardiac muscle. J. gen. Physiol. 48, 797–823 (1965).
Behrens, M. E., and V. J. Wulff: Light initiated responses of retinula and eccentric cells in the Limulus lateral eye. J. gen. Physiol. 48, 1081–1093 (1965).
Bennett, M. V. L., Y. Nakajima, and G. D. Pappas: Physiology and ultrastructure of electrotonic junctions. I. Supramedullary neurons. J. Neurophysiol. 30, 161–179 (1967a).
—: Physiology and ultrastructure of electrotonic junctions. III. Giant electromotor neurons of Malapterurus electricus. J. Neurophysiol. 30, 209–235 (1967b).
—, G. D. Pappas, E. Aljure, and Y. Nakajima: Physiology and ultrastructure of electrotonic junctions. II. Spinal and medullary electromotor nuclei in mormyrid fish. J. Neurophysiol. 30, 180–209 (1967c).
—, M. Gimenez, and Y. Nakajima: Physiology and ultrastructure of electrotonic junctions. IV. Medullary electromotor nuclei in gymnotid fish. J. Neurophysiol. 30, 236–300 (1967d).
Berlin, J. D.: The localization of acid mucopolysaccharides in the Golgi complex of intestinal goblet cells. J. Cell Biol. 32, 760–768 (1967).
Bonnett, H. T. J., and E. H. Newcomb: Coated vesicles and other cytoplasmic components of growing root hairs of radish. Protoplasma (Wien) 62, 59–75 (1966).
Bullock, T. H., and G. A. Horridge: Structure and function in the nervous systems of invertebrates. 1719 pp. San Francisco: W. H. Freeman & Co. 1965.
Dowling, J. E.: Discrete potentials in the dark-adapted eye of the crab Limulus. Nature (Lond.) 217, 28–31 (1968).
Dumont, J. N., E. Anderson, and E. Chomyn: The anatomy of the peripheral nerve and its ensheathing artery in the horseshoe crab, Xiphosura (Limulus) polyphemus. J. Ultrastruct. Res. 13, 38–64 (1965).
Eguchi, E., and T. H. Waterman: Fine structure patterns in crustacean rhabdoms. In: The functional organization of the compound eye (C. G. Bernhard, ed.) p. 105–124. Oxford: Pergamon Press 1966.
—: Changes in retinal fine structure induced in the crab Libinia by light and dark adaptation. Z. Zellforsch. 79, 209–229 (1967).
—: Cellular basis for polarized light perception in the spider crab, Libinia. Z. Zellforsch. 84, 87–101 (1968).
Fahrenbach, W. H.: The fine structure of a nauplius eye. Z. Zellforsch. 62, 182–197 (1964).
—: The morphology of the eyes of Limulus. I. Cornea and epidermis of the compound eye. Z. Zellforsch. 87, 278–291 (1968).
—, L. B. Sanderberg, and E. G. Cleary: Ultrastructural studies on early elastogenesis. Anat. Rec. 155, 563–576 (1966).
Farquhar, M. G., and G. E. Palade: Junctional complexes in various epithelia. J. Cell Biol. 17, 375–412 (1963).
Fernández-Morán, H.: Cell membrane ultrastructure. Low temperature electron microscopy and X-ray diffraction studies of lipoprotein components in lamellar system. Circulation 26, 1039–1065 (1962).
Friend, D. S., and M. G. Farquhar: Functions of coated vesicles during protein absorption in the rat vas deferens. J. Cell Biol. 35, 357–376 (1967).
Fuortes, M. G. F.: Electric activity of cells in the eye of Limulus. Amer. J. Ophthal. 46, 210–223 (1958).
—: Initiation of impulses in visual cells of Limulus. J. Physiol. (Lond.) 148, 14–28 (1959).
Furshpan, E., and D. D. Potter: Transmission at the giant motor synapses of the crayfish. J. Physiol. (Lond.) 145, 289–325 (1959).
Goldsmith, T. H.: Fine structure of the retinulae in the compound eye of the honeybee. J. Cell Biol. 14, 494 (1962).
Gordon, G. B., L. R. Miller, and K. G. Bensch: Studies on the intracellular digestive process in mammalian tissue culture cells. J. Cell Biol. 25, Pt. 2, 41–55 (1965).
Grenacher, H.: Untersuchungen über das Sehorgan der Arthropoden, insbesondere der Spinnen, Insecten und Crustaceen. 188 pp. Göttingen: Vandenhoek & Ruprecht 1879.
Griffin, J. L.: Fixation and visualization of microfilaments and microtubules and their significance in the movement of four types of ameboid cells (Abstr.). J. Cell Biol. 27, 39A (1965).
Harper, E., S. Seifter, and B. Scharrer: Electron microscopic and biochemical characterization of collagen in blattarian insects. J. Cell Biol. 33, 385–393 (1967).
Hartline, H. K., N. A. Coulter, and H. G. Wagner: Effects of electric current on responses of single photoreceptor units in the eye of Limulus. Fed. Proc. 11, 65–66 (1952a).
—, G. A. Wagner, and E. F. Macnichol: The peripheral origin or nervous activity in the visual system. Cold Spr. Harb. Symp. quant. Biol. 17, 125–141 (1952b).
—, H. G. Wagner, and F. Ratliff: Inhibition in the eye of Limulus. J. gen. Physiol. 39, 651–673 (1956).
Hesse, R.: Untersuchungen über die Organe der Lichtempfindung bei niederen Thieren. VII. Von den Arthropoden-Augen. Z. wiss. Zool. 70, 347–473 (1901).
Horridge, G. A.: The retina of the locust. In: the functional organization of the compound eye (C. G. Bernhard, ed.), p. 513–541. Oxford: Pergamon Press 1966.
—, and P. B. T. Barnard: Movement of palisade in locust retinula cells when illuminated. Quart. J. micr. Sci. 107, 135–136 (1965).
Hubbard, R., and G. Wald: Visual pigment of the horseshoe crab, Limulus polyphemus. Nature (Lond.) 186, 212–215 (1960).
Kuffler, S. W., and D. D. Potter: Glia in the leech central nervous system: physiological properties and neuron-glia relationship. J. Neurophysiol. 27, 290–320 (1964).
Lane, N., L. Caro, L. R. Otero-Vilardebó, and G. C. Godman: On the site of sulfation in colonic goblet cells. J. Cell Biol. 21, 339–351 (1964).
Lasansky, A.: Cell junctions in ommatidia of Limulus. J. Cell Biol. 33, 365–384 (1967).
Lipetz, L. E.: Response pathways to electric stimulation in the Limulus eye. Am. J. Ophthal. 46, Ser. 3, No. 3, pt. 2, 5–19 (1958).
—: The Limulus eye as an information converter: Mechanisms for the transfer of information from the light image to the optic nerve discharge. Advanc. biol. med. Phys. 7, 131–173 (1960).
Macnichol, E. F.: Visual receptors as biological transducers. In: Molecular structure and functional activity of nerve cells (R. G. Grenell and L. J. Mullins, ed.), p. 1–34. Washington: American Institute of Biological Sciences Publications 1956.
Manton, I.: Observations on the fine structure of the zoospore and young germling of Stigeoclonium. J. exp. Bot. 15, 399–411 (1964).
Maunsbach, A. B.: The influence of different fixatives and fixation methods on the ultrastructure of rat kidney proximal tubule cells. I. Comparison of different perfusion fixation methods and of glutaraldehyde, formaldehyde and osmium tetroxide fixatives. J. Ultrastruct. Res. 15, 242–282 (1966a).
—: The influence of different fixatives and fixation methods on the ultrastructure of rat kidney proximal tubule cells. II. Effects of varying osmolality, ionic strength, buffer system and fixative concentration of glutaraldehyde solutions. J. Ultrastruct. Res. 15, 283–309 (1966b)
Melamed, J., and O. Trujillo-Cenóz: The fine structure of the visual system of Lycosa (Araneae: Lycosidae). I. Retina and optic nerve. Z. Zellforsch. 74, 12–31 (1966).
Miller, W. H.: The neural structure and function of an invertebrate eye (Abstr.) Bull. Johns Hopk. Hosp. 91, 72 (1952).
—: Comparative study of fine structure of some invertebrate photoreceptors (Abstr.). Science 126, 1234 (1957a).
—: Morphology of the ommatidia of the compound eye of Limulus. J. biophys. biochem. Cytol. 3, 421–427 (1957b).
—: Fine structure of some invertebrate photoreceptors. Ann. N. Y. Acad. Sci. 74, 204–209 (1958).
—, F. Ratliff, and H. K. Hartline: How cells receive stimuli. Sci. Amer. 205, 222–238 (1961).
Neutra, M., and C. P. Leblond: Synthesis of the carbohydrate of mucus in the Golgi complex as shown by electron microscope radioautography of goblet cells from rats injected with glucose-H3. J. Cell Biol. 30, 119–136 (1966a).
—, and C. P. Leblond: Radioautographic comparison of the uptake of galactose-H3 and glucose-H3 in the Golgi region of various cells secreting glycoproteins or mucopolysaccharides. J. Cell Biol. 30, 137–150 (1966b).
Newcomb, E. H.: A spiny vesicle in slime-producing cells of the bean root. J. Cell Biol. 35, No. 2, Pt. 1, C17-C22 (1967).
Nicholls, J. G., and S. W. Kuffler: Extracellular space as a pathway for exchange between blood and neurons in the central nervous system of the leech: ionic composition of glial cells and neurons. J. Neurophysiol. 27, 645–671 (1964).
Nilsson, S. E. G.: The ultrastructure of the receptor outer segments in the retina of the leopard frog (Rana pipiens). J. Ultrastruct. Res. 12, 207–231 (1965).
Pappas, G. D., Y. Asada, and M. V. L. Bennett: Morphological and physiological changes in junctional sites of crayfish septate axons (Abstr.). Anat. Rec. 157, 297 (1967).
Peterson, M., and C. P. Leblond: Synthesis of complex carbohydrates in the Golgi region as shown by radioautography after injection of labeled glucose. J. Cell Biol. 21, 143–148 (1964).
Peterson-Neutra, M.: Synthesis of complex carbohydrates in Golgi saccules: an electron microscope-autoradiographic study (Abstr.). Anat. Rec. 151, 399 (1965).
Revel, J. P., and M. J. Karnovsky: Hexagonal array of subunits in intercellular junctions of the mouse heart and liver (Abstr.). J. Cell Biol. 33, C7 (1967).
Robertson, J. D.: Recent electron microscope observations on the ultrastructure of the crayfish median-to-motor giant synapse. Exp. Cell Res. 8, 226–229 (1955).
—: Ultrastructure of excitable membranes and the crayfish median-giant synapse. Ann. N. Y. Acad. Sci. 94, 339–389 (1961).
Rosenbluth, J.: Subsurface cisternae and their relationship to the neuronal plasma membrane. J. Cell Biol. 13, 405–421 (1962).
Roth, T. F., and K. R. Porter: Yolk protein uptake in the oocyte of the mosquito Aedes aegypti L. J. Cell Biol. 20, 313–332 (1964).
Ruck, P.: On photoreceptor mechanisms of retinula cells. Biol. Bull. 123, 618–634 (1962).
Smith, T. G., F. Baumann, and M. G. F. Fuortes: Electrical connections between visual cells in the ommatidium of Limulus. Science 147, 1446–1448 (1965).
—, and J. E. Brown: A photoelectric potential in invertebrate cells. Nature (Lond.) 212, 1217–1219 (1966).
Sorokin, S. P.: Centrioles and the formation of rudimentary cilia by fibroblasts and smooth muscle cells. J. Cell Biol. 15, 363–378 (1962).
Stieve, H.: Interpretation of the generator potential in terms of ionic processes. Cold Spr. Harb. Symp. quant. Biol. 30, 451–456 (1965).
Tomita, R., R. Kikuchi, and I. Tanaka: Excitation and inhibition in lateral eye of horseshoe crab. In: Electrical activity of single cells (Y. Katsuki, ed.), p. 11–23. Tokyo: Igakushoin 1960.
Trujillo-Cenóz, O.: Some aspects of the structural organization of the arthropod eye. Cold Spr. Harb. Symp. quant. Biol. 30, 371–382 (1965).
Waddington, C. H., and M. M. Perry: The ultra-structure of the developing eye of Drosophila. Proc. roy. Soc. B 153, 155–178 (1960).
Watáse, S.: On the morphology of the compound eye of arthropods. Studies Biol. Lab. Johns. Hopk. Univ. 4, 387–334 (1890).
Waterman, T. H., and C. A. G. Wiersma: The functional relation between retinal cells and optic nerve in Limulus. J. exp. Zool. 126, 59–85 (1954).
White, R. H.: The effect of light and light deprivation upon the ultrastructure of the larval mosquito eye. II. The rhabdom. J. exp. Zool. 166, 405–425 (1967).
Wolbarsht, M. L., and S. Yeandle: Visual processes in the Limulus eye. Ann. Rev. Physiol. 29, 513–542 (1967).
Author information
Authors and Affiliations
Additional information
This study constitutes publication No. 329 from the Oregon Regional Primate Research Center, supported by Grants FR00163 and NB07717-01 from the National Institutes of Health and by a Bob Hope Fight-for-Sight Grant-in-Aid of the National Council to Combat Blindness, Inc.
The author wishes to thank Mrs. Audrey Griffin for patient and excellent technical assistance, Mr. Joel Ito for the execution of the drawing, and Dr. C. J. Russell for constructive criticism.
Rights and permissions
About this article
Cite this article
Fahrenbach, W.H. The morphology of the eyes of Limulus . Z. Zellforsch. 93, 451–483 (1968). https://doi.org/10.1007/BF00338531
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00338531