Summary
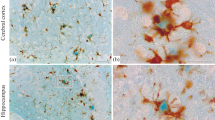
Several studies have demonstrated that the accurate visualization and quantification of pathological lesions in neurodegenerative disorders depend on the reliability of staining methods. In an attempt to gain a better assessment of the density and distribution of the neuropathological markers of Alzheimer's disease, we compared the staining efficiency of a modified thioflavine S protocol for neurofibrillary tangles (NFT) and senile plaques (SP) to different argentic impregnation techniques (Bielchowsky, Gallyas, Globus, Campbell-Switzer-Martin) and to immunohistochemical stainings obtained with two different antibodies against the amyloid β protein A4 and the microtubule-associated tau protein. The modified thioflavine S technique (MTST) detects up to 60% more SP and up to 50% more NFT than the Bielschowsky and Globus methods, respectively. The results obtained with the specific antibodies are comparable to those obtained with the MTST, but these immunotechniques are more expensive and time consuming for routine neuropathological evaluation, and the appropriate antibodies are not always commercially available. Furthermore, the morphological appearance of NFT and SP with MTST is greatly improved when compared to the classical thioflavine S and the increased signal-to-noise ratio between specifically stained structures and background permits an accurate semi-automatic quantification.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arnold SE, Hyman BT, Flory J, Damasio AR, Van Hoesen GW (1991) The topographical and neuroanatomical distribution of neurofibrillary tangles and neurite plaques in the cerebral cortex of patients with Alzheimer's disease. Cerebral Cortex 1:103–116
Behrouz N, Défossez A, Delacourte A, Hublau P, Mazzuca M (1989) Alzheimer's disease: glycolytic pretreatment dramatically enhances immunolabeling of senile plaques and cerebrovascular amyloid substance. Lab Invest 61:576–580
Benson DF, Davis RJ, Snyder BD (1988) Posterior cortical atrophy. Arch Neurol 45:789–793
Bielschowsky M (1904) Die Silberimprägnation der Neurofibrillen. J Psychol Neurol 3:169–188
Braak H, Braak E, Grundke-Iqbal I, Iqbal K (1986) Occurrence of neuropil threads in the senile human brain and in Alzheimer's disease: a third location of paired helical filaments outside of neurofibrillary tangles and neuritic plaques. Neurosci Lett 65:351–355
Campbell SK, Switzer RC, Martin TL (1987) Alzheimer's plaques and tangles: a controlled and enhanced method. Soc Neurosci Abstr 13:678
Défossez A, Beauvillain JC, Delacourte A, Mazzuca M (1990) Alzheimer's disease: a new evidence for common epitopes between monotubule associated protein tau and paired helical filaments (PHF): demonstration at the electron microscope by a double immunogold labelling. Virchows Arch [A] 413:141–145
Delacourte A, Flament S, Dibe EM, Hublau P, Sablonnière B, Hémon B, Scherrer V, Défossez A (1990) Pathological proteins Tau 64 and 69 are specifically expressed in the somatodendritic domain of the degenerating cortical neurons during Alzheimer's disease. Demonstration with a panel of antibodies against Tau proteins. Acta Neuropathol 80:111–117
Duyckaerts C, Brion JP, Hauw JJ, Flament-Durand J (1987) Quantitative assessment of the density of neurofibrillary tangles and senile plaques in senile dementia of the Alzheimer type. Comparison of immunocytochemistry with a specific antibody and Bodian's protargol method. Acta Neuropathol (Berl) 73:167–170
Duyckaerts C, Delaère P, Hauw JJ, Abbamondi-Pinto AL, Sorbi S, Allen I, Brion JP, Flament-Durand J, Duchen L, Kauss J, Schlote W, Lowe J, Probst A, Ravid R, Swaab DF, Renkawek K, Tomlinson B (1990) Rating of the lesions in senile dementia of the Alzheimer type: concordance between laboratories. A European multicenter study under the auspices of EURAGE. J Neurol Sci 97:295–323
Flament S, Delacourte A, Delaère P, Duyckaerts C, Hauw JJ (1990) Correlation between microscopical changes and tau 64 and 69 biochemical detection in senile dementia of the Alzheimer type. Tau 64 and 69 are reliable markers of the neurofibrillary degeneration. Acta Neuropathol 80:212–215
Gallyas F (1971) Siver staining of Alzheimer's neurofibrillary changes by means of physical development. Acta Morphol Acad Sci Hung 19:1–8
Gentleman SM, Bruton C, Allsop D, Lewis SJ, Polak JM, Roberts GW (1989) A demonstration of the advantages of immunostaining in the quantification of amyloid plaque deposits. Histochemistry 92:355–358
Globus JH (1927) The Cajal and Hortega glia staining methods. Arch Neurol Psychiatry 8:263–271
Golaz J, Bouras C, Hof PR (1991) Motor cortex involvement in presenile dementia: report of a case. J Geriatr Psychiatry Neurol (in press)
Guntern R, Bouras C, Vallet PG, Hof PR (1989) An improved staining method for senile plaques and neurofibrillary tangles in Alzheimer's disease: quantitative comparison with other techniques. Soc Neurosci Abstr 15:1038
Guntern R, Vallet PG, Bouras C, Constantinidis J (1989) An improved immunohistostaining procedure for peptides in human brain. Experientia 45:159–161
Guntern R, Bouras C, Hof PR, Vallet PG (1991) An improved staining method for senile plaques and neurofibrillary tangles in Alzheimer's disease. Experientia (in press)
Hauw JJ, Duyckaerts C, Delaère P (1988) Neuropathology of aging and DAT: how can age-related changes be distinguished from those due to disease processes. In: Henderson AS, Henderson JH (eds) Etiology of dementia of Alzheimer's type. John Wiley & Sons, Chichester, pp 195–211
Henderson VW, Finch CE (1989) The neurobiology of Alzheimer's disease. J Neurosurg 70:335–353
Hof PR, Bouras C (1991) Object recognition deficit in Alzheimer's disease: possible disconnection of the occipitotemporal component of the visual system. Neurosci Lett 122:53–56
Hof PR, Morrison JH (1990) Quantitative analysis of a vulnerable subset of pyramidal neurons in Alzheimer's disease. II. Primary and secondary visual cortex. J Comp Neurol 301:55–64
Hof PR, Bouras C, Constantinidis J, Morrison JH (1989) Balint's syndrome in Alzheimer's disease: specific disruption of the occipito-parietal visual pathway. Brain Res 493:368–375
Hof PR, Bouras C, Constantinidis J, Morrison JH (1990) Selective disconnection of specific visual association pathways in cases of Alzheimer's disease presenting with Balint's syndrome. J Neuropathol Exp Neurol 49:168–184
Hof PR, Cox K, Morrison JH (1990) Quantitative analysis of a vulnerable subset of pyramidal neurons in Alzheimer's disease. I. Superior frontal and inferior temporal cortex. J comp Neurol 301:44–54
Hof PR, Knabe R, Bovier P, Bouras C (1991) Neuropathological observations in a case of autism presenting with self-injury behavior. Acta Neuropathol (in press)
Hof PR, Perl DP, Loerzel AJ, Morrison JH (1991) Neurofibrillary tangles distribution in the cerebral cortex of Parkinson dementia cases from Guam: differences with Alheimer's disease. Brain Res (in press)
Ikeda SI, Allsop D, Glenner GG (1989) Morphology and distribution of plaque and related deposits in the brains of Alzheimer's disease and control cases. Lab Invest 60:113–122
Jagust WJ, Davies P, Tillerborcich JK (1990) Focal Alzheimer's disease. Neurology 40:14–19
Katzman R, Saitoh T (1991) Advances in Alzheimer's disease. FASEB J 5:278–286
Khachaturian ZS (1985) Diagnosis of Alzheimer's disease. Arch Neurol 42:1097–1104
Kim KS, Miller DL, Sapienza VG, Chen CJ, Vai C, Grundke-Iqbal I, Curry JR, Wisniewski HM (1988) Production and characterization of monoclonal antibodies reactive to synthetic cerebrovascular amyloid peptide. Neurosci Res Commun 2:121–130
Lamy C, Duyckaerts P, Delaère P, Payan CH, Fermanian J, Poulain V, Hauw JJ (1989) Comparison of seven staining methods for senile plaques and neurofibrillary tangles in a prospective series of 15 elderly patients. Neuropathol Appl Neurobiol 15:563–578
Lewis DA, Campbell MJ, Terry RD, Morrison JH (1987) Laminar and regional distribution of neurofibrillary tangles and neuritic plaques in Alzheimer's disease: a quantitative study of visual cortex and auditory cortices. J Neurosci 7:1799–1808
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA work group under the auspices of the Department of health and Human services task Force on Alzheimer's disease. Neurology 34:939–944
Mirra SS, Heyman A, McKeel D, Sumi SM, Crain BJ, Brownlee LM, Vogel FS, Hughes JP, van Belle G, Berg L and CERAD neuropathologists (1991) The Consortium to establish a registry for Alzheimer's disease (CERAD). II. Standardization of the neuropathologic assessment of Alzheimer's disease. Neurology 41:479–486
Mutrux S (1947) Diagnostic différentiel histologique de la maladie d'Alzheimer et de la démence sénile-Pathophobie de la zone de projection corticale. Monatsschr Psychiatr Neurol 113:100–117
Pappola MA, Omar RA, Robakis NK (1991) Immunohistochemical demonstration of antioxidant enzymes in Alzheimer's disease. Am J Pathol (in press)
Pearson RCA, Esiri MM, Hiorns RW, Wilcock GK, Powell TPS (1985) Anatomical correlates of the distribution of the pathological changes in the neocortex in Alzheimer's disease. Proc Natl Acad Sci USA 82:4531–4534
Rogers J, Morrison JH (1985) Quantitative morphology and regional and laminar distributions of senile plaques in Alzheimer's disease. J Neurosci 5:2801–2808
Rudelli RD, Ambler MV, Wisniewski HM (1984) Morphology and distribution of Alzheimer neuritic (senile) and amyloid plaques in striatum and diencephalon. Acta Neuropathol (Berl) 64:273–281
Wilcock GK, Esiri MM (1982) Plaques, tangles and dementia. A quantitative study. J Neurol Sci 56:343–356
Wisniewski HM, Rabe A, Zigman W, Silverman W (1989) Neuropathological diagnosis of Alzheimer disease. J Neuropathol Exp Neurol 48:606–609
Wisniewski HM, Wen GY, Kim KS (1989) Comparison of four staining methods on the detection of neuritic plaques. Acta Neuropathol 78:22–27
Yamaguchi H, Hirai S, Morimatsu M, Shoji M, Harigaya Y (1988) Diffuse type of senile plaques in the brains of Alzheimer-type dementia. Acta Neuropathol 77:113–119
Yamamoto T, Hirano A (1986) A comparative study of modified Bielschowsky, Bodian and thioflavine S stains on Alzheimer's neurofibrillary tangles. Neuropathol Appl Neurobiol 12:3–9
Author information
Authors and Affiliations
Additional information
Supported in part by the Brookdale Foundation and the American Health Assistance Foundation (to P.R.H.) and the NIH (AG05138 and AGO8200 to N.K.R.)
Rights and permissions
About this article
Cite this article
Vallet, P.G., Guntern, R., Hof, P.R. et al. A comparative study of histological and immunohistochemical methods for neurofibrillary tangles and senile plaques in Alzheimer's disease. Acta Neuropathol 83, 170–178 (1992). https://doi.org/10.1007/BF00308476
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00308476