Abstract
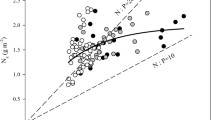
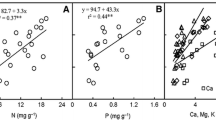
Among species, photosynthetic capacity (Amax) is usually related to leaf nitrogen content (N), but variation in the species-specific relationship is not well understood. To address this issue, we studied Amax-N relationships in 23 species in adjacent Amazonian communities differentially limited by nitrogen (N), phosphorus (P), and/or other mineral nutrients. Five species were studied in each of three late successional forest types (Tierra Firme, Caatinga and Bana) and eight species were studied on disturbed sites (cultivated and early secondary successional Tierra Firme plots). Amax expressed on a mass basis (Amass) was correlated (p<0.05) with Nmass in 17 of 23 species, and Amax on an area basis (Aarea) was correlated (p<0.05) with Narea in 21 of 23 species. The slopes of Amax-N relationships were greater and intercepts lower for disturbance adapted early successional species than for late successional species. On a mass basis, the Amax-N slope averaged ≈15 μmol CO2 [g N]-1 s-1 for 7 early secondary successional species and ≈4 μmol CO2 [g N]-1 s-1 for 15 late successional species, respectively. Species from disturbed sites had shorter leaf life-span and greater specific leaf area (SLA) than late successional species. Across all 23 species, the slope of the Amass-Nmass relationship was related (p<0.001) positively to SLA (r2=0.70) and negatively to leaf life-span (r2=0.78) and temporal niche during secondary succession (years since cutting-and-burning, r2=0.90). Thus, disturbance adapted early successional species display a set of traits (short leaf life-span, high SLA and Amax and a steep slope of Amax-N) conducive to resource acquisition and rapid growth in their high resource regeneration niches. The significance and form of the Amax-N relationship were associated with the relative nutrient limitations in the three late successional communities. At species and community levels, Amax was more highly dependent on N in the N-limited Caatinga than in the P-and N-limited Bana and least in the P-and Ca-limited Tierra Firme on oxisol-and differences among these three communities in their massbased Amax-N slope reflects this pattern (6.0, 2.4, and 0.7 μmol CO2 [g N]-1 s-1, respectively). Among all 23 species, the estimated leaf Nmass needed to reach compensation (net photosynthesis ≈ zero) was positively related to the Amass-Nmass slope and to dark respiration rates and negatively related to leaf life-span. Variation among species in the Amax-N slope was well correlated with potential photosynthetic N use efficiency, Amax per unit leaf N. The dependence of Amax on N and the form of the relationship vary among Amazonian species and communities, consistent with both relative availabilities of N, P, and other mineral nutrients, and with intrinsic ecophysiological characteristics of species adapted to habitats of varying resource availability.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Chapin FS III (1980) The mineral nutrition of wild plants. Ann Rev Ecol Syst 11:233–260
Chazdon RL, Field CB (1987) Determinants of photosynthetic capacity in six rainforest Piper species. Oecologia 73:222–230
Cuevas E, Medina E (1986) Nutrient dynamics within amazonian forest ecosystems. I. Nutrient flux in fine litterfall and efficiency of nutrient utilization. Oecologia 68:466–472
Cuevas E, Medina E (1988) Nutrient dynamics within amazonian forest ecosystems. II. Fine root growth, nutrient availability and leaf litter decomposition. Oecologia 76:222–235
DeJong TM, Day KR, Johnson RS (1989) Partitioning of leaf nitrogen with respect to within canopy light exposure and nitrogen availability in peach (Prunus persica). Trees 3:89–95
Ellsworth DS (1991) Variation in leaf structure, nitrogen, and photosynthesis across light gradients in a temperate and tropical forest. Ph.D. Dissertation, University of Wisconsin, 146 pp
Ellsworth DS, Reich PB (1992) Leaf mass per area, nitrogen content and photosynthetic carbon gain in Acer saccharum seedlings in contrasting forest light environments. Functional Ecology 6:423–435
Ellsworth DS, Reich PB (1993) Canopy structure and vertical patterns of photosynthesis and related leaf traits in a deciduous forest. Oecologia 96:169–178
Evans JR (1989) Photosynthesis and nitrogen relationships in leaves of C3 plants. Oecologia 78:9–19
Field C, Mooney HA (1986) The photosynthesis-nitrogen relationship in wild plants. In: Givnish T (ed) On the economy of plant form and function, Cambridge University Press, pp 25–55
Field C, Merino J, Mooney HA (1983) Compromises between water-use efficiency and nitrogen-use efficiency in five species of California evergreens. Oecologia 60:84–89
Grubb PJ (1977) The maintenance of species richness in plant communities and the importance of the regeneration niche. Biol Rev 52:107–145
Gulmon SL, Chu CC (1981) The effects of light and nitrogen on photosynthesis, leaf characteristics and dry matter allocation in the Chaparral shrub Diplacus aurantiacus. Oecologia 49:207–212
Lloyd J, Syvertsen JP, Kriedemann PE, Farquhar GD (1992) Low conductances for CO2 diffusion from stomata to the sites of carboxylation in leaves of woody species. Plant Cell Environ 15:873–899
Medina E, Cuevas E (1990) Patterns of nutrient accumulation and release in Amazonian forests of the upper Rio Negro basin. In: Proctor J (ed) Mineral Nutrients in Tropical Forests and Savann Ecosystems, Blackwell Scientific Publications, Oxford, UK pp 217–240
Montagnini F, Buschbacher R (1989) Nitrification rates in two undisturbed tropical rain forests and three slash-and-burn sites of the Venezuelan Amazon. Biotropica 21:9–14
Poorter H, Remkes C, Lambers H (1990) Carbon and nitrogen economy of 24 wild species differing in relative growth rate. Plant Physiol 94:621–627
Reich PB, Schoettle AW (1988) Role of phosphorus and nitrogen in photosynthetic and whole plant carbon gain and nutrientuse efficiency in eastern white pine. Oecologia 77:25–33
Reich PB, Walters MB (1994) Photosynthesis-nitrogen relation in Amazonian tree s ecies. II. Variation in nitrogen vis-a-vis specific leaf area influences mass- and area-based expressions. Oecologia 97:73–81
Reich PB, Abrams MD, Ellsworth DS, Kruger EL, Tabone T (1990) Fire affects ecophysiology and community dynamics of central Wisconsin oak forest regeneration. Ecology 71:2179–2190
Reich PB, Uhl C, Walters MB, Ellsworth DS (1991a) Leaf lifespan as a determinant of leaf structure and function among 23 tree species in Amazonian forest communities. Oecologia 86:16–24
Reich PB, Walters MB, Ellsworth DS (1991b) Leaf age and season influence the relationships between leaf nitrogen, leaf mass per area, and photosynthesis in maple and oak trees. Plant, Cell and Environment 14:251–259
Reich PB, Walters MB, Ellsworth DS (1992) Leaf life-span in relation to leaf, plant and stand processes in diverse ecosystems. Ecological Monographs 62:365–392
Sage RF, Pearcy R (1987) The nitrogen use efficiency of C3 and C4 plants. II. Leaf nitrogen effects on the gas exchange characteristics of Chenopodium album (L.) and Amaranthus retroflexus (L.). Plant Physiol 84:959–963
SAS (1985) SAS User's Guide: Statistics, Version 5, ed, S.P. Joyner, SAS Institute, Inc., Cary, NC
Saldarriaga JG, West DC, Tharp ML, Uhl C (1988) Long-term chronosequence of forest succession in the upper Rio Negro of Colombia and Venezuela. J Ecol 76:938–958
Sanford R, Saldarriaga J, Clark K, Uhl C, Herrera R (1985) Amazon rain-forest fires. Science 227:53–55
Uhl C (1987) Factors controlling succession following slash-and-burn agriculture in Amazonia. J Ecol 75:377–407
Vitousek PM, Field CB, Matson PA (1990) Variation in foliar d13C in Hawaiian Metrosideros polymorpha: a case of internal resistance? Oecologia 84:362–370
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Reich, P.B., Walters, M.B., Ellsworth, D.S. et al. Photosynthesis-nitrogen relations in Amazonian tree species. Oecologia 97, 62–72 (1994). https://doi.org/10.1007/BF00317909
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00317909