Abstract
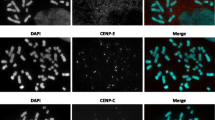
We have screened for the presence of two centromere autoantigens, CENP-B (80 kDa) and CENP-C (140 kDa) at the inactive centromere of a naturally occurring stable dicentric chromosome using specific antibodies that do not cross-react with any other chromosomal proteins. In order to discriminate between the active and inactive centromeres on this chromosome we have developed a modification of the standard methanol/acetic acid fixation procedure that allows us to obtain high-quality cytological spreads that retain antigenicity with the anti-centromere antibodies. We have noted three differences in the immunostaining patterns with specific anti-CENP-B and CENP-C antibodies. (1) The amount of detectable CENP-B varies from chromosome to chromosome. The amount of CENPC appears to be more or less the same on all chromosomes. (2) CENP-B is present at both active and inactive centromeres of stable dicentric autosomes. CENP-C is not detectable at the inactive centromeres. (3) While immunofluorescence with anti-CENP-C antibodies typically gives two discrete spots, staining with anti-CENP-B often appears as a single bright bar connecting both sister centromeres. This suggests that while CENP-C may be confined to the outer centromere in the kinetochore region, CENP-B may be distributed throughout the entire centromere. Our data suggest that CENP-C is likely to be a component of some invariant chromosomal substructure, such as the kinetochore. CENPB may be involved in some other aspect of centromere function, such as chromosome movement or DNA packaging.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CENP:
-
centromere protein
References
Balczon RD, Brinkley BR (1987) Tubulin interaction with kinetochore proteins: Analysis by in vitro assembly and chemical cross-linking. J Cell Biol 105:855–862
Bravo R, Macdonald-Bravo H (1987) Existence of two populations of cyclin/proliferating cell nuclear antigen during the cell cycle: association with DNA replication sites. J Cell Biol 105:1549–1554
Brenner S, Pepper D, Berns MW, Tan E, Brinkley BR (1981) Kinetochore structure, duplication and distribution in mammalian cells: analysis by human autoantibodies from scleroderma patients. J Cell Biol 91:95–102
Cherry LM, Wang R-Y (1988) Kinetochore staining in multicentric chromosomes of murine cancer cell lines. Cytobios 53:173–183
Cole A (1967) Chromosome structure. Theor Biophys 1:305–375
Distèche C, Hagemeijer A, Frederic J, Progneaux D (1972) An abnormal large human chromosome identified as an end-to-end fusion of two X's by combined results of the new banding techniques and microdensitometry. Clin Genet 3:338–395
Earnshaw WC, Laemmli UK (1983) Architecture of metaphase chromosomes and chromosome scaffolds. J Cell Biol 96:84–93
Earnshaw WC, Migeon BR (1985) Three related centromere proteins are absent from the inactive centromere of a stable isodicentric chromosome. Chromosoma 92:290–296
Earnshaw WC, Rothfield NF (1985) Identification of a family of human centromere proteins using autoimmune sera from patients with scleroderma. Chromosoma 91:313–321
Earnshaw WC, Halligan N, Cooke CA, Rothfield NF (1984) The kinetochore is part of the metaphase chromosome scaffold. J Cell Biol 98:352–357
Earnshaw WC, Bordwell BJ, Marino C, Rothfield NF (1986) Three human chromosomal autoantigens are recognized by sera from patients with anti-centromere antibodies. J Clin Invest 77:426–430
Earnshaw WC, Sullivan KF, Machlin PS, Cooke CA, Kaiser DA, Pollard TD, Rothfield NF, Cleveland DW (1987) Molecular cloning of cDNA for CENP-B, the major human centromere autoantigen. J Cell Biol 104:817–829
Gorbsky GJ, Sammak PJ, Borisy GG (1987) Chromosomes move poleward in anaphase along stationary microtubules that coordinately disassemble from their kinetochore ends. J Cell Biol 104:9–18
Hsu TC, Pathak S, Chen TR (1975) The possibility of latent centromeres and a proposed nomenclature system for total chromosome and whole arm translocations. Cytogenet Cell Genet 15:41–49
Jabs EW, Wolf SF, Migeon BR (1984) Characterization of a cloned DNA sequence that is present at centromeres of all human autosomes and the X chromosome and shows polymorphic variation. Proc Natl Acad Sci USA 81:4884–4888
Kirschner MW, Mitchison TJ (1986) Beyond self-assembly: from microtubules to morphogenesis. Cell 45:329–342
Koshland D, Rutledge L, Fitzgerald-Hayes M, Hartwell EH (1987) A genetic analysis of dicentric minichromosomes in Saccharomyces cerevisiae. Cell 48:801–812
Koshland DE, Mitchison TJ, Kirschner MW (1987) Polewards chromosome movement driven by microtubule depolymerization in vitro. Nature 331:499–504
Labhart P, Koller T (1981) Electron microscope preparation of rat liver chromatin by a modified Miller procedure. Eur J Cell Biol 24:309–316
Mann R, Davis R (1983) Instability of dicentric plasmids in yeast. Proc Natl Acad Sci USA 80:228–232
McClintock B (1939) The behavior in successive nuclear divisions of a chromosome broken in meiosis. Proc Natl Acad Sci USA 25:405–416
McKeon FD, Tuffanelli DL, Kobayashi S, Kirschner MW (1984) The redistribution of a conserved nuclear envelope protein during the cell cycle suggests a pathway for chromosome condensation. Cell 36:83–92
Merry DW, Pathak S, Hsu TC, Brinkley BR (1985) Anti-kinetochore antibodies: use as probes for inactive centromeres. Am J Hum Genet 37:425–430
Moroi Y, Peebles C, Fritzler MJ, Steigerwald J, Tan EM (1980) Autoantibody to centromere (kinetochore) in scleroderma sera. Proc Natl Acad Sci USA 77:1627–1631
Oertal W, Mayer M (1984) Structure and mitotic stability of minichromosomes originating in yeast cells transformed with tandem dimers of CEN11 plasmids. Mol Gen Genet 198:300–306
Peretti D, Maraschio P, Lambiase S, Lo Curto F, Zuffardi O (1986) Indirect immunofluorescence of inactive centromeres as indicator of centromeric function. Hum Genet 73:12–16
Rieder CL (1982) The formation, structure and composition of the mammalian kinetochore and kinetochore fiber. Int Rev Cytol 79:1–58
Rijhsinghani AG, Hruban RH, Stetten G (1988) Fetal anomalies associated with an inversion duplication 13 chromosome. Obstet Gynecol 71:991–994
Snyder M, Elledge S, Sweetser D, Young RA, Davis RW (1988) γgt11: Gene isolation with antibody probes and other applications. Methods Enzymol 154:107–128
Thoma F, Koller T, Klug A (1979) Involvement of histone H1 in the organization of the nucleosome and of the salt-dependent superstructures of chromatin. J Cell Biol 83:403–427
Vig BK, Paweletz N (1988) Sequence of centromere separation: generation of unstable multicentric chromosomes in a rat cell line. Chromosoma 96:275–282
Warburton D, Henderson AS, Shapiro LR, Hsu LYF (1973) A stable human dicentric chromosome, tdic(12;14)(p13;p13), including an intercalary satellite region between centromeres. Am J Hum Genet 25:439–445
Wataya-Kaneda M, Kaneda Y, Sakurai T, Sugawa H, Uchida T (1987) A monoclonal antibody against the nucleus reveals the presence of a common protein in the nuclear envelope, the perichromosomal region, and cytoplasmic vesicles. J Cell Biol 104:1–7
Williamson DH, Fennell DJ (1975) The use of fluorescent DNA-binding agent for detecting and separating yeast mitochondrial DNA. Methods Cell Biol 12:335–351
Young RA, Davis RB (1983) Efficient isolation of genes by using antibody probes. Proc Natl Acad Sci USA 80:1194–1198
Young RA, Davis RB (1983) Yeast polymerase II genes: isolation with antibody probes. Science 222:778–782
Zinkowski RP, Vig BK, Broccoli D (1986) Characterization of kinetochores in multicentric chromosomes. Chromosoma 94:243–248
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Earnshaw, W.C., Ratrie, H. & Stetten, G. Visualization of centromere proteins CENP-B and CENP-C on a stable dicentric chromosome in cytological spreads. Chromosoma 98, 1–12 (1989). https://doi.org/10.1007/BF00293329
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00293329