Summary
We tested the prediction that plants grown in elevated CO2 environments are better able to compensate for biomass lost to herbivory than plants grown in ambient CO2 environments. The herbaceous perennial Plantago lanceolata (Plantaginaceae) was grown in either near ambient (380 ppm) or enriched (700 ppm) CO2 atmospheres, and then after 4 weeks, plants experienced either 1) no defoliation; 2) every fourth leaf removed by cutting; or 3) every other leaf removed by cutting. Plants were harvested at week 13 (9 weeks after simulated herbivory treatments). Vegetative and reproductive weights were compared, and seeds were counted, weighed, and germinated to assess viability.
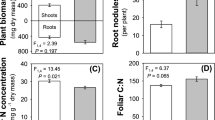
Plants grown in enriched CO2 environments had significantly greater shoot weights, leaf areas, and root weights, yet had significantly lower reproductive weights (i.e. stalks + spikes + seeds) and produced fewer seeds, than plants grown in ambient CO2 environments. Relative biomass allocation patterns further illustrated differences in plants grown in ambient CO2 environments. Relative biomass allocation patterns further illustrated differences in plant responses to enriched CO2 atmospheres: enriched CO2-grown plants only allocated 10% of their carbon resources to reproduction whereas ambient CO2-grown plants allocated over 20%. Effects of simulated herbivory on plant performance were much less dramatic than those induced by enriched CO2 atmospheres. Leaf area removal did not reduce shoot weights or reproductive weights of plants in either CO2 treatment relative to control plants. However, plants from both CO2 treatments experienced reductions in root weights with leaf area removal, indicating that plants compensated for lost above-ground tissues, and maintained comparable levels of reproductive output and seed viability, at the expense of root growth.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Antonovics J (1980) Concepts of resource allocation and partitioning in plants. In: Staddon JER (ed) Limits to Action: the Allocation of Individual Behavior. Academic Press, NY pp 1–34
Bazzaz FA (1990) Response of natural ecosystems to the rising global CO2 level. Annu Rev Ecol Syst 21: 167–196
Bazzaz FA, Garbutt K, Williams WE (1985) Effects of increased atmospheric carbon dioxide concentration on plant communities. In: Strain BR, Cure JD (eds) Direct Effects of Increased Carbon Dioxide on Vegetation. US Depart Energy, Washington, DC, pp 155–170
Belsky AJ (1986) Does herbivory benefit plants? A review of evidence. Am Nat 127: 870–892
Carter DR, Peterson KM (1983) Effects of a CO2-enriched atmosphere on the growth and competitive interactions of a C3 and a C4 grass. Oecologia 58: 188–193
Chapin FS, McNaughton SJ (1989) Lack of compensatory growth under phosphorus deficiency in grazing-adapted grasses from the Serengeti Plains. Oecologia 79: 551–557
Curtis PS, Drake BG, Leadley PW, Arp WJ, Whigham DF (1989) Growth and senescence in plant communities exposed to elevated CO2 concentrations on an estuarine marsh. Oecologia 78:20–26
Fajer ED, Bowers MD, Bazzaz FA (1989) The effects of enriched carbon dioxide atmospheres on plant/insect herbivore interactions. Science 243: 1198–1200
Garbutt K, Bazzaz FA (1984) The effects of elevated CO2 on plants. III. Flower, fruit and seed production and abortion. New Phytol 98:433–445
Garbutt K, Williams WE, Bazzaz FA (1990) Analysis of the differential response of five annuals to elevated CO2 during growth. Ecology 71:1185–1194
Harper JL (1977) Population biology of plants. Academic Press, London, pp 892
Kramer PJ (1981) Carbon dioxide concentration, photosynthesis and dry matter production. Bioscience 31:29–33
Lemon ER (1983) CO2 and plants: the response of plants to rising levels of atmospheric carbon dioxide. AAAS, Selected Symposium 84. Westview Press, Boulder, CO
Lincoln DE, Couvet D, Sionit N (1986) Response of an insect herbivore to host plants grown in enriched carbon dioxide atmospheres. Oecologia 69:566–570
Maschinski J, Whitham TG (1989) The continuum of plant responses to herbivory: the influence of plant association, nutrient availability, and timing. Am Nat 134:1–19
Mihaliak CA, Lincoln DE (1989) Plant biomass partitioning and chemical defense: response to defoliation and nitrate limitation. Oecologia 80:122–126
Paez A, Hellmers H, Strain BR (1980) CO2 effects on apical dominance in Pisum sativum. Physiol Plant 50:43–46
Primack RB, Antonovics J (1982) Experimental ecological genetics in Plantago. VII. Reproductive effort in populations of P. lanceolata L. Evolution 36:742–752
Solbrig OT (1981) Studies on the population biology of the genus Viola. II. The effect of plant size on the fitness in Viola sororia. Evolution 35:1080–1093
Wulff RD, Alexander HM (1985) Intraspecific variation in the response to CO2 enrichment in seeds and seedlings of Plantago lanceolata L. Oecologia 66:458–460
Zangerl AR, Bazzaz FA (1984) The response of plants to elevated CO2. II. Competitive interactions among annual plants under varying light and nutrients. Oecologia 62:412–417
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fajer, E.D., Bowers, M.D. & Bazzaz, F.A. Performance and allocation patterns of the perennial herb, Plantago lanceolata, in response to simulated herbivory and elevated CO2 environments. Oecologia 87, 37–42 (1991). https://doi.org/10.1007/BF00323777
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00323777