Abstract
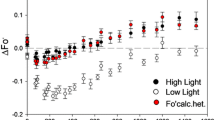
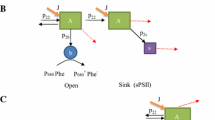
The light-dependent rate of photosystem-II (PSII) damage and repair was measured in photoautotrophic cultures of Dunaliella salina Teod. grown at different irradiances in the range 50–3000 μmol photons · m−2· s−1. Rates of cell growth increased in the range of 50–800 μmol photons·m−2·s−1, remained constant at a maximum in the range of 800–1,500 μmol photons·m−2 ·s−1, and declined due to photoinhibition in the range of 1500–3000 μmol photons·m−2·s−1. Western blot analyses, upon addition of lincomycin to the cultures, revealed first-order kinetics for the loss of the PSII reaction-center protein (D1) from the 32-kDa position, occurring as a result of photodamage. The rate constant of this 32-kDa protein loss was a linear function of cell growth irradiance. In the presence of lincomycin, loss of the other PSII reaction-center protein (D2) from the 34-kDa position was also observed, occurring with kinetics similar to those of the 32-kDa form of D1. Increasing rates of photodamage as a function of irradiance were accompanied by an increase in the steady-state level of a higher-molecular-weight protein complex (≈ 160-kDa) that cross-reacted with D1 antibodies. The steady-state level of the 160-kDa complex in thylakoids was also a linear function of cell growth irradiance. These observations suggest that photodamage to D1 converts stoichiometric amounts of D1 and D2 (i.e., the D1/D2 heterodimer) into a ≈160-kDa complex. This complex may help to stabilize the reaction-center proteins until degradation and replacement of D1 can occur. The results indicated an intrinsic half-time of about 60 min for the repair of individual PSII units, supporting the idea that degradation of D1 after photodamage is the rate-limiting step in the PSII repair process.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Chl:
-
chlorophyll
- PSI:
-
photosystem I
- PSII:
-
photosystem II
- D1:
-
the 32-kDa reaction-center protein of PSII, encoded by the chloroplast psbA gene
- D2:
-
the 34-kDa reactioncenter protein of PSII, encoded by the chloroplast psbD gene
- QA :
-
primary electron-accepting plastoquinone of PSII
References
Adir N, Shochat S, Ohad I (1990) Light-dependent D1 protein synthesis and translocation is regulated by reaction center II: Reaction center II serves as an acceptor for the D1 precursor. J Biol Chem 265: 12563–12568
Anderson J, Osmond CB (1987) Shade-sun responses: compromises between acclimation and photoinhibition. In: Kyle DJ, Osmond CB, Arntzen CJ (eds) Photoinhibition. Elsevier, Amsterdam, pp 1–38
Arnon D (1949) Copper enzymes in isolated chloroplasts. Polyphenol oxidase in Beta vulgaris. Plant Physiol 24: 1–15
Aro E-M, Virgin I, Andersson B (1993) Photoinhibition of photosystem II. Inactivation, protein damage and turnover. Biochim Biophys Acta 1143: 113–134
Barbato R, Friso G, Rigoni F, Frizzo A, Giacometti, GM (1992) Characterization of a 41 kDa photoinhibition adduct in isolated photosystem II reaction centres. FEBS Lett 309: 165–169
Barber J, Andersson B (1992) Too much of a good thing: light can be bad for photosynthesis. Trends Biochem Sci 17: 61–66
Cleland RE, Melis A, Neale PJ (1986) Mechanism of photoinhibition: photochemical reaction center inactivation in system II of chloroplasts. Photosynth Res 9: 79–88
Greer DM, Berry JA, Björkman O (1986) Photoinhibition of photosynthesis in intact bean leaves: role of light and temperature, and requirement for chloroplast-encoded protein synthesis during recovery. Planta 168: 253–260
Guenther JE, Melis, A (1990) Dynamics of photosystem II heterogeneity in Dunaliella salina (green algae). Photosynth Res 23: 195–204
Holden, M (1976) Chlorophylls. In: Goodwin TW (ed) Chemistry and biochemistry of plant pigments. Academic Press, New York, pp 6–9
Keren N, Gong H, Ohad I (1995) Oscillations of reaction center II-D1 protein degradation in vivo induced by repetitive light flashes. J Biol Chem 270: 806–814
Kim JH, Nemson JA, Melis A (1993) Photosystem II reaction center damage and repair in Dunaliella salina (green alga): Analysis under physiological and irradiance-stress conditions. Plant Physiol 103: 181–189
Kok B (1956) On the inhibition of photosynthesis by intense light. Biochim Biophys Acta 21: 234–244
Krömer S, Malmberg G, Gardeström P (1993) Mitochondrial contribution to photosynthetic metabolism. A study with barley (Hordeum vulgare L.) leaf protoplasts at different light intensities and CO2 concentrations. Plant Physiol 102: 947–955
Laemmli U (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Long SP, Humphries S, Falkowski PG (1994) Photoinhibition of photosynthesis in nature. Annu Rev Plant Physiol Plant Mol Biol 45: 633–662
Mattoo A, Hoffman-Falk H, Marder J, Edelman M (1984) Regulation of protein metabolism: coupling of photosynthetic electron transport to in vivo degradation of the rapidly metabolized 32 kDa protein of chloroplast membranes. Proc Natl Acad Sci USA 81: 1380–1384
Mattoo AK, Edelman M (1987) Intramembrane translocation and posttranslational palmitoylation of the chloroplast 32 kDa herbicide-binding protein. Proc Natl Acad Sci USA 84: 1497–1501
Maxwell, DP, Falk S, Trick CG, Huner NPA (1994) Growth at low temperature mimics high-light acclimation in Chlorella vulgaris Plant Physiol 105: 535–543
Maxwell DP, Falk S, Huner NPA (1995) Photosystem II excitation pressure and development of resistance to photoinhibition. 1. Light harvesting complex II abundance and zeaxanthin content in Chlorella vulgaris. Plant Physiol 107: 687–694
Melis A, Nemson JA (1995) Characterization of a 160 kD photosystem II reaction center complex isolated from photoinhibited Dunaliella salina thylakoids. Photosynth Res 46: 207–211
Melis A, Spangfort M, Andersson B (1987) Light-absorption and electron transport balance between photosystem-II and photosystem-I in spinach chloroplasts. Photochem Photobiol 45: 129–136
Mori H, Yamashita Y, Akasaka T, Yamamoto Y (1995) Further characterization of the loss of antenna chlorophyll-binding protein CP43 from Photosystem II during donor-side photoinhibition. Biochim Biophys Acta 1228: 37–42
Naus J, Melis A (1991) Changes of photosystem stoichiometry during cell growth in Dunaliella salina cultures. Plant Cell Physiol 32: 569–576
Neale PJ, Melis A (1990) Activation of a reserve pool of photosystem II in Chlamydomonas reinhardtii counteracts photoinhibition. Plant Physiol 92: 1196–1204
Neale PJ, Melis A (1991) Dynamics of Photosystem II heterogeneity during photoinhibition: Depletion of PSIIβ from non-appressed thylakoids during strong-irradiance exposure of Chlamydomonas reinhardtii. Biochim Biophys Acta 1056: 195–203
Park Y-I, Chow WS, Anderson JM (1995a) The quantum yield of photoinactivation of photosystem-II in pea leaves is greater at low than high photon exposure. Plant Cell Physiol 36: 1163–1167
Park Y-I, Chow WS, Anderson JM (1995b) Light inactivation of functional photosystem II in leaves of peas grown in moderate light depends on photon exposure. Planta 196: 401–411
Pick U, Karni L, Avron M (1986) Determination of ion content and ion fluxes in the halotolerant alga Dunaliella salina. Plant Physiol 81: 92–96
Powles S (1984) Photoinhibition of photosynthesis induced by visible light. Annu Rev Plant Physiol 35: 15–44
Prasil O, Adir N, Ohad I (1992) Dynamics of photosystem II: mechanism of photoinhibition and recovery processes. In: Barber J (ed) The photosystems: structure, function and molecular biology. Elsevier, Amsterdam, pp 295–348
Shibata K (1958) Spectrophotometry of biological materials. J Biochem 45: 559–604
Smith BM, Morrissey PJ, Guenther JE, Nemson JA, Harrison MA, Allen JF, Melis A (1990) Response of the photosynthetic apparatus in Dunaliella salina (green algae) to irradiance stress. Plant Physiol 93: 1433–1440
Sukenik A, Bennett J, Falkowski PG (1988) Changes in the abundance of individual apoproteins of light harvesting chlorophyll a/b-protein complexes of photosystem I and II with growth irradiance in the marine chlorophyte Dunaliella tertiolecta. Biochim Biophys Acta 932: 206–215
Vasilikiotis C, Melis A (1994) Photosystem II reaction center damage and repair cycle: chloroplast acclimation strategy to irradiance stress. Proc Natl Acad Sci USA 91: 7222–7226
Author information
Authors and Affiliations
Corresponding author
Additional information
The work was supported by grant 94-37100-7529 from the US Department of Agriculture, National Research Initiative Competitive Grants Program.
Rights and permissions
About this article
Cite this article
Baroli, I., Melis, A. Photoinhibition and repair in Dunaliella salina acclimated to different growth irradiances. Planta 198, 640–646 (1996). https://doi.org/10.1007/BF00262653
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00262653