Summary
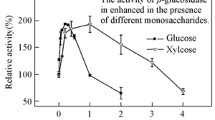
Clostridium stercorarium cultures grown on cellobiose contain both an extracellular and a cell-bound β-glucosidase activity. A substantial portion of the cell-bound enzyme could be extracted by osmotic shock, suggesting a periplasmic localization. The β-glucosidase present in culture supernatants was purified to homogeneity. It was found to be identical in all aspects tested with the cell-bound β-glucosidase. The enzyme exists as a monomer with an apparent molecular weight of 85.000 (SDS-PAGE) and a pI of 4.8. It shows optimal activity as pH 5.5 and 65° C. Thiol groups are essential for enzyme activity. In the presence of reducing agents and divalent cations the half-life of the purified enzyme was more than 5 h at 60°C. The enzyme hydrolyses at different rates a wide range of substrates including aryl-β-glucosides, cellobiose, and disordered cellulose. K m values were determined as 0.8 mM for p-nitrophenyl-β-glucoside (PNPG) and 33 mM for cellobiose. The cellular localization and the substrate specificity pattern are consistent with a dual role of the C. stercorarium β-glucosidase in cellulose saccharification: (1) Cleavage of cellobiose formed by exoglucanase and (2) degradation of cellodextrins produced by endoglucanase action.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aiba S, Kitai K, Imanaka T (1983) Cloning of a thermostable alpha-amylase gene from Bacillus stearothermophilus in Bacillus stearothermophilus and Bacillus subtilis. Appl Environ Microbiol 46:1059–1065
Ait N, Creuzet N, Cattaneo J (1979) Characterization and purification of thermostable β-glucosidase from Clostridium thermocellum. Biochem Biophys Res Commun 90:537–546
Ait N, Creuzet N, Cattaneo J (1982) Properties of β-glucosidase purified from Clostridium thermocellum. J Gen Microbiol 128:569–577
Alexander JK (1968) Purification and specificity of cellobiose phosphorylase from Clostridium thermocellum. J Biol Chem 243:2899–2904
Barras F, Chambost JP, Chippaux M (1984) Cellobiose metabolism in Erwinia: A genetic study. Mol Gen Genet 197:490–496
Breuil C, Wojtczak G, Saddler JN (1986) Production and localization of cellulases and β-glucosidase from the thermophilic fungus Thielavia terrestris. Biotechnol Lett 8:673–676
Bronnenmeier K, Staudenbauer WL (1988) Resolution of Clostridium stercorarium cellulase by fast protein liquid chromatography (FPLC). Appl Microbiol Biotechnol 27:432–436
Coughlan MP (1985) The properties of fungal and bacterial cellulases with comment on their production and application. Biotechnol Genet Eng Rev 3:39–109
Creuzet N, Frixon C (1983) Purification and characterization of an endoglucanase from a newly isolated thermophilic anaerobic bacterium. Biochimie 65:149–156
Hägerdal B, Ferchack JD, Pye EK (1980) Saccharification of cellulose by the cellulolytic enzyme system of Thermomonospora sp. Biotechnol Bioeng 22:1515–1526
Johnson EA, Madia A, Demain AL (1981) Chemically defined minimal medium for growth of the anaerobic cellulolytic thermophile Clostridium thermocellum. Appl Environ Microbiol 41:1060–1062
Johnson EA, Sakajoh M, Halliwell G, Madia A, Demain AL (1982) Saccharification of complex cellulosic substrates by the cellulase system from Clostridium thermocellum. Appl Environ Microbiol 43:1125–1132
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Madden RH (1983) Isolation and characterization of Clostridium stercorarium sp. nov., cellulolytic thermophile. Int J Syst Bacteriol 33:837–840
Nanda M, Bisaria VS, Ghose TK (1982) Localization and release mechanism of cellulases in Trichoderma reesei QM 9414. Biotechnol Lett 4:633–638
Ohmiya K, Shiria M, Kurachi Y, Shimizu S (1985) Isolation and properties of β-glucosidase from Ruminococcus albus. J Bacteriol 161:432–434
Patchett ML, Daniel RM, Morgan HW (1987) Purification and properties of a stable β-glucosidase from an extremely thermophilic anaerobic bacterium. Biochem J 243:779–787
Schimz KL, Broll B, John B (1983) Cellobiose phosphorylase (EC 2.4.1.20) of Cellulomonas: occurrence, induction, and its role in cellobiose metabolism. Arch Microbiol 135:241–249
Schmid G, Wandrey C (1987) Purification and partial characterization of a cellodextrin glucohydrolase (β-glucosidase) from Trichoderma reesei strain QM 9414. Biotechnol Bioeng 30:571–585
Schwarz WH, Bronnenmeier K, Gräbnitz F, Staudenbauer WL (1987) Activity staining of cellulases in polyacrylamide gels containing mixed linkage β-glucans. Anal Biochem 164:72–77
Sedmark JJ, Grossberg SE (1977) A rapid, sensitive assay for protein using Coomassie brilliant blue G250. Anal Biochem 79:544–552
Sheth K, Alexander JK (1969) Purification and properties of β-1,4-oligoglucan: orthophosphate glucosyltransferase from Clostridium thermocellum. J Biol Chem 244:457–464
Stoppok W, Rapp P, Wagner F (1982) Formation, location, and regulation of endo-1,4-β-glucanases and β-glucosidases from Cellulomonas uda. Appl Environ Microbiol 44:44–53
Tsukagoshi N, Ihara H, Yamagata H, Udaka S (1984) Cloning and expression of a thermostable α-amylase gene from Bacillus stearothermophilus in Escherichia coli. Mol Gen Genet 193:58–63
Wood TM (1971) The cellulases of Fusarium solani. Purification and specificity of the β-1,4-glucanase and β-glucosidase components. Biochem J 121:353–362
Wood TM, McCrae SI (1982) Purification and some properties of a (1→4)-β-d-glucan glucohydrolase associated with the cellulase from the fungus Penicillium funiculosum. Carbohydrate Res 110:291–303
Woodward J, Wiseman A (1982) Fungal and other β-glucosidases—their properties and applications. Enzyme Microb Technol 4:73–79
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bronnenmeier, K., Staudenbauer, W.L. Purification and properties of an extracellular β-glucosidase from the cellulolytic thermophile Clostridium stercorarium . Appl Microbiol Biotechnol 28, 380–386 (1988). https://doi.org/10.1007/BF00268200
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00268200