Summary
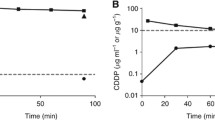
The phamacokinetics of carboplatin, ultrafilterable platinum, and total platinum after intraperitoneal (i. p.) administration were studied in peritoneal fluid, plasma, red blood cells (RBCs), and urine during a phase-I trial in patients with minimal, residual ovarian cancer. Samples were collected from 7 patients who had recived carboplatin (200–500 mg/m2) in 21 dialysis fluid. The fluid was withdrawn after a 4-h dwell. Platinum concentrations were measured by flameless atomic absorption spectrometry, and intact carboplatin was determined by HPLC with electrochemical detection. Peak concentrations of carboplatin in plasma were obtained 2 h after the end of instillation. The mean ratio of peak concentrations of carboplatin in instilled fluid and plasma was 24±11. The peritoneal clearance of carboplatin was 8±3 ml/min, which was 12 times less than the plasma clearance (93±32 ml/min). Due to this clearance ratio, the AUCs for the peritoneal cavity were about 10 times higher than those for plasma. On average, 34%±14% of the dose was still present in the instillation fluid that had been withdrawn after a dwell time of 4 h. In plasma, the mean value of AUC/Dnet (Dnet=Dose — amount recovered from the peritoneal cavity) after i.p. administration was comparable with that of AUC/D after i.v. administration. This means that unrecovered carboplatin (66%) was completely absorbed from the peritoneal cavity. It may be expected from this bioavailability that the maximum tolerated dose (MTD) of i.p.-administered carboplatin with a 4-h dwell is around 1.5 times higher than that after i.v. administration. Overall pharmacokinetic parameters of carboplatin and platinum in plasma were comparable after i.p. and i.v. administration.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Calvert AH, Harland SJ, Newell DR, Siddik ZH, Harrap KR (1985) Phase I studies with carboplatin at the Royal Marsden Hospital. Cancer Treat Rev 12 [Suppl A]: 51
Canetta R, Rozencweig M, Carter SK (1985) Carboplatin: the clinical spectrum to date. Cancer Treat Rev 12 [Suppl A]: 125
Casper ES, Kelsen DP, Alcock NW, Lewis JL Jr (1983) Ip cisplatin in patients with malignant ascites: pharmacokinetic evaluation and comparison with the i.v. route. Cancer Treat Rep 67: 235
Cleare MJ, Hydes PC, Malerbi BW, Watkins DM (1978) Anti-tumour platinum complexes: relationships between chemical properties and activity. Biochimie 60: 835
Dedrick RL (1985) Theoretical and experimental bases of intraperitoneal chemotherapy. Semin Oncol 12 [Suppl 4]: 1
Dedrick RL, Meyers CE, Bungay PM, DeVita VT Jr (1978) Pharmacokinetic rationale for peritoneal drug administration in the treatment of ovarian cancer. Cancer Treat Rep 62: 1
Egorin MJ, Van Echo DA, Tipping SJ, Olman EA, Whitacre MY, Thompson BW, Aisner J (1984) Pharmacokinetics and dosage reduction of cis-diammine (1,1-cyclobutanedicarboxylato)-platinum in patients with impaired renal function. Cancer Res 44: 5432
Elferink F, Van der Vijgh WJF, Klein I, Pinedo HM (1986a) Interaction of cisplatin and carboplatin with sodium thiosulfate: reaction rates and protein binding. Clin Chem 32: 641
Elferink F, Van der Vijgh WJF, Pinedo HM (1986b) On-line differential pulse polarographic detection of carboplatin in biological samples after chromatographic separation. Anal Chem 58: 2293
Elferink F, Van der Vijgh WJF, Klein I, Vermorken JB, Gall HE, Pinedo HM (1988) Pharmacokinetics of carboplatin after intravenous administration. Cancer Treat Rep (in press)
Gibaldi M, Perrier D (1982) Pharmacokinetics, 2nd edn. Marcel Dekker Inc, New York
Howell SB, Pfeifle CE, Wung WE, Olshen RA (1983) Intraperitoneal cisdiamminedichloroplatinum with systemic thiosulfate protection. Cancer Res 43: 1426
Levin VA (1986) Clinical anticancer pharmacology: some pharmacokinetic considerations. Cancer Treat Rev 13: 61
McVie JG, Ten Bokkel Huinink WW, Dubbelman R, Franklin H, Van der Vijgh WJF, Klein I (1985) Phase I study and pharmacokinetics of intraperitoneal carboplatin. Cancer Treat Rev 12 [Suppl A]: 35
Myers C (1984) The use of intraperitoneal chemotherapy in the treatment of ovarian cancer. Semin Oncol 11: 275
Neijt JP, Van der Burg MEL, Vriesendorp R, Van Lindert ACM, Van Lent M, Ten Bokkel Huinink WW, Van Oosterom AT, Kooyman CD, Hamerlynck JVTH, Van Houwelingen JC, Pinedo HM (1984) Kandomized trial comparing two combination chemotherapy regimens (HEXA-CAF vs CHAP-5) in advanced ovarian carcinoma. Lancet 2: 594
Perrier D, Mayersohn M (1982) Noncompartmental determination of the steady-state volume of distribution for any mode of administration. J Pharm Sci 71: 372
Ten Bokkel Huinink WW, Dubbelman R, Aartsen E, Franklin H, McVie JG (1985) Experimental and clinical results with intraperitoneal cisplatin. Semin Oncol 12 [Suppl 4]: 43
Van der Vijgh WJF, Klein I (1986) Protein binding of five platinum compounds. Comparison of two ultrafiltration systems. Cancer Chemother Pharmacol 18: 129
Vermorken JB, Van der Vijgh WJF, Klein I, Gall HE, Van Groeningen CJ, Hart GAM, Pinedo HM (1986) Pharmacolkinetics of free and total platinum species after rapid and prolonged infusions of cisplatin. Clin Pharmacol Ther 39: 136
Wiltshaw E (1985) Ovarian trials at the Royal Marsden. Cancer Treat Rev 12 [Suppl A]: 67
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Elferink, F., van der Vijgh, W.J.F., Klein, I. et al. Pharmacokinetics of carboplatin after intraperitoneal administration. Cancer Chemother. Pharmacol. 21, 57–60 (1988). https://doi.org/10.1007/BF00262740
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00262740