Summary
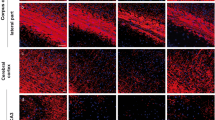
An immunohistochemical study was performed to compare glial reactions in recent and old lesions of central pontine myelinolysis (CPM). Regions of demyelination and destruction of oligodendrocytes, showed reduced immunoreactivity of myelin basic protein (MBP), myelin-associated glycoprotein (MAG), transferrin, and carbonic anhydrase C (CA C). In addition, labeling of glial fibrillary acidic protein (GFAP) and S-100 protein revealed distinct dystrophic alterations of the astroglia. Remarkably, immunolabeling of GFAP was drastically reduced in astrocytic cytoplasm within freshly demyelinated lesions. Immunostaining of vimentin revealed a differential intracytoplasmic decoration of hypertrophic and dystrophic astrocytes in recent and old CPM lesions. Immunolabeling of desmin failed to stain glial cells. Monoclonal antibodies against HNK-1 exhibited greatly increased immunoreactivity both of persisting oligodendrocytes and of reactive fibrillary astrocytes in old CPM foci. In freshly demyelinated lesions, enhanced immunoreactivity of the X-hapten (3-fucosyl-N-acetyllactosamine) was prominent in astroglia and oligodendrocytes. Simultaneously, reactive astrocytes revealed intracytoplasmic labeling of laminin. Quantitation of GFAP+ astroglia in fresh CPM and control cases revealed an increase in the number of astrocytes within the demyelinated foci and in the surrounding nondemyelinated pontine tissue of CPM cases. The occurrence of astroglial alterations in the demyelinated foci of CPM could be interpreted as “astroglial dystrophy” which may represent a pathogenic factor in CPM. Furthermore, it is possible that changes of the glial microenvironment may influence the astroglia to revert transiently back to an immature phenotype as indicated by the enhanced expression of the X-hapten and HNK-1, and the de novo synthesis of vimentin and laminin.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abo T, Balch CM (1981) A differentiation antigen of human NK and K cells identified by a monoclonal antibody (HNK-1). J Immunol 127:1024–1029
Adams RD, Victor M, Mancall EL (1959) Central pontine myelinolysis. A hitherto undescribed disease occurring in alcoholic and malnourished patients. Arch Neurol Psychiatry 81:154–172
Bartsch U, Kirchhoff F, Schachner M (1989) Immuno-histological localization of the adhesion molecules L1, N-CAM, and MAG in the developing and adult optic nerve of mice. J Comp Neurol 284:451–462
Bloch B, Popovici T, Levin MJ, Tuil D, Kahn A (1985) Transferrin gene expression visualized in oligodendrocytes of the rat brain by using in situ hybridization and immunohistochemistry. Proc Natl Acad Sci USA 82:6706–6710
Dahl D, Bignami A (1982) Immunohistological localization of desmin, the muscle-type 100 Å filament protein, in rat astrocytes and Müller glia. J Histochem Cytochem 30:207–213
Dahl D, Bignami A, Weber K, Osborn M (1981) Filament proteins in rat optic nerves undergoing Wallerian degeneration. Localization of vimentin, the fibroblastic 100-Å filament protein, in normal and reactive astrocytes. Exp Neurol 73:496–506
Dahl D, Rueger DC, Bignami A, Weber K, Osborn M (1981) Vimentin, the 57000 molecular weight protein of fibroblast filaments, is the major cytoskeletal component in immature glia. Eur J Cell Biol 24:191–196
DeArmond SJ, Eng LF, Rubinstein LJ (1980) The application of glial fibrillary acidic (GFA) protein immunohistochemistry in neurooncology. A progress report. Pathol Res Pract 168:374–394
Deck JHN, Rubinstein LJ (1981) Glial fibrillary acidic protein in stromal cells of some capillary hemangioblastomas. Significance and possible implications of an immunoperoxidase study. Acta Neuropathol (Berl) 54:173–181
Eng LF, DeArmond SJ (1982) Immunocytochemical studies of astrocytes in normal development and disease. In: Fedoroff S, Hertz L (eds) Advances in cellular neurobiology, vol 3. Academic Press, London, pp 145–171
Eng LF, DeArmond SJ (1983) Immunocytochemistry of the glial fibrillary acidic protein. In: Zimmermann HM (ed) Progress in neuropathology, vol 5. Raven Press, New York, pp 19–39
Fedoroff S, White RV, Neal J, Subrahmanyan L, Kalnins VI (1983) Astrocyte cell lineage. II. Mouse fibrous astrocytes and reactive astrocytes in cultures have vimentin- and GFP-containing intermediate filaments. Dev Brain Res 7:303–315
Gallyas F, Merchenthaler I (1988) Copper-H2O2 oxidation strikingly improves silver intensification of the nickeldiaminobenzidine (Ni-DAB) end-product of the peroxidase reaction. J Histochem Cytochem 36:807–810
Giftochristos N, David S (1988) Laminin and heparan sulphate proteoglycan in the lesioned adult mammalian central nervous system and their possible relationship to axonal sprouting. J Neurocytol 17:385–397
Gocht A, Colmant HJ (1987) Central pontine and extrapontine myelinolysis. A report of 58 cases. Clin Neuropathol 6:262–270
Goebel HH, Herman-Ben Zur P (1976) Central pontine myelinolysis. Handb Clin Neurol 28:285–316
Graeber MB, Streit WJ, Kreutzberg GW (1989) Formation of microglia-derived brain macrophages is blocked by adriamycin. Acta Neuropathol 78:348–358
Graham RC, Karnovsky MJ (1966) The early stages of absorption of injected horseradish peroxidase in the proximal tubules of mouse kidney. Ultrastructural cytochemistry by a new technique. J Histochem Cytochem 14:291–301
Hanjan SNS, Kearney JF, Cooper MD (1982) A monoclonal antibody (MMA) that identifies a differentiation antigen on human myelomonocytic cells. Clin Immunol Immunopathol 23:172–188
Hsu SM, Raine L, Fanger H (1981) Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques. A comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 9:577–580
Illowsky B, Laureno R (1987) Encephalopathy and myelinolysis after rapid correction of hyponatraemia. Brain 110:855–867
Itoyama Y, Sternberger NH, Kies MW, Cohen SR, Richardson EP, Webster HdeF (1980) Immunocytochemical method to identify myelin basic protein in oligodendroglia and myelin sheaths of the human nervous system. Ann Neurol 7:157–166
Jellinger K (1968) Zur Neuropathologie des Komas und postkomatöser Encephalopathien. Wien klin Wochenschr 80:505–517
Kleinman HK, Ogle RC, Cannon FB, Little CD, Sweeney TM, Luckenbill-Edds L (1988) Laminin receptors for neurite formation. Proc Natl Acad Sci USA 85:1281–1286
Kleinschmidt-DeMasters BK, Norenberg MD (1981) Rapid correction of hyponatremia causes demyelination. Relation to central pontine myelinolysis. Science 211:1068–1070
Kumpulainen T, Korhonen LK (1982) Immunohistochemical localization of carbonic anhydrase isoenzyme C in the central and peripheral nervous system of the mouse. J Histochem Cytochem 30:283–292
Kumpulainen T, Nyström SHM (1981) Immunohistochemical localization of carbonic anhydrase isoenzyme C in human brain. Brain Res 220:220–225
Liesi P (1985) Do neurons in the vertebrate CNS migrate on laminin? EMBO J 4:1163–1170
Liesi P, Kaakkola S, Dahl D, Vaheri A (1984) Laminin is induced by astrocytes of adult brain by injury. EMBO J 3:683–686
Ludwin SK, Kosek JC, Eng LF (1976) The topographical distribution of S-100 and GFA proteins in the adult rat brain. An immunohistochemical study using horseradish peroxidase-labelled antibodies. J Comp Neurol 165:197–208
Mai JK, Reifenberger G (1988) Distribution of the carbohydrate epitope 3-fucosyl-N-acetyllactosamine (FAL) in the adult human brain. J Chem Neuroanat 1:255–285
Manthorpe M, Engvall E, Ruoslahti E, Longo FM, Davis GE, Varon S (1983) Laminin promotes neuritic regeneration from cultured peripheral and central neurons. J Cell Biol 97:1882–1890
Mason DY, Woolston R-E (1982) Double immunoenzymatic labelling. In: Bullock GR, Petrusz P (eds) Techniques in immunocytochemistry, vol 1. Academic Press, London, pp 135–153
Niedieck B, Löhler J (1987) Expression of 3-fucosyl-N-acetyllactosamine on glia cells and its putative role in cell adhesion. Acta Neuropathol (Berl) 75:173–184
Norenberg MD (1981) Ultrastructural observations in electrolyte-induced myelinolysis (abstract). J Neuropathol Exp Neurol 40:319
Norenberg MD (1983) A hypothesis of osmotic endothelial injury. A pathogenetic mechanism in central pontine myelinolysis. Arch Neurol 40:66–69
Norenberg MD, Papendick RE (1984) Chronicity of hyponatremia as a factor in experimental myelinolysis. Ann Neurol 15:544–547
Norenberg MD, Leslie KO, Robertson AS (1982) Association between rise in serum sodium and central pontine myelinolysis. Ann Neurol 11:128–135
Okeda R, Kitano M, Sawabe M, Yamada I, Yamada M (1986) Distribution of demyelinating lesions in pontine and extrapontine myelinolysis. Three autopsy cases including one case devoid of central pontine myelinolysis. Acta Neuropathol (Berl) 69:259–266
Perentes E, Rubinstein LJ (1985) Immunohistochemical recognition of human nerve sheath tumors by anti-Leu 7 (HNK-1) monoclonal antibody. Acta Neuropathol (Berl) 68:319–324
Pogacar S (1980) Iatrogenic precipitation of central pontine myelinolysis with formation of Rosenthal fibers (abstract). J Neuropathol Exp Neurol 39:383
Powers JM, McKeever PE (1976) Central pontine myelinolysis. An ultrastructural and elemental study. J Neurol Sci 29:65–81
Reifenberger G, Bilzer T, Seitz RJ, Wechsler W (1989) Expression of vimentin and glial fibrillary acidic protein in ethylnitrosourea-induced rat gliomas and glioma cell lines. Acta Neuropathol 78:270–282
Rutishauser U, Jessell TM (1988) Cell adhesion molecules in vertebrate neural development. Physiol Rev 68:819–857
Schwarting GA, Yamamoto M (1988) Expression of glycoconjugates during development of the vertebrate nervous system. Bio Essay 9:19–23
Seitelberger F (1973) Zentrale pontine Myelinolyse. Schweiz Arch Neurol Neurochir Psychiatr 112:285–297
Solter D, Knowles BB (1978) Monoclonal antibody defining a stage-specific mouse embryonic antigen (SSEA-1). Proc Natl Acad Sci USA 75:5565–5569
Sosale A, Robson JA, Stelzner DJ (1988) Laminin distribution during corticospinal tract development and after spinal cord injury. Exp Neurol 102:14–22
Stagaard M, Møllgård K (1989) The developing neuro-epithelium in human embryonic and fetal brain studied with vimentin-immunocytochemistry. Anat Embryol 180:17–28
Sternberger LA (1986) Immunocytochemistry, 3rd edn. Wiley, New York
Sternberger NH, Quarles RH, Itoyama Y, Webster HdeF (1979) Myelin-associated glycoprotein demonstrated immunocytochemically in myelin and myelin-forming cells of developing rat. Proc Natl Acad Sci USA 76:1510–1514
Timpl R, Rhode H, Robey PG, Rennard SI, Foidart J-M, Martin GR (1979) Laminin. A glycoprotein from basement membranes. J Biol Chem 254:9933–9937
Van der Meulen JDM, Houthoff HJ, Ebels EJ (1978) Glial fibrillary acidic protein in human gliomas. Neuropathol Appl Neurobiol 4:177–190
Wasterlain CG, Torack RM (1968) Cerebral edema in water intoxication. II. An ultrastructural study. Arch Neurol 19:79–87
Weibel ER (1979) Stereological methods. Practical methods for biological morphometry, vol 1. Academic Press, London
Wildi E (1973) Contribution à l'anatomo-clinique de la myélinolyse du pont. Schweiz Arch Neurol Neurochir Psychiatr 112:271–284
Wright DG, Laureno R, Victor M (1979) Pontine and extrapontine myelinolysis. Brain 102:361–385
Author information
Authors and Affiliations
Additional information
The Heinrich-Pette-Institut is financially supported by Freie und Hansestadt Hamburg and Bundesministerium für Jugend, Familie, Frauen und Gesundheit
Rights and permissions
About this article
Cite this article
Gocht, A., Löhler, J. Changes in glial cell markers in recent and old demyelinated lesions in central pontine myelinolysis. Acta Neuropathol 80, 46–58 (1990). https://doi.org/10.1007/BF00294221
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00294221