Summary
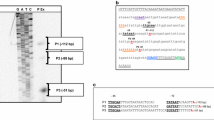
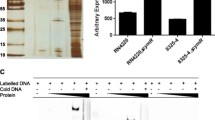
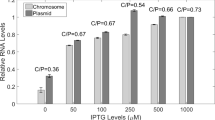
Transcriptional fusions were constructed between the promoter for the epidermolytic toxin A (eta) gene of Staphylococcus aureus and the luxAB and xylE reporter gene systems. The expression of the fusion products was found to be dependent upon the accessory gene regulator (agr) locus and was observed to increase significantly during the transition from the exponential to the stationary phase of growth. Furthermore the expression of the eta gene promoter was found to be osmotically regulated, with the expression levels of the eta fusions being inversely related to the osmolyte levels. The ability of environmental factors to influence DNA topology (and thence gene expression) was investigated. High osmolarity (0.7 M NaCl) resulted in an increase in the degree of negative supercoiling of plasmid DNA in the S. aureus strain 8325-4 (Agr+) but not in strain ISP546 (Agr−). Furthermore the eta promoter was strongly induced in S. aureus cultures grown in the presence of sub-inhibitory concentrations of novobiocin, a DNA gyrase inhibitor. However this induction was independent of agr, suggesting that the eta promoter is subject to both agr-dependent (osmolarity, growth phase) and -independent (DNA topology) regulatory processes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahmed KA, Stewart GSAB (1988) Cloning of the lux genes into Lactobacillus casei and Streptococcus lactis: phosphate-dependent light production. Biochem Soc Trans 16:1068
Ahmed KA, Stewart GSAB (1991) The production of bioluminescent lactic acid bacteria suitable for the rapid assessment of starter culture activity in milk. J Appl Bacteriol 70:113–120
Balke V, Gralla J (1987) Changes in the linking number of super-coiled DNA accompany growth transitions in E. coli. J Bacteriol 169:4499–4506
Bayles KW, Iandolo JJ (1989) Genetic and molecular analysis of the gene encoding staphylococcal enterotoxin D. J Bacteriol 171:4799–4806
Bernardini ML, Fontaine A, Sansonetti PJ (1990) The two component regulatory system OmpR-EnvZ controls the virulence of Shigella flexneri. J Bacteriol 172:6274–6281
Bjorklind A, Arvidson S (1980) Mutants of Staphylococcus aureus affected in the regulation of exoprotein synthesis. FEMS Lett 7:203–206
Chatfield SN, Dorman CJ, Hayward C, Dougan G (1991) Role of ompR dependent genes in Salmonella typhimurium virulence: mutants deficient in OmpC and OmpF are attenuated in vivo. Infect Immun 59:449–452
Dorman CJ (1991) DNA supercoiling and environment regulation of gene expression in pathogenic bacteria. Infect Immun 59:745–749
Dorman CJ, Barr GC, Ni Bhriain N, Higgins CF (1988) DNA supercoiling and the anaerobic and growth phase regulation of tonB expression. J Bacteriol 170:2816–2826
Dorman CJ, Chatfield S, Higgins CF, Hayward C, Dougan G (1989) Characterisation of porin and ompR mutants of a virulent strain of Salmonella typhimurium: ompR mutants are attenuated in vivo. Infect Immun 57:2136–2140
Drlica K (1990) Bacterial topoisomerases and the control of DNA supercoiling. Trends Genet 6:433–437
Easmon CSF, Adlam C (eds) (1983) Staphylococci and staphylococcal infections. vols 1 and 2. Academic Press, London
Galan JE, Curtiss R III (1990) Expression of Salmonella typhimurium genes required for invasion is regulated by changes in DNA supercoiling. Infect Immun 58:1879–1885
Gaskill ME, Khan SA (1988) Regulation of enterotoxin B gene in Staphylococcus aureus. J Biol Chem 263:6276–6280
Gasson MJ, Anderson PH (1985) High copy number plasmid vectors for use in lactic streptococci. FEMS Lett 30:193–196
Higgins CF, Dorman CJ, Stirling DA, Waddell L, Booth IR, May G, Bremer E (1988) A physiological role for DNA supercoiling in the osmotic regulation of gene expression in Salmonella typhimurium and E. coli. Cell 52:569–584
Hopewell R, Oram M, Briesewitz R, Fischer LM (1990) DNA cloning and organisation of the Staphylococcus aureus gyrA and gyrB genes: close homology among proteins and implications for 4-quinolone action and resistance. J Bacteriol 172:3481–3484
Janzon L, Arvidson S (1990) The role of the δ-lysin gene (hld) in the regulation of virulence genes by the accessory gene regulator (agr) of Staphylococcus aureus. EMBO J 9:1391–1399
Janzon L, Lofdahl S, Arvidson S (1986) Evidence for a coordinate transcriptional control of alpha-toxin and protein A synthesis in Staphylococcus aureus. FEMS Lett 33:193–198
Janzon L, Lofdahl S, Arvidson S (1989) Identification and nucleotide sequence of the delta-lysin gene, hld, adjacent to the accessory gene regulator (agr) of Staphylococcus aureus. Mol Gen Genet 219:480–485
Kreiswirth B, Lofdahl S, Betley MJ, O'Reilly M, Schlievert PM, Bergdoll MS, Novick RP (1983) The toxic shock syndrome exotoxin structural gene is not transmitted by a prophage. Nature 305:709–712
Lee CY, Schmidt JJ, Johnson-Winegar AD, Spero L, Iandolo JJ (1987) Sequence determination and comparison of the exfoliative toxin A and toxin B genes from Staphylococcus aureus. J Bacteriol 169:3904–3909
Mahmood R, Khan SA (1990) Role of upstream sequences in the expression of the staphylococcal enterotoxin B gene. J Biol Chem 2655:4652–4656
Mallonee DH, Glatz BA, Pattee PA (1982) Chromosomal mapping of a gene affecting enterotoxin A production in Staphylococcus aureus. Appl Environ Microbiol 43:397–402
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning: a laboratory manual. Cold Spring Harbour Laboratory Press, Cold Spring Habour, New York
Meighen EA (1988) Enzymes and genes from the lux operons of bioluminescent bacteria. Annu Rev Microbiol 42:151–176
Menzel R, Gellert M (1987) Fusions of the Escherichia coli gyrA and gyrB control regions to the galactokinase gene are inducible by coumermycin treatment. J Bacteriol 169:1272–1278
Miller JF, Mekalanos JJ, Falkow S (1989) Coordinate regulation and sensory transduction in the control of bacterial virulence. Science 243:916–922
Morfeldt T, Janzon L, Arvidson S, Lofdahl S (1988) Cloning of a chromosomal locus (exp) which regulates the expression of several exoprotein genes in Staphylococcus aureus. Mol Gen Genet 211:435–440
Novick RP (1963) Properties of a cryptic high-frequency transducing phase in Staphylococcus aureus. Virology 33:155–166
Oskouian B, Stewart GC (1990) Repression and catabolite repression of the lactose operon of Staphylococcus aureus. J Bacteriol 172:3804–3812
Peng HI, Novick RP, Kreiswirth B, Kornblum J, Schlievert P (1988) Cloning, characterisation and sequencing of an accessory gene regulator (agr) in Staphylococcus aureus. J Bacteriol 170:4365–4372
Ray C, Hay RE, Carter HL, Moran CP (1985) Mutations that affect utilisation of a promoter in stationary-phase Bacillus subtilis. J Bacteriol 163:610–614
Rescei P, Kreiswirth B, O'Reilly M, Schlievert P, Gruss A, Novick RP (1986) Regulation of exoprotein gene expression in Staphylococcus aureus by agr. Mol Gen Genet 202:58–61
Ronson CW, Nixon BT, Ausubel FM (1987) Conserved domains in bacterial regulatory proteins that respond to environmental stimuli. Cell 49:579–581
Sakuri S, Suzuki H, Kondo I (1988) DNA sequencing of the eta gene coding for staphylococcal exfoliative toxin serotype A. J Gen Microbiol 134:711–717
Stock JB, Ninfa AJ, Stock AM (1989) Protein phosphorylation and regulation of adaptive responses in bacteria. Microbiol Rev 53:450–490
Stock JB, Stock AM, Mottonen J (1990) Signal transduction in bacteria. Nature 344:395–400
Sutcliffe JA, Gootz TD, Barrett JF (1989) Biochemical characteristics and physiological significance of major DNA topoisomerases. Antimicrob Agents Chemother 33:2027–2033
O'Toole P, Foster TJ (1986) Molecular cloning and expression of the epidermolytic toxin A gene of Staphylococcus aureus. Microbial Pathogenesis 1:583–594
O'Toole P, Foster TJ (1987) Nucleotide sequence of the epidermolytic toxin A gene of Staphylococcus aureus. J Bacteriol 169:3910–3915
Wilson C, Skinner S, Shaw W (1981) Analysis of two chloramphenicol resistance plasmids from Staphylococcus aureus: Insertional inactivation of chloramphenicol resistance, mapping of restriction sites, and construction of cloning vehicles. Plasmid 5:245–258
Yanisch-Perron C, Vieira J, Messing J (1983) Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC vectors. Gene 33:103–119
Zukowski MM, Gaffney DF, Speck D, Kauffmann M, Findeli A, Wisecup A, Lecocq J (1983) Chromogenic identification of genetic regulatory signals in Bacillus subtilis based on expression of a cloned Pseudomonas gene. Proc Natl Acad Sci USA 80:1101–1105
Author information
Authors and Affiliations
Additional information
Communicated by W. Goebel
Rights and permissions
About this article
Cite this article
Sheehan, B.J., Foster, T.J., Dorman, C.J. et al. Osmotic and growth-phase dependent regulation of the eta gene of Staphylococcus aureus: a role for DNA supercoiling. Molec. Gen. Genet. 232, 49–57 (1992). https://doi.org/10.1007/BF00299136
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00299136