Abstract
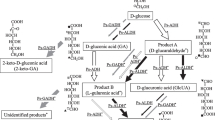
The hyperthermophilic archaeon Pyrococcus furiosus was grown on pyruvate as carbon and energy source. The enzymes involved in gluconeogenesis were investigated. The following findings indicate that glucose-6-phosphate formation from pyruvate involves phosphoenolpyruvate synthetase, enzymes of the Embden-Meyerhof pathway and fructose-1,6-bisphosphate phosphatase.
Cell extracts of pyruvate-grown P.furiosus contained the following enzyme activities: phosphoenolpyruvate synthetase (0.025 U/mg, 50 °C), enolase (0.9 U/mg, 80 °C), phosphoglycerate mutase (0.13 U/mg, 55 °C), phosphoglycerate kinase (0.01 U/mg, 50 °C), glyceraldehyde-3-phosphate dehydrogenase reducing either NADP+ or NAD+ (NADP+: 0.019 U/mg, NAD+: 0.009 U/mg; 50 °C), triosephosphate isomerase (1.4 U/mg, 50 °C), fructose-1,6-bisphosphate aldolase (0.0045 U/mg, 55 °C), fructose-1,6-bisphosphate phosphatase (0.026 U/mg, 75 °C), and glucose-6-phosphate isomerase (0.22 U/mg, 50 °C). Kinetic properties (V max values and apparent K m values) of the enzymes indicate that they operate in the direction of sugar synthesis. The specific enzyme activities of phosphoglycerate kinase, glyceraldehyde-3-phosphate dehydrogenase (NADP+-reducing) and fructose-1,6-bisphosphate phosphatase in pyruvate-grown P. furiosus were by a factor of 3, 10 and 4, respectively, higher as compared to maltose-grown cells suggesting that these enzymes are induced under conditions of gluconeogenesis. Furthermore, cell extracts contained ferredoxin: NADP+ oxidoreductase (0.023 U/mg, 60 °C); phosphoenolpyruvate carboxylase (0.018 U/mg, 50 °C) acts as an anaplerotic enzyme.
Thus, in P. furiosus sugar formation from pyruvate involves reactions of the Embden-Meyerhof pathway, whereas sugar degradation to pyruvate proceeds via a modified “non-phosphorylated” Entner-Doudoroff pathway.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abdelal T, Schlegel HG (1974) Purification and regulatory properties of fructose 1,6-diphosphatase from Hydrogenomonas eutropha. J Bacterial 120: 304–310
Altekar W, Rangaswamy V (1991) Ketohexokinase (ATP: D-fructose 1-phosphotransferase) initiates fructose breakdown via the modified EMP pathway in halophilic archaebacteria. FEMS Microbiol Lett 83: 241–246
Altekar W, Rangaswamy V (1992) Degradation of endogenous fructose during catabolism of sucrose and mannitol in halophilic archaebacteria. Arch Microbiol 158: 356–363
Amachi T, Bowien B (1979) Characterization of two fructose bisphosphatase isoenzymes from the hydrogen bacteria Nocardia opaca 1b. J Gen Microbiol 113: 347–356
Bode CH, Goebel H, Stähler E (1968) Zur Eliminierung von Trübungsfehlern bei der Eiweißbestimmung mit der Biuretmethode. Z Klin Chem Klin Biochem 5: 419–422
Buchanan BB (1974) Orthophosphate requirement for the formation of phosphoenolpyruvate from pyruvate by enzyme preparations from photosynthetic bacteria. J Bacteriol 119: 1066–1068
Cooper RA, Kornberg HL (1974) Phosphoenolpyruvate synthetase and pyruvate, phosphate dikinase In: Boyer PD (ed) The enzymes, vol. 10. Academic Press, New York, pp 631–649
Daldal F, Fraenkel DG (1983) Assessment of a futile cycle involving reconversion of fructose-6-phosphate to fructose 1,6-biphosphate during gluconeogenetic growth of Escherichia coli. J Bacteriol 153: 390–394
Danson MJ (1988) Archaebacteria: the comparative enzymology of their central metabolic pathways. Adv Microb Physiol 29: 165–231
Eden G, Fuch G (1983) Autotrophic CO2 fixation in Acetobacterium woodii. II. Demonstration of enzymes involved. Arch Microbiol 135: 68–73
Eyzaguirre J, Jansen K, Fuchs G (1982) Phosphoenolpyruvate synthetase in Methanobacterium thermoautotrophicum. Arch Microbiol 132: 67–74
Fabry S, Hensel R (1987) Purification and characterization of d-glyceraldehyde-3-phosphate dehydrogenase from the thermophilic archaebacterium Methanothermus fervidus. Eur J Biochem 165: 147–155
Fiala G, Stetter KO (1986) Pyrococcus furiosus sp. nov. represents a novel genus of marine heterotrophic archaebacteria growing optimally at 100 °C. Arch Microbiol 145: 56–61
Frings W, Schlegel HG (1971) Synthese von Phosphoenolpyruvat aus Pyruvat durch Extrakte aus Hydrogenomonas eutropha Stamm H 16. Arch Mikrobiol 79: 220–230
Fuchs G, Stupperich E (1982) Autotrophic CO2 fixation in Methanobacterium thermoautotrophicum. Zentralbl Bakteriol Parasitenk Infektionskr Hyg Abt 1 Orig C 3: 277–288
Fuchs G, Stupperich E (1986) Carbon assimilation pathways in archaebacteria. Syst Appl Microbiol 7: 364–369
Fuchs G, Winter H, Steiner I, Stupperich E (1983) Enzymes of gluconeogenesis in the autotroph Methanobacterium thermoautotrophicum. Arch Microbiol 136: 160–162
Gottschalk G (1986) Bacterial metabolism, 2nd edn. Springer, Berlin Heidelberg New York
Hensel R, Laumann S, Lang J, Heumann H, Lottspeich F (1987) Characterization of two d-glyceraldehyde-3-phosphate dehydrogenases from the extremely thermophilic archaebacterium Thermoproteus tenax. Eur J Biochem 170: 325–333
Hochstein LI (1988) The physiology and metabolism of the extremely halophilic bacteria. In: Rodriguez-Valera F (ed) Halophilic bacteria, vol 2. CRC Press, Boca Raton, pp 67–83
Jansen K, Stupperich E, Fuchs G (1982) Carbohydrate synthesis from acetyl CoA in the autotroph Methanobacterium thermoautotrophicum. Arch Microbiol 132: 355–364
Josse J (1966) Constitutive inorganic pyrophosphatase of Escherichia coli I. Purification and catalytic properties. J Biol Chem 241: 1938
Krishnan G, Altekar W (1991) An unusual class I (Schiff base) fructose-1,6-bisphosphate aldolase from the halophilic archaebacterium Haloarcula vallismortis. Eur J Biochem 195: 343–350
Lanzotti V, Trincone A, Nicolaus B, Zillig W, DeRosa M, Gambacorta M (1989) Complex lipids of Pyrococcus and AN1, thermophilic members of archaebacteria belonging to Thermococcales. Biochim Biophys Acta 1004: 44–48
Mukund S, Adams MWW (1991) The novel tungsten-iron-sulfur protein of the hyperthermophilic archaebacterium, Pyrococcus furiosus, is an aldehyde ferredoxin oxidoreductase. J Biol Chem 266: 14208–14216
Pontremoli S, Horecker BL (1971) Fructose-1,6-diphosphatases. In: Boyer PD (ed) The enzymes, vol 4. Academic Press, New York, pp 612–646
Schäfer S, Barkowski C, Fuchs G (1986) Carbon assimilation by the autotrophic thermophilic archaebacterium Thermoproteus neutrophilus. Arch Microbiol 146: 301–308
Schäfer T, Schönheit P (1991) Pyruvate metabolism of the hyperthermophilic archaebacterium Pyrococcus furiosus. Acetate formation from acetyl-CoA and ATP synthesis are catalyzed by an acetyl-CoA synthetase (ADP forming). Arch Microbiol 155: 366–377
Schäfer T, Schönheit P (1992) Maltose fermentation to acetate, CO2 and H2 in the anaerobic hyperthermophilic archaeon Pyrococcus furiosus: evidence for the operation of a novel sugar fermentation pathway. Arch Microbiol 158: 188–202
Schäfer T, Selig M, Schönheit P (1993) Acetyl-CoA synthetase (ADP-forming) in archaea, a novel enzyme involved in acetate formation and ATP synthesis. Arch Microbiol 159: 72–83
Schönheit P, Wäscher C, Thauer RK (1978) A rapid procedure for the purification of ferredoxin from Clostridia using polyethyleneimine. FEBS Lett 89: 219–222
Scholz S, Sonnenbichler J, Schäfer W, Hensel R (1992) Di-myoinositol-1,1′-phosphate — a new inositol phosphate isolated from Pyrococcus woesei. FEBS Lett 306: 239–242
Thauer RK, Jungermann K, Decker K (1977) Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev 41: 100–180
Tomlinson GA, Koch TK, Hochstein LI (1974) The metabolism of carbohydrates by extremely halophilic bacteria: glucose metabolism via a modified Entner-Doudoroff pathway. Can J Microbiol 20: 1085–1091
Woese CR (1987) Bacterial evolution. Microbiol Rev 51: 221–271
Woese CR, Kandler O, Wheelis ML (1990) Towards a natural system of organisms: proposal for the domains archaea, bacteria and eucarya. Proc Natl Acad Sci USA 87: 4576–4579
Zeikus JG, Fuchs G, Kenealy W, Thauer RK (1977) Oxidoreductases involved in cell carbon synthesis of Methanobacterium thermoautotrophicum. J Bacteriol 132: 604–613
Zwickl P, Fabry S, Bogedain C, Haas A, Hensel R (1990) Glyceraldehyde-3-phosphate dehydrogenase from the hyperthermophilic archaebacterium Pyrococcus woesei: characterization of the enzyme, cloning and sequencing of the gene, and expression in Escherichia coli. J Bacteriol 172: 4329–4338
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schäfer, T., Schönheit, P. Gluconeogenesis from pyruvate in the hyperthermophilic archaeon Pyrococcus furiosus: involvement of reactions of the Embden-Meyerhof pathway. Arch. Microbiol. 159, 354–363 (1993). https://doi.org/10.1007/BF00290918
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00290918