Summary
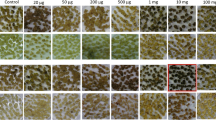
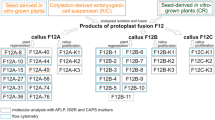
Protoplasts isolated from a 5-methyltryptophan resistant nonregenerable Nicotiana tabacum (L.) Xanthi suspension culture were fused via the dextran method (Kameya et al. 1981) with protoplasts isolated from N. glutinosa L. leaf mesophyll cells. Prospective somatic hybrids were selected by their ability to produce chlorophyll and to grow on medium containing 5-methyltryptophan and eventually to regenerate into complete plants. Twenty-eight plants regenerated from selected colonies were classified into three groups: (1) Four plants had intermediate morphology, were sterile, had hybrid isozyme patterns for four different enzymes, and contained fraction 1 protein large subunit patterns indicative of a N. glutinosa chloroplast origin. (2) One plant had N. tabacum morphology, was fertile, had two hybrid isozyme patterns, and contained fraction 1 protein large subunits indicative of the N. tabacum chloroplast type. Although this hybrid contained both parental small subunit patterns of fraction 1 protein, two-dimensional gel electrophoresis showed the N. glutinosa-type small subunit to consist of several molecular weights suggestive of errors in the processing steps. Progeny of the Type 2 hybrid (selfed) contained no N. glutinosa characteristics. (3) Twenty-three plants were N. glutinosa escapes. Chromosome numbers ranged from 60 (some chromosome loss) in two hybrids to 34–35 (extensive chromosome loss) in three hybrids.
These results suggest that amino acid analog resistance and regeneration capability can be utilized as complementing markers for selecting and identifying somatic hybrids after protoplast fusion. Mesophyll protoplasts from N. glutinosa can also be regenerated into morphologically normal fertile plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- 5MT:
-
DL-5-methyltryptophan
- GOT:
-
glutamate oxaloacetate transaminase
- LAP:
-
leucine aminopeptidase
- PER:
-
peroxidase
- RuBPCase:
-
ribulose bisphosphate carboxylase
- PVP:
-
polyvinylpyrolidone
- IEF:
-
isoelectric focusing
- PVPP:
-
polyvinylpolypyrolidone
References
Brewbaker JL, Upadhya MD, Makinen Y, MacDonald T (1968) Isoenzyme polymorphism in flowering plants. III. Gel electrophoretic methods and applications. Physiol Plant 21:930–940
Chua NH (1980) Electrophoretic analysis of chloroplast proteins. Methods Enzymol 69:434–446
Douglas GC, Wetter LR, Nakamura C, Keller WA, Setterfield G (1981) Somatic hybridization between Nicotiana rustica and N. tabacum. III. Biochemical, morphological, and cytological analysis of somatic hybrids. Can J Bot 59:228–237
Evans DA, Wetter LR, Gamborg OL (1980) Somatic hybrid plants of Nicotiana glauca and Nicotiana tabacum obtained by protoplast fusion. Physiol Plant 48:225–230
Harms CT, Potrykus I, Widholm JM (1981) Complementation and dominant expression of amino acid analogue resistance markers in somatic hybrid clones from Daucus carota after protoplast fusion. Z Pflanzenphysiol 101:377–390
Harms CT, Oertli JJ, Widholm JM (1982) Characterization of amino acid analogue resistant somatic hybrid cell lines of Daucus carota L. Z Pflanzenphysiol 106:239–249
Kameya T, Horn ME, Widholm JM (1981) Hybrid shoot formation from fused Daucus carota and D. capillifolius protoplasts. Z Pflanzenphysiol 104:459–466
Kameya T (1982) The method for fusion with dextran. In: Fujiwara A (ed) Proc 5th Intl Congr Plant Tissue and Cell Culture. Maruzen Co Ltd, Tokyo, Japan, pp 613–614
Kao KN (1975) A chromosomal staining method for cultured cells. In: Gamborg OL, Wetter LR (eds) Plant tissue culture methods. National Research Council of Canada, Saskatoon, Saskatchewan, pp 63–64
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
O'Farrell PH (1975) High resolution two-dimensional gel electrophoresis of proteins. J Biol Chem 250:4007–4021
Thomas E, King PJ, Potrykus I (1979) Improvement of crop plants via single cells in vitro — an assessment. Z Pflanzenzüchtg 82:1–30
Uchimiya H (1982) Somatic hybridization between male sterile Nicotiana tabacum and N. glutinosa through protoplast fusion. Theor Appl Genet 61:69–72
Uchimiya H, Akiyama T, Ohgawara T, Harada H (1982) Expression of nuclear and cytoplasmic genes in the progeny of a somatic hybrid between male sterile Nicotiana tabacum and N. glutinosa. Jpn J Genet 57:343–347
Wetter LR (1977) Isoenzyme patterns in soybean-Nicotiana somatic hybrid cell lines. Mol Gen Genet 150:231–235
White DWR, Vasil IK (1979) Use of amino acid analogue-resistant cell lines for selection of Nicotiana sylvestris somatic hybrids. Theor Appl Genet 55:107–112
Widholm JM (1972) Cultured Nicotiana tabacum cells with an altered anthranilate synthetase which is less sensitive to feedback inhibition. Biochim Biophys Acta 261:52–58
Widholm JM (1973) Measurement of the five enzymes which convert chorismate to tryptophan in cultured Daucus carota cell extracts. Biochim Biophys Acta 320:217–226
Widholm JM (1980) Differential expression of amino acid biosynthetic control isoenzymes in plants and cultured cells. In: Sala F, Parisi B, Cella R, Ciferri O (eds) Plant cell cultures: results and perspectives. Elsevier/North Holland Biomedical Press, Amsterdam, pp 157–159
Widholm JM (1982) Selection of protoplast fusion hybrids. In: A Fujiwara (ed) Proc 5th Intl Congr Plant Tissue and Cell Culture. Maruzen Co Ltd, Tokyo, Japan, pp 609–612
Author information
Authors and Affiliations
Additional information
Communicated by R.B. Goldberg
Rights and permissions
About this article
Cite this article
Horn, M.E., Kameya, T., Brotherton, J.E. et al. The use of amino acid analog resistance and plant regeneration ability to select somatic hybrids between Nicotiana tabacum and N. glutinosa . Molec Gen Genet 192, 235–240 (1983). https://doi.org/10.1007/BF00327672
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00327672