Summary
The topography of the funnel-canal organs of Carcinus maenas (Decapoda, Crustacea) and their stimulus-receiving cuticular and sensory apparatus were studied in the light and electron microscopes.
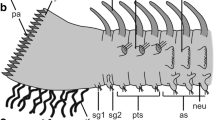
About 4000 funnel-canal organs are situated within the exoskeleton of Carcinus. Almost all of them are on the distal segments of the walking legs, in particular on the epicuticular cap at the tip of the dactyl. They were not found to be arranged in groups or sensilla fields, and no sex-specific differences were observed.
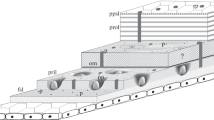
Characteristic features of the funnel-canal organs are as follows: (a) There is a terminal pore (0.5×0.8 μm diameter) in the cuticle, at the tip of a small projection. It is closed by a plug of electron-dense material. (b) The terminal sections of the dendrites are enclosed in a dendritic sheath up to ca. 10 μm below the pore. (c) The dendrites, 3–24 in number, end below the plug; none of the dendrites exhibits a tubular body; two of the dendrites are distinguished from the others by the greater number of microtubules in their outer segments.
The structural characteristics, in particular the “gustatory pore” and the number of dendrites, are typical of bimodal receptors in arthropods. In such receptors, as in the contact chemoreceptors of insects and arachnids, mechano-and chemosensitive sensory cells are combined.
This interpretation of the function of the funnel-canal organs is supported by electrophysiological data of other authors.
The morphological parameters we find for the funnelcanal organs, in comparison with those of insect campaniform sensilla, provide clear evidence against the reclassification of the funnel-canal organs as “crustacean campaniform organs” proposed by Shelton and Laverack (1968).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Altner H (1977) Insektensensillen: Bau- und Funktionsprinzipien. Verh Dtsch Zool Ges 1977, pp 139–153
Altner H, Prillinger L (1980) Ultrastructure of invertebrate chemothermo-and hygroreceptors and its functional significance. Int Review Cytol 67:69–139
Altner I, Hatt H, Altner H (1983) Structural properties of bimodal chemo- and mechanosensitive setae on the pereiopod chelae of the crayfish, Austropotamobius torrentium. Cell Tissue Res 228:357–374
Barth FG (1971) Der sensorische Apparat der Spaltsinnesorgane (Cupiennius salei Keys. Araneae). Z Zellforsch 112:212–246
Barth FG (1972) Die Physiologie der Spaltsinnesorgane II. Funktionelle Morphologie eines Mechanorezeptors. J Comp Physiol 81:159–186
Barth FG (1980) Campaniform sensilla: another vibration receptor in the crab leg. Naturwiss 67:201
Barth FG (1981) Strain detection in the arthropod exoskeleton. In: Laverack MS, Cosens DJ (eds) Sense organs. Blackie, Glasgow London pp 112–141
Barth FG, Libera W (1970) Ein Atlas der Spaltsinnesorgane von Cupiennius salei Keys. Chelicerata (Araneae). Z Morphol Tiere 68:343–369
Blaney WM, Chapman RF (1969) The fine structure of the terminal sensilla on the maxillary palps of Schistocerca gregaria (Forskal) (Orthoptera, Acrididae). Z Zellforsch 99:74–97
Burck HC (1973) Histologische Technik. Thieme, Stuttgart
Case J, Gwilliam GT (1961) Amino acid sensitivity of the dactyl chemoreceptors of Carcinides maenas. Biol Bull Woods Hole 121:449–455
Chapman KM, Duckrow RB, Moran DT (1973) Form and role of deformation in excitation of an insect mechanoreceptor. Nature 244:453–454
Derby CD (1982) Structure and function of cuticular sensilla of the lobster Homarus americanus. J Crust Biol 2:1–21
Dumpert K, Gnatzy W (1977) Cricket combined mechanoreceptors and kicking response. J Comp Physiol 122:9–25
Foelix RF, Chu-Wang I-Wu (1973a) The morphology of spider sensilla I. Mechanoreceptors. Tissue Cell 5:451–460
Foelix RF, Chu-Wang I-Wu (1973b) The morphology of spider sensilla II. Chemoreceptors. Tissue Cell 5:461–478
Gaffal KP (1979) An ultrastructural study of the tips of four classical bimodal sensilla with one mechanosensitive and several chemosensitive receptor cells. Zoomorphologie 92:273–291
Gnatzy W, Schmidt M (1982) Sind die Trichterkanalorgane die ‘campaniformen Sensillen’ der Crustaceen? Verh Dtsch Zool Ges 1982. Gustav Fischer, Stuttgart, p 249
Gnatzy W, Tautz J (1980) Ultrastructure and mechanical properties of an insect mechanoreceptor: Stimulus-transmitting structures and sensory apparatus of the cercal filiform hairs of Gryllus. Cell Tissue Res 213:441–463
Hansen K (1978) Insect Chemoreception. In: Hazelbauer GL (ed) Receptors and Recognition B. 5. Chapman and Hall, London, pp 233–292
Hansen K, Heumann HG (1971) Die Feinstruktur der tarsalen Schmeckhaare der Fliege Phormia terranovae Rob.-Desv. Z Zellforsch 117:419–442
Krämer K, Markl H (1978) Flight-inhibition on ground contact in the american cockroach, Periplaneta americana. I. Contact receptors and a model for their central connections. J Insect Physiol 24:577–586
Laverack MS (1963) Aspects of chemoreception in crustacea. Comp Biochem Physiol 8:141–151
Laverack MS (1976) External proprioceptors. In: Mill PJ (ed) Structure and function of proprioceptors in the invertebrates. Chapman and Hall, London, pp 1–63
Leduc E, Bernhard W (1967) Recent modifications of the glycol methacrylate embedding procedure. J Ultrastruct Res 19:196–199
Luther W (1930) Versuche über die Chemorezeption der Brachyuren. Z Vergl Physiol 12:177–205
Pflugstaedt H (1912) Die Halteren der Dipteren. Z Wiss Zoologie 100:2–59
Pringle JWS (1955) The function of the lyriform organs of arachnids. J Exp Biol 32:270–278
Reynolds ES (1963) The use of lead citrate at high pH as an electron opaque stain in electron microscopy. J Cell Biol 17:208–212
Romeis B (1968) Mikroskopische Technik. R Oldenbourg, München
Richards AG (1951) The Integument of Arthropods. University of Minnesota Press, Minneapolis
Schmidt K, Gnatzy W (1972) Die Feinstruktur der Sinneshaare auf den Cerci von Gryllus bimaculatus Deg (Saltatoria, Gryllidae). III. Die kurzen Borstenhaare. Z Zellforsch 126:206–222
Schmidt M, Gnatzy W (in press) Are the funnel-canal organs the “campaniform sensilla” of the shore crab Carcinus maenas (Crustacea, Decapoda) II. Ultrastructure. Cell Tissue Res
Shelton RGJ, Laverack MS (1968) Observations on a redescribed crustacean cuticular sense organ. Comp Biochem Physiol 25:1049–1059
Stürckow B (1967) Occurence of a viscous substance at the tip of the labellar taste hair of the blowfly. In: Hayashi T (ed) ‘Olfaction and Taste II. Pergamon Press, New York, pp 707–720
Thurm U, Stedtler A, Foelix R (1975) Reizwirksame Verformung der Terminalstrukturen eines Mechanorezeptors. Verh Dtsch Zool Ges 67, pp 37–41
Weatherby TM (1981) Ultrastructural study of the sinus gland of the crab Cardisoma carnifex. Cell Tissue Res 220:295–312
Zacharuk RY (1980) Ultrastructure and function of insect chemosensilla. Ann Rev Entomol 25:27–47
Zill SN, Moran DT (1981) The exoskeleton and insect proprioception. I. Responses of tibial campaniform sensilla to external and musclegenerated forces in the american cockroach Periplaneta americana. J Exp Biol 91:1–24
Author information
Authors and Affiliations
Additional information
Supported by the Deutsche Forschungsgemeinschaft (W.G., SFB 45/A1)
We thank Professor Dr. F.G. Barth for valuable discussion and Mr. K. Grommet for drawing the Figs. 1 and 6c
Rights and permissions
About this article
Cite this article
Gnatzy, W., Schmidt, M. & Römbke, J. Are the funnel-canal organs the “campaniform sensilla” of the shore crab Carcinus maenas (Crustacea, Decapoda)?. Zoomorphology 104, 11–20 (1984). https://doi.org/10.1007/BF00312166
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00312166