Summary
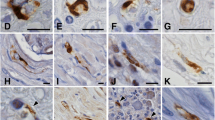
Sural nerve specimens from ten patients with amyloidosis (hereditary, associated with lymphoproliferative disorders, or of unknown origin) and peripheral neuropathy were investigated by immunohistochemistry at the light and electron microscopic level. Peroxidase-antiperoxidase and immunogold techniques were applied to glutaraldehyde-fixed, osmicated and epoxy-embedded tissue. In five cases, four of which associated with lymphoproliferative disorders, amyloid deposits strongly and exclusively reacted with antibodies to kappa or lambda light chains, respectively. By electron microscopy, bundles of immunogold-labelled amyloid fibrils could be identified in coated and uncoated single membrane-bound vesicles of endoneurial macrophages. Schwann cells did not contain intracellular amyloid but their processes were entangled in amyloid fibrils and their basement membranes were sometimes fused with the fibrillar masses. It is concluded that immunoglobulin light chains in AL (amyloid of immunoglobulin light chain origin) amyloidosis precipitate, forming amyloid fibrils, in the presence of, and presumably with the assistence of, endoneurial cells. Inefficiency of phagocytosis appears to be one of the major causes for the deleterious effects of amyloid.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bendayan M, Zollinger M (1983) Ultrastructural localization of antigenic sites on osmium-fixed tissues applying the protein A-gold technique. J Histochem Cytochem 31:101–109
Beyreuther K, Multhaupt G, Simms G, Pottgiesser J, Schröder W, Martins RH, Masters CC (1986) Neurofibrillary tangles of Alzheimer's disease and “aged” Down's syndrome contain the same protein as the amyloid of plaque cores and blood vessels. Disc Neurosci 3:68–80
Cohen AS, Rubinow A (1984) Amyloid neuropathy. In: Dyck PJ, Thomas PK, Lambert EN, Bunge R (eds) Peripheral neuropathy. Saunders, Philadelphia London, pp 1866–1898
Coimbra S, Andrade C (1971) Familial amyloid polyneuropathy: an electron microscope study of the peripheral nerve in five cases. Brain 94:199–221
Donini U, Casanova S, DalBosco F, Linke RP (1984) Immunohistochemical typing of amyloid on hydroxyethyl-methacrylate-embedded renal biopsies. Appl Pathol 2:299–307
Durie BGM, Persky B, Soehnlen BJ, Grogan TM, Salmon SE (1982) Amyloid production in human myeloma stemcell culture, with morphological evidence of amyloid secretion by associated macrophages. N Engl J Med 307:1689–1692
Dyck PJ, Lambert EH (1969) Dissociated sensation in amyloidosis. Compound action potential, quantitative histology and teased-fibers, and electron microscopic studies of sural nerve biopsies. Arch Neurol 20:490–507
Franklin EC, Zucker-Franklin D (1972) Current concepts of amyloid. Adv Immunol 15:249–304
Fuks A, Zucker-Franklin D (1985) Impaired Kupffer cell function precedes development of secondary amyloidosis. J Exp Med 161:1013–1028
Ghadially FN (1982) Ultrastructural pathology of the cell and matrix. Butterworths, London, p 971
Glenner GG (1980) Amyloid deposits and amyloidosis. N Engl J Med 302:1283–1292
Gueft B, Ghidoni JJ (1963) The site of formation and ultrastructure of amyloid. Am J Pathol 43:837–854
Hanyu N, Ikeda S, Nakadai A, Yanagisawa N, Powell H (1989) Peripheral nerve pathological findings in familial amyloid polyneuropathy: a correlative study of proximal sciatic nerve and sural nerve lesions. Ann Neurol 25:340–350
Hikita K, Tateishi J, Nagara H (1985) Morphogenesis of amyloid plaques in mice with Creutzfeldt-Jakob disease. Acta Neuropathol (Berl) 68:138–144
Janssen S, Elema JD, van Rijswijk MH, Limburg PC, Meijer S, Mandema E (1985) Classification of amyloidosis: immunohistochemistry versus the potassium permanganate method in differentiating AA from AL amyloidosis. Appl Pathol 31:29–38
Janssen S, Martin H, van Rijswijk MH, Meijer S, Ruinen L, van der Hem K (1986) Clinical evaluation of AA and AL amyloid disease. In: Marrink J, VanRijswijk M (eds) Amyloidosis. Martinus Nijhoff, Dordrecht, pp 61–72
Kirkpatrick CJ, Curry A, Galle J, Melzner I (1986) Systemic kappa light chain deposition and amyloidosis in multiple myeloma: novel morphological observations. Histopathology 10:1065–1076
Kitamoto T, Tateishi J, Hikita K, Nagara H, Takeshita I (1985) A new method to classify amyloid fibril proteins. Acta Neuropathol (Berl) 67:272–278
Kjeldsberg CR, Eyre HJ, Totzke H (1977) Evidence for intracellular amyloid formation in myeloma. Blood 50:493–504
Koeppen AM, Mitzen EJ, Hans MB, Peng SK (1985) Familial amyloid polyneuropathy. Muscle Nerve 8:733–749
Krücke W (1963) Zur pathologischen Anatomie der Paramyloidose. Acta Neuropathol (Berl) [Suppl] II:74–93
Linke RP (1982) Immunohistochemical identification and cross reaction of amyloid fibril proteins in senile heart and amyloid in familial polyneuropathy. Clin Neuropathol 1:172–182
Marrink J, van Rijswijk M (1986) Amyloidosis. Martinus Nijhoff, Dordrecht, p 378
Nakagawa S (1984) Ultrastructural investigation of amyloidosis. Pathogenesis of systemic amyloidosis. Appl Pathol 2:328–340
Neundörfer B, Meyer JG, Volk B (1977) Amyloid neuropathy due to monoclonal gammopathy. J Neurol 216:207–215
Numura S, Kano T, Uchino H (1982) Intracellular formation of amyloid fibrils in myeloma: cytochemical, immunochemical and electron microscopic observations. Acta Haematol Jpn 45:615–621
Pajor L, Kelenyi G (1984) Primary cardiovascular amyloidosis with benign monoclonal gammopathy. Virchows Arch [A] 403:195–203
Peters A, Palay SL, Webster H deF (1976) The fine structure of the nervous system. Saunders, Philadelphia, pp 33–34
Said G, Ropert A, Faux N (1984) Length-dependent degeneration of fibrils in Portuguese amyloid neuropathy. Neurology 34:1025–1032
Shirahama AT, Cohen AS (1970) The association of hemidesmosome-like plaques and dense coating with the pinocytotic uptake of a heterologous fibrillar protein (amyloid)by macrophages. J Ultrastruct Res 33:587–597
Shirahama T, Cohen AS (1971) Lysosomal breakdown of amyloid fibrils by macrophages. Am J Pathol 63:463–485
Shirahama T, Cohen AS (1973) An analysis of the close relationship of lysosomes to early deposits of amyloid. Am J Pathol 73:97–114
Shiramaha T, Cohen AS (1975) Intralysosomal formation of amyloid fibrils. Am J Pathol 81:101–116
Shirahama T, Cohen AS (1985) Immunocytochemical study of hepatocyte syntheses of amyloid AA. Am J Pathol 118:108–115
Shirahama T, Skinner M, Sipe JD, Cohen AS (1985) Widespread occurrence of AP in amyloidotic tissue. Virchows Arch [B] 48:197–206
Sternberger LA (1986) Immunocytochemistry, 3rd edn. Wiley & Sons, New York
Takahashi K, Naito M, Takatsuki K, Kono F, Chitose M, Ooshima S, Mori N, Sakuma H, Uchino F (1987) Multiple myeloma, IgA kappa type, accompanying crystal-storing histiocytosis and amyloidosis. Acta Pathol Jpn 37:141–154
Thomas PK, King RHM (1974) Peripheral nerve changes in amyloid neuropathy. Brain 97:395–406
Uchino F, Takahashi M, Yokota T, Ishihara T (1985) Experimental amyloidosis. The role of Kupffer cells in amyloid formation. Appl Pathol 3:78–87
Van Rijswijk MH, van Heusden CWGJ (1979) The potassium permanganate method. Am J Pathol 97:43–58
Viale G, Dell'Orto P, Colombi R, Coggi G (1986) Ultrastructural localization of extracellular immunoglobulins in immune-complex-mediated glomerulonephritis. Histochemistry 84:1–4
Vital C, Lacoste D, Deminiere C, Lagueny A, Boisseau C, Reiffer J, Amouretti M, Broustet A (1983) Amyloid neuropathy and multiple myeloma. Eur Neurol 22:106–112
Vital C, Vallat JM (1988) Ultrastructural study of the human diseased peripheral nerve, 2nd edn. Elsevier, New York, pp 113–116, 149–166
Westermark P (1986) Amyloid of the islets ofLangerhans and its connection with diabetes mellitus. In: Marrink J, Van Rijswijk N (eds) Amyloidosis. Martinus Nijhoff, Dordrecht, pp 255–259
Zucker-Franklin D (1970) Immunophagocytosis of human amyloid fibrils by leucocytes. J Ultrastruct Res 32:247–257
Author information
Authors and Affiliations
Additional information
Supported in part by a grant from the Deutsche Forschungsgemeinschaft (FE 255/1/1) (CS). Presented in Part at the Fourth International Meeting of the Periveral Nerve Association of America, Halifax, July 19–23, 1988
Rights and permissions
About this article
Cite this article
Sommer, C., Schröder, J.M. Amyloid neuropathy: immunocytochemical localization of intra- and extracellular immunoglobulin light chains. Acta Neuropathol 79, 190–199 (1989). https://doi.org/10.1007/BF00294378
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00294378