Summary
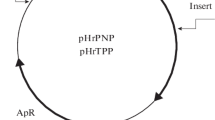
The hemB gene of Escherichia coli K12, coding for porphobilinogen synthase (PBG-S; syn., 5-aminolevulinic acid dehydratase, ALA-D), was cloned following insertion of an EcoRI fragment of plasmid F′13 into the mobilizable vector pCR1. The hybrid plasmid carrying the hemB gene was able to complement a hemB mutant of E. coli K12: not only was the PBG-S activity of the mutant restored after the acquisition of the hemB gene, but it was about ten times higher than that of the wild type. Subcloning of the original EcoRI fragment (14.6 kb) enabled us to locate the hemB gene on an Nrul-HpaI fragment of about 1.1 kb. The hemB promoter was located toward the NruI end of the fragment, as shown by the use of the pKO promoter-probe series of vectors. Sequencing of the hemB gene indicated the presence of an open reading frame (ORF) of 1051 nucleotides, which should correspond to the HemB protein. Primer extension experiments enabled us to identify the 5′ end of the hemB mRNA, and to deduce the-10 and-35 regions of the hemB promoter. Protein synthesis performed by an in vitro coupled transcription-translation system, showed the presence of a protein of about 35 kDa. This is in agreement with the molecular weight of the HemB protein (35.6 kDa), as deduced from the nucleotide sequence of the gene. Comparison of the amino acid sequences of E. coli and human PBG-S allowed the detection of several regions of strong homology between the two proteins. Two of these regions correspond, as expected, to the putative zinc-binding and catalytic sites of the human PBG-S.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aiba H, Adhya S, de Crombrugghe B (1981) Evidence for two functional gal promoters in intact Escherichia coli cells. J Biol Chem 256:11905–11910
Aldrich TL, Chakrabarty AM (1988) Transcriptional regulation, nucleotide sequence, and localization of promoter of the catBC operon in Pseudomonas putida. J Bacteriol 170:1297–1304
Anderson PM, Desnick RJ (1979) Purification and properties of Δ-aminolevulinate dehydrase from human erythrocytes. J Biol Chem 254:6924–6930
Barnard GF, Itoh R, Hohbergr LH, Shemin D (1977) Mechanism of porphobilinogen synthase. Possible role of essential thiol groups. J Biol Chem 252:8965–8974
Birnboim HC, Doly J (1979) A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res 7:1513–1523
Bishop JR, Cohen PJ, Boyer SH, Noyes AN, Frelin LP (1986) Isolation of a rat liver Δ-aminolevulinate dehydratase (ALAD) cDNA clone: Evidence for unequal ALAD gene dosage among inbred mouse strains. Proc Natl Acad Sci USA 83:5568–5572
Coleman DL (1966) Purification and properties of Δ-aminolevulinate dehydratase from tissues of two strains of mice. J Biol Chem 241:5511–5517
Dayhoff MO, Eck RV, Park CM (1972) A model of evolutionary change in proteins. In: Dayhoff MO (ed) Atlas of protein sequence and structure 1972, vol. 5. National Biomedical Research Foundation, Washington DC, pp 89–99
Dymetryszyn J (1985) Clonage du gène hemB chez E. coli K-12. Memoire, M.Sc., Université de Montréal
Gibbs PNB, Jordan PM (1981) 5-aminolaevulinate dehydratase: zinc-65 binding and exchange with the enzyme from human erythrocytes. Biochem Soc Trans 9:232–233
Gibbs PNB, Jordan PM (1986) Identification of lysine at the active site of human 5-aminolaevulinate dehydratase. Biochem J 236:447–451
Granick S, Beale SI (1978) Hemes, chlorophylls, and related compounds: biosynthesis and metabolic regulation. Adv Enzymol 46:33–203
Gornall AG, Bardawill CJ, David MM (1949) Determination of serum proteins by means of the Biuret reaction. J Biol Chem 177:751
Hansen JB, Olsen RH (1978) Isolation of large plasmids and characterization of the P2 incompatibility group plasmids pMG1 and pMG5. J Bacteriol 135:227–238
Holmes DS, Quigley M (1981) A rapid boiling method for the preparation of bacterial plasmids. Anal Biochem 114:193–197
Hu S, Ohtsubo E, Davidson N (1975) Electron microscope heteroduplex studies of sequence relations among plasmids of Escherichia coli: Structure of F13 and related F-primes. J Bacteriol 122:749–763
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Li JM, Umanoff H, Proenca R, Russell CS, Cosloy SD (1988) Cloning of the Escherichia coli K-12 hemB gene. J Bacteriol 170:1021–1025
Liedgens W, Lutz C, Schneider HAW (1983) Molecular properties of 5-aminolevulinic acid dehydratase from Spinacia olereiea. Eur J Biochem 135:75–79
Low KB (1972) Escherichia coli K-12 F-prime factors, old and new. Bacteriol Rev 36:587–607
Mandel M, Higa J (1970) Calcium-dependent bacteriophage DNA infection. J Mol Biol 53:159–162
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Maralihalli GB, Rao SR, Bhagwat AS (1985) Histidine residues at the active site of maize Δ-aminolevulinic acid dehydratase. Phytochem 24:2533–2536
McConville ML, Charles HP (1979) Isolation of haemin-requiring mutants of Escherichia coli K-12. J Gen Microbiol 113:155–164
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Nandi DL, Shemin D (1968) Δ-aminolevulinic acid dehydratase of Rhodopseudomonas spheroides. J Biol Chem 243:1231–1235
Nandi DL, Shemin D (1973) Δ-aminolevulinic acid dehydratase of Rhodopseudomonas capsulata. Arch Biochem Biophys 158:305–311
Norrander J, Kempe T, Messing J (1983) Construction of improved M13 vectors using oligodeoxynucleotide-directed mutagenesis. Gene 26:101–106
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Sasarman A, Horodniceanu T (1967) Locus determining normal colony formation on the chromosome of Escherichia coli K-12. J Bacteriol 94:1268–1269
Sasarman A, Surdeanu M, Szegli G, Horodniceanu T, Greceanu V, Dumitrescu A (1968) Hemin deficients mutants of Escherichia coli K-12. J Bacteriol 96:570–572
Sasarman A, Nepveu A, Echelard Y, Dymetryszyn J, Drolet M, Goyer C (1987) Molecular cloning and sequencing of the hemD gene of Escherichia coli K-12 and preliminary data on the Uro operon. J Bacteriol 169:4257–4262
Sassa S (1982) Delta-aminolevulinic acid dehydratase assay. Enzyme 28:133–145
Shibata H, Ochiai H (1976) Purification and properties of Δ-aminolevulinic acid dehydratase from radish cotyledons. Plant Cell Physiol 18:421–429
Silhavy TJ, Berman ML, Enquist LW (1984) Experiments with gene fusions. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Staden R (1982) Automation of the computer handling of gel reading data produced by the shotgun method of DNA sequencing. Nucleic Acids Res 10:4731–4751
Thomas SD, Jordan PM (1986) Nucleotide sequence of the hemC locus encoding porphobilinogen deaminase of Escherichia coli K12. Nucleic Acids Res 14:6215–6226
Tsukamoto I, Yoshinaga T, Sano S (1979) The role of zinc with special reference to the essential thiol groups in Δ-aminolevulinic acid dehydratase of bovine liver. Biochim Biophys Acta 570:167–178
Vögtli M, Hütter R (1987) Characterization of the hydroxystreptomycin phosphotransferase gene (sph) of Streptomyces glaucescens: nucleotide sequence and promoter analysis. Mol Gen Genet 208:195–203
Wetmur JG, Bishop DF, Ostasiewicz L, Desnick RJ (1986a) Molecular cloning of cDNA for human Δ-aminolevulinic acid dehydratase. Gene 43:123–130
Wetmur JG, Bishop DF, Cantelmo C, Desnick RJ (1986b) Human Δ-aminolevulinate dehydratase: Nucleotide sequence of a full-length cDNA clone. Proc Natl Acad Sci USA 83:7703–7707
Wu WH, Shemim D, Richardo KE, Williams RC (1974) The quaternary structure of Δ-aminolevulinic acid dehydratase from bovine liver. Proc Natl Acad Sci USA 71:1767–1770
Author information
Authors and Affiliations
Additional information
Communicated by H. Hennecke
Rights and permissions
About this article
Cite this article
Echelard, Y., Dymetryszyn, J., Drolet, M. et al. Nucleotide sequence of the hemB gene of Escherichia coli K12. Mol Gen Genet 214, 503–508 (1988). https://doi.org/10.1007/BF00330487
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00330487