Abstract
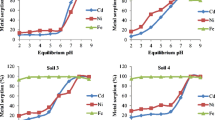
Cadmium sorption experiments were performed using four soil separates of different chemical and mineralogical composition, adding Cd solutions with initial concentrations ranging from 15 to 150 μg l−1. At the soils pHs, the sorption isotherms were a mixture of a constant partition isotherm with a high affinity one. Also, more than 90% of the initially added Cd was sorbed by all four soils. These results indicate a high affinity of these soils for trace amounts of Cd. The effect of pH was, in general, to decrease the amount of sorbed Cd as the pH decreased. The sorption isotherms were linear at all pH's. Also, the data fitted the Freundlich's sorption isotherm in all cases, but not Langmuir's. Freundlich's k values were found to be a good index of the relative Cd sorption affinities of the four soils at all the pH's used. It was found that the structural and chemical nature of the soils sorbent complex was a more important parameter than the CEC when studying the sorption of these trace amounts of Cd by soils.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Bittell, J. E. and Miller, R. J.: 1974, J. Env. Qual. 3, 250.
Friberg, L., Piscator, M., and Nordberg, G.: 1971, Cadmium in the Environment, Chemical Rubber Co. Press, Cleveland, Ohio.
Frink, C. R. and Peech, M.: 1963, Soil Sci. Soc. Am. Proc. 27, 527.
García-Miragaya, J. and Page, A. L.: 1976, Soil Sci. Soc. Am. J. 40, 658.
García-Miragaya, J.: 1975, ‘Sorption and Desorption of Cadmium by Soils and Soil Materials’, pH.D. Diss., Univ. of California, Riverside.
Giles, C. H:, MacEwan, T. H., Nakhwa, S. N., and Smith, D.: 1960, J. Chem. Soc. 3973.
Hahne, H. C. and Kroontje, W.: 1973, J. Env. Qual. 2, 444.
Jenne, E. A.: 1968, Adv. Chem. Series 73, 337.
John, M. K.: 1971, Env. Letters 2, 173.
Lagerwerff, J. V.: 1972, in J. J. Mortvedt, P. M. Giordano, and W. L. Lindsay (eds.), Micronutrients in Agriculture, Soil Sci. Soc. Am., Inc., Madison, Wisconsin, p. 593.
Lagerwerff, J. V. and Brower, D. L.: 1972, Soil Sci. Soc. Amer. Proc. 36, 734.
Levi-Minzi, R., Soldatini, G. F., and Riffaldi, R.: 1976, J. Soil Sci. 27, 10.
Mehra, O. P. and Jackson, M. L.: 1960, Clays Clay Miner. 7, 317.
Page, A. L.: 1974, U.S. Env. Prot. Agency, Cincinnati, Ohio, Rpt. No. EPA-670/2-74-005.
Page, A. L. and Bingham, F. T.: 1973, Resd. Reviews 48, 1.
Pratt, P. F. and Bair, F. L.: 1962, Hilgardia 33, 689.
Ragland, J. L. and Coleman, N. T.: 1960, Soil Sci. Soc. Am. Proc. 24, 457.
Santillán-Medrano, J. and Jurinak, J. J.: 1975, Soil Sci. Soc. Am. Proc. 39, 851.
Tiller, K. G.: 1967, Nature 214, 852.
Tiller, K. G.: 1968, Soil Sci. Trans. 9 (Adelaide, Australia) 2, 567.
Whittig, L. D.: 1965, in C. A. Black (ed.), ‘Methods of Soil Analysis’, Part 2, Agronomy 9, 671.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
García-Miragaya, J., Page, A.L. Sorption of trace quantities of cadmium by soils with different chemical and mineralogical composition. Water Air Soil Pollut 9, 289–299 (1978). https://doi.org/10.1007/BF00280677
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00280677