Summary
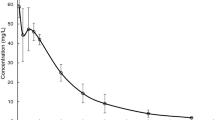
Six normal, healthy adult males received a single dose of allopurinol intravenously, orally in the form of a commercial tablet, and rectally in the form of an extemperaneously prepared suppository (either in a cocoa butter or in polyethylene glycol base). Plasma allopurinol and oxipurinol concentrations were measured over a period of at least 60 h. The following mean (±SD) values were obtained from the intravenous allopurinol experiment: clearance, 9.62±3.49 ml · kg-1 · min-1; Vd, 1.61±0.74 l/kg; t1/2, 1.62 h. Oxipurinol had a mean t1/2 of 16.90 h. The absolute systemic bioavailability of the oral tablet was 67%±23%, while the allopurinol rectal suppositories produced no measurable plasma concentrations of allopurinol or oxipurinol in any of the subjects. Current use of rectal dosage forms as an adjunct in cancer chemotherapy should therefore be re-examined.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Appelbaum SJ, Mayersohn M, Perrier D, Dorr RT (1980) Allopurinol absorption from rectal suppositories. Drug Intell Clin Pharmacy 14:789
Benezra SA, Bennett TR (1978) Allopurinol. In: Florey K (ed) Analytical profiles of drug substances, vol 7. Academic Press, New York, pp 1–17
Boxenbaum HG, Riegelman RM, Elashoff M (1974) Statistical estimations in pharmacokinetics. J Pharmacokin Biopharm 2:123
Chang S-L, Kramer WG, Feldman S, Ballentine R, Frankel LS (1981) Bioavailability of allopurinol oral and rectal dosage forms. Am J Hosp Pharm 38:365
DeConti RC, Calabresi P (1966) Use of allopurinol for prevention and control of hyperuricemia in patients with neoplastic disease. N Engl J Med 274:481
Elion GB, Kovensky A, Hitchings GH, Metz E, Rundles RW (1966) Metabolic studies of allopurinol, an inhibitor of xanthine oxidase. Biochem Pharmacol 15:863
Elion GB, Yu T-F, Gutman AB, Hitchings GH (1968) Renal clearance of oxipurinol, the chief metabolite of allopurinol. Am J Med 45:69
Gibaldi M, Perrier D (1975) Pharmacokinetics. Marcel Dekker, New York, pp 73–75
Hande K, Reed E, Chabner B (1978) Allopurinol kinetics. Clin Pharmacol Ther 23:598
Kjellstrand CM, Campbell DC, von Hartitzsch B, Buselmeier TJ (1974) Hyperuricemic acute renal failure. Arch Intern Med 133:349
Klinenberg JR, Goldfinger SE, Seegmiller JE (1965) The effectiveness of the xanthine oxidase inhibitor allopurinol in the treatment of gout. Ann Intern Med 62:639
Krakoff CW, Meyer RL (1965) Prevention of hyperuricemia in leukemia and lymphoma. JAMA 193:89
Metzler CM (1969) A Users Manual for NONLIN. Upjohn Co., Kalamazoo
Rundles RW, Wyngaarden JB, Hitchings GH, Elion GB, Silberman HR (1963) Effects of xanthine oxidase inhibitor on thiopurine metabolism, hyperuricemia and gout. Trans Assoc Am Physicians 76:126
Vogler WR, Bain JA, Huguley CM, Palmer HG, Lowery ME (1966) Metabolic and therapeutic effects of allopurinol in patients with leukemia and gout. Am J Med 40:548
Yu TF, Gutman AB (1964) Effects of allopurinol (4-hydroxypyrazolo-(3,4-d) pyrimidine) on serum and urinary uric acid in primary and secondary gout. Am J Med 37:885
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Appelbaum, S.J., Mayersohn, M., Dorr, R.T. et al. Allopurinol kinetics and bioavailability. Cancer Chemother. Pharmacol. 8, 93–98 (1982). https://doi.org/10.1007/BF00292878
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00292878