Summary
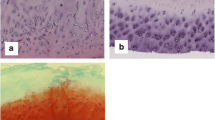
In the cartilage-pannus junction of 14 patients with rheumatoid arthritis (RA) and seven patients with osteoarthritis (OA), monoclonal antibodies to keratan sulphate (KS) and chondroitin sulphate (CS) stained a transitional fibroblastic zone (TFZ) within the pannus in nine RA patients and one OA patient. In three patients this was clearly localised to the cytoplasm of cells in this zone, but in all remaining cases KS and CS could be demonstrated in the surrounding matrix. This area was distinguished from adjacent pannus which contained many blood vessels and cells positive for MHC Class II antigen. Specific markers for glycosaminoglycans have been employed to demonstrate that chondrocyte-derived cells and matrix contribute to the changes seen at the cartilage-pannus junction in RA-affected joints.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Harris ED Jr (1976) Recent insights into the pathogenesis of the proliferative lesion in rheumatoid arthritis. Arthritis Rheum 19:68–72
Ball J (1969) Pathological aspects of rheumatoid arthritis. In: Hijmans W, Paul WD, Herschel H (eds) Early synovectomy in rheumatoid arthritis. Excerpta Medica Foundation, Amsterdam, pp 23–27
Gardner DL (1978) Pathology of rheumatoid arthritis. In: Scott JT (ed) Copeman's textbook of rheumatic diseases, 6th edn. Livingstone, Edingburgh London Melbourne New York, pp 199–250
Kobayashi I, Ziff M (1975) Electron microscopic studies of the cartilage-pannus junction in rheumatoid arthritis. Arthritis Rheum 18:475–483
Shiozawa S, Shiozawa K, Fujita T (1983) Morphologic observations in the early phase of the cartilage-pannus junction. Arthritis Rheum 26:472–478
Tateishi H (1973) Ultrastructure of the synovio-cartilage junction in rheumatoid arthritis. Kobe J Med Sci 19:51–66
Muirden KD (1982) Microscopic studies of the synovialcartilage junction in rheumatoid arthritis. Eur J Rheumatol Inflamm 5:30–38
Fassbender HG (1983) Histomorphological basis of articular cartilage destruction in rheumatoid arthritis. Coll Relat Res 3:141–151
Barrie HJ (1981) Histological changes in rheumatoid disease of the metacarpal and metatarsal heads as seen in surgical material. J Rheumatol 8:246–257
Mills K (1970) Pathology of the knee joint in rheumatoid arthritis. J Bone Joint Surg [Br] 52:746–756
Bromley M, Wooley DE (1984) Histopathology of the rheumatoid lesion. Arthritis Rheum 27:857–863
Mohr W, Wessinghage D (1978) The relationship between polymorphonuclear granulocytes and cartilage destruction in rheumatoid arthritis. Z Rheumatol 37:81–86
Mitrovic D (1985) The mechanism of cartilage destruction in rheumatoid arthritis (letter). Arthritis Rheum 28:1192–1193
Cooke TDV (1985) Rheumatoid arthritis pannus: true or false? (letter). Arthritis Rheum 28:1195–1197
Ropes MW, Bennett GA, Cobb S, Jacox R, Jessar RA (1958) 1958 revision of diagnostic criteria for rheumatoid arthritis. Bull Rheum Dis 9:175–176
Bancroft JD (1982) Enzyme histochemistry. In: Bancroft JD, Stevens A (eds) Theory and practice of histological techniques, 2nd edn. Livingstone, Edinburgh London Melbourne New York, p 403
Zanetti M, Ratcliffe A, Watt FM (1985) Two subpopulations of differentiated chondrocytes identified with a monoclonal antibody to keratan sulfate. J Cell Biol 101:53–59
Couchman JR, Caterson B, Christner JE, Baker JR (1984) Mapping by monoclonal antibody detection of glycosaminoglycans in connective tissues. Nature 307:650–652
Adams TE, Bodmer JG, Bodmer WF (1983) Production and characterization of monoclonal antibodies recognizing the α-chain subunits of human Ia alloantigens. Immunology 50:613–624
McKenzie JL, Fabre JW (1981) Human Thy-1: Unusual localization and possible functional significance in lymphoid tissues. J Immunol 126:843–850
Papadimitriou CS, Stein H, Papacharalampous NX (1980) Presence of α1-antichymotrypsin and α1-antitrypsin in haematopoietic and lymphoid tissue cells as revealed by the immunoperoxidase method. Pathol Res Pract 169:287–297
Stefansson K, Wollman RL, Moore BW, Arnason BGW (1982) S-100 protein in human chondrocytes. Nature 295:63–64
Palmer DG, Selvendran Y, Allen C, Revell PA, Hogg N (1985) Features of synovial membrane identified with monoclonal antibodies. Clin Exp Immunol 59:529–538
Hammerman D, Barland P, Janis R (1969) The structure and function of the synovial membrane in health and disease. In: Bittar EE, Bittar N (eds) Biological basis of medicine, vol 3. Academic Press, London New York, pp 269–309
Ziff M (1983) Factors involved in cartilage injury. J Rheumatol 11 (suppl):13–25
Mohr W, Kuhn C, Pelster B, Wessinghage D (1985) S100 protein in normal, osteoarthritic and arthritic cartilage. Rheumatol Int 5:273–277
Miossec P, Cavender D, Ziff M (1986) Production of interleukin 1 by human endothelial cells. J Immunol 136:2486–2491
Dingle JT, Saklatvala J, Hembry R, Tyler J, Fell HB, Jubb R (1979) A cartilage catabolic factor from synovium. Biochem J 184:177–180
Klareskog L, Johnell O, Hulth A (1984) Expression of HLA-DR and HLA-DQ antigens on cells within the cartilage-pannus junction in rheumatoid arthritis. Rheumatol Int 4 (suppl):11–15
Burmester GR, Menche D, Merryman P, Klein M, Winchester R (1983) Application of monoclonal antibodies to the characterization of cells eluted from human articular cartilage. Arthritis Rheum 26:1187–1195
Fell HB, Jubb RW (1977) The effect of synovial tissue on the breakdown of articular cartilage in organ culture. Arthritis Rheum 20:1359–1371
Mark K von der, Gauss V, Mark H von der, Müller P (1977) Relationship between cell shape and type of collagen synthesised as chondrocytes lose their cartilage phenotype in culture. Nature 267:531–532
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Allard, S.A., Muirden, K.D., Camplejohn, K.L. et al. Chondrocyte-derived cells and matrix at the rheumatoid cartilage-pannus junction identified with monoclonal antibodies. Rheumatol Int 7, 153–159 (1987). https://doi.org/10.1007/BF00270363
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00270363