Abstract
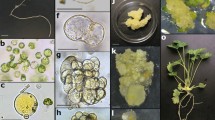
Conditions were established for efficient plant regeneration from four freshmarket cultivars of Lycopersicon esculentum. In order to increase the yield of viable protoplasts which are able to sustain cell divisions, the donor plants are preconditioned by incubation at 25°C in the dark for 18 hours, followed by a cold treatment at 4°C in the dark for the last 6 hours, prior to protoplast isolation. Browning of the dividing cell colonies can be prevented by culturing protoplasts in 100 μl droplets of low-melting agarose, surrounded by liquid medium. Alternatively, protoplasts can be cultured in liquid medium. In both procedures the plating efficiencies and percentage of shoot regeneration are increased, only when dilutions were performed with auxin-free culture medium. Shoot regeneration is obtained by using a two step procedure: initiation of greening of microcalli on a medium containing 0.2 M mannitol and 7.3 mM sucrose, which is followed by shoot development on a mannitol-free medium containing 0.5 M sucrose. In this way, plants can be regenerated within 3 months from the hybrid cultivars Bellina, Abunda, Sonatine and also from the true seedline Moneymaker. The latter one showed the highest regeneration frequency (30%).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BAP:
-
6-Benzylamino purine
- 2,4-D:
-
2,4-dichlorophenoxy acetic acid
- IAA:
-
indole acetic acid
- MES:
-
2-(N-morpholino)- ethane sulfonic acid
- NAA:
-
naphthalene acetic acid
- PE:
-
plating efficiency
References
Adams TL and JA Townsend (1983) Plant Cell Rep., 2, 165–168.
Brouwer R and JJM Dons (1986) Acta Bot. Neerl., 35, 45.
Cassels AC and M Barlass (1976) Physiol. Plant., 37, 239–246.
Cassels AC and M Barlass (1978) Physiol. Plant., 42, 236–242.
Frearson EM, JB Power and EC Cocking (1973) Dev. Biol., 33, 130–137.
Gamborg OL, RA Miller and K Ojima (1968) Exp. Cell. Res., 50, 151–158.
Haberlach GT, BA Cohen, NA Reichert, MA Baer, LE Towill, and JP Helgeson (1985) Plant Sci. Lett., 39, 67–74.
Hassanpour-Estahbanati A and Y Demarly (1985) J. Plant Physiol., 121, 171–174
Kao KN and MR Michayluk (1975) Planta 126, 105–110.
Murashige T and F Skoog (1962) Physiol. Plant., 15, 473–479.
Niedz RP, SM Rutter, LW Handley and KC Sink (1985) Plant Sci. Lett., 39, 199–204.
Nitsch JP and C Nitsch (1969) Science 163, 85–87.
Rick, CM (1982) In: Vasil, I.K., W.R. Scowcroft and K.J. Frey (eds): Plant Improvement and Somatic Cell Genetics pp. 1–28. Academic Press, New York.
Shahin EA (1985) Totipotency of tomato protoplasts Theor. Appl. Genet., 69, 235–240.
Shepard JF and RE Totten (1977) Plant Physiol., 60, 313–316.
Tabaeizadeh Z, C Bunniset-Bergounioux and C Perennes (1984) Physiol Veg. 22 (2), 223–229.
Tan MMC, F Van der Mark, J Hoogendijk, GAM Van Marrewijk and AJ Kool (1986) Acta Bot. Neerl., 35, 45.
Tan MMC, HS Boerrigter and AJ Kool (1987) A rapid procedure for plant regeneration from protoplast isolated from suspension cultures and leaf mesophyll cells of three Solanum species. Plant Sci. Lett., (in press).
Author information
Authors and Affiliations
Additional information
Communicated by L. K. Grill
Rights and permissions
About this article
Cite this article
Tan, ML.M.C., Rietveld, E.M., van Marrewijk, G.A.M. et al. Regeneration of leaf mesophyll protoplasts of tomato cultivars (L. esculentum): factors important for efficient protoplast culture and plant regeneration. Plant Cell Reports 6, 172–175 (1987). https://doi.org/10.1007/BF00268470
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00268470