Abstract
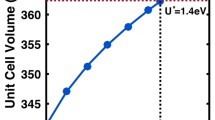
Tetrahedrites of composition (Cu, Ag)10(Cu2, Fe, Zn)2(Sb, As)4S13 or Cu12Sb14/3S13 have 208 valence electrons per unit cell and are expected to be semiconductors. The bands are full in these cases, whereas compositions towards the classical formula Cu12Sb4S13 (204 valence electrons per unit cell) have only partially filled bands and are therefore expected to be metallic. These predictions are supported by new optical absorption spectra of tetrahedrites with 205 and 208 valence electrons per unit cell. The gap between valence and conduction bands of the semiconductor is about 1.7 (±0.2) eV. A further prediction based on a nearly-free electron model is that 208 valence electrons per unit cell represent a compositional limit for tetrahedrites, and that the stability increases as compositions approach this limit. Existing data indicate an exponential increase in the number of occurrences as the limit is approached.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Johnson ML, Jeanloz R (1983) A Brillouin-zone model for compositional variation in tetrahedrite. Am Mineral 68:220–226
Knittle E, Jeanloz R (1984) Structural and bonding changes in CsI at high pressures Science 223:53–56
Pankove JI (1971) Optical Processes in Semiconductors. New York: Dover
Pauling L, Neumann EW (1934) The crystal structure of binnite, (Cu, Fe)12As4S13, and the chemical composition and structure of minerals in the tetrahedrite group. Z Kristallogr 88:544–562
Shuey RT (1975) Semiconducting Ore Minerals. Amsterdam: Elsevier
Wernick JH, Benson KE (1957) New semiconducting ternary compounds. J Phys Chem Solids 3:157–159
Author information
Authors and Affiliations
Additional information
A.P. Sloan Foundation Fellow
Rights and permissions
About this article
Cite this article
Jeanloz, R., Johnson, M.L. A note on the bonding, optical spectrum and composition of tetrahedrite. Phys Chem Minerals 11, 52–54 (1984). https://doi.org/10.1007/BF00309375
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00309375