Summary
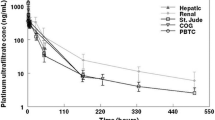
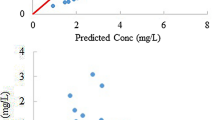
A flow-limited physiologic pharmacokinetic model using volume terms, flow rates, distribution ratios, metabolic rate constants, and clearance terms restricted to physiologic or measured values was used to simulate the disposition of cisplatin in children and adolescents. Physiologic model simulations of parent cisplatin and total platinum serum concentrations were not statistically different from concentrations of these platinum species measured in 14 patients. A simplified first-order multicompartment operational model was also developed, and produced comparable simulations of parent cisplatin disposition but less accurate simulations of total platinum serum concentrations. These data provide further clarification of cisplatin disposition in humans and provide the basis for previously observed changes in the renal clearance of total platinum.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bannister SJ, Sternson LA, Repta AJ, James GW (1977) Measurement of free circulating cis-dichlorodiammineplatinum (II) in plasma. Clin Chem 23:2258–2262
Bischoff KB (1975) Some fundamental considerations of the applications of pharmacokinetics to cancer chemotherapy. Cancer Chemother Rep 59:777–793
Bischoff KB, Dedrick RL, Zaharko DS, Longsheth JA (1971) Methotrexate pharmacokinetics. J Pharm Sci 60:1128–1133
Casper ES, Kelsen DP, Alcock NW, Yung CW (1979) Platinum concentrations in bile and plasma following rapid and 6-hour infusions of cis-dichlorodiammineplatinum (II). Cancer Treat Rep 63:2023–2025
Crom WR, Evans WE, Pratt CB, Senzer N, Denison M, Green AA, Hayes FA, Yee GC (1981) Cisplatin disposition in children and adolescents with cancer. Cancer Chemother Pharmacol 6:95–99
Evans WE, Crom WR, Green AA, Hayes FA, Pratt CB (1980) cis-Diamminedichloroplatinum disposition in children: Parameters of a multicompartment kinetic model. In: Nelson JD, Grassi C (eds) Current chemotherapy and infectious disease. American Society for Microbiology, Washington, pp 1508–1510
Holdener EE, Park CH, Belt JR, et al. (1978) Effect of mannitol and human plasma on the cytotoxicity of cis-dichlorodiammineplatinum. Clin Res 26:4364
Leroy AF, Lutz RJ, Dedrick RL, Litterst CL, Guarino AM (1979) Pharmacokinetic study of cis-diamminedichloroplatinum (II) (DDP) in the beagle dog: Thermodynamic and kinetic behavior of DDP in a biologic milieu. Cancer Treat Rep 63:59–71
Lowrey GH (1978) Growth and development of children, 7th edn. Year Book Medical Publishers, Chicago, p 207
Pennington RH (1965) Introductory computer methods and numerical analysis. Macmillan, New York
Pratt CB, Hayes A, Green AA, Evans WE, Senzer N, Howarth CB, Ransom VL, Crom WR (1981) Pharmacokinetic evaluation of cisplatin in children with malignant solid tumors: a phase II study. Cancer Treat Rep 65:1021–1026
Prestayko AW, D'Aoust JC, Issell BF, Croule ST (1979) Cisplatin (cis-diamminedichloroplatinum-II). Cancer Treat Rev 6:17–39
Sawchuk RJ, Zaske DE (1976) Pharmacokinetics of dosing regimens which utilize multiple intravenous infusions: gentamicin in burn patients. J Pharmacokinet Biopharm 4:183–195
Smith PHS, Taylor DM (1974) Distribution and retention of the antitumor agent 195mPt-cis-dichlorodiammineplatinum (II) in man. J Nucl Med 15:349–351
Author information
Authors and Affiliations
Additional information
Supported in part by Cancer Center Support (CORE) Grant CA 21765, Solid Tumor Program Project Grant CA 23099, Biomedical Research Support Grant RR 05584, and by ALSAC
Rights and permissions
About this article
Cite this article
Evans, W.E., Crom, W.R., Tsiatis, A. et al. Pharmacokinetic modeling of cisplatin disposition in children and adolescents with cancer. Cancer Chemother. Pharmacol. 10, 22–26 (1982). https://doi.org/10.1007/BF00257231
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00257231