Summary
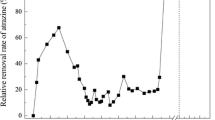
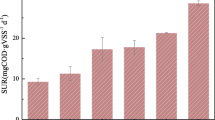
Anaerobic phenol degrading consortia were selected in sewage sludge and culture conditions were improved to allow maximum degradation rates of 0.9 g/l·d. Phenol had to be added in two portions of 0.45 g/l at intervals of 12 h to keep the fermentation at stable conditions. From U-14C-phenol little benzoate and acetate were formed as intermediates under a N2:CO2 gas phase. Final products were methane and CO2. When methanogenesis was inhibited by BESA, less labeled methane and CO2 were formed and labeled acetate remained undegraded. Turnover rates of phenol were significantly reduced in the presence of a H2:CO2 gas atmosphere and benzoate was formed from phenol and CO2. Acetate did not accumulate remarkably. After the H2:CO2 was converted to methane or was exchanged by N2:CO2 the accumulated benzoate was further degraded to methane and CO2. Elevated pools of acetate in sewage sludge led also to a reduction of the phenol degradation rates and presumably to an increased concentration of benzoate. In fresh sewage sludge benzoate degradation proceeds immediately, while the degradation of phenol starts only after a lag-phase of 3–10 days.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Balch WE, Fox GE, Magrum LJ, Woese CR, Wolfe RS (1979) Methanogens: Reevaluation of a unique biological group. Microbiol Rev 43:260–296
Barik S, Brulla WJ, Bryant MP (1985) PA1, a versatile anaerobe obtained in pure culture, catabolizes benzoids and other compounds in syntrophy with hydrogenotrophs, and P-2 plus Wolinella sp. degrades benzoids. Appl Environ Microbiol 50:304–310
Bettmann H, Rehm HJ (1985) Degradation of phenol by polymer entrapped microorganisms. Appl Microbiol Biotechnol 20:285–290
Bray GA (1960) A simple efficient liquid scintillator for counting aqueous solutions in a liquid scintillation counter. Anal Biochem 1:279–285
Chmielowski J, Grossmann A, Labazek S (1965) Biochemical degradation of some phenols during methane fermentation. Zesz Nauk Politech Slask Inz Sanit 8:97–122
DFG-Forschungsbericht (1982) Schadstoffe im Wasser, Vol. II Phenole. Deutsche Forschungsgemeinschaft (ed), D-5300 Bonn 2, Harald Boldt Verlag, Boppard
Dutton PH, Evans WC (1967) Dissimilation of aromatic substrates by Rhodopseudomonas palustris. Biochem J 104:30–31
Ehrhardt HM, Rehm HJ (1985) Phenol degradation by microorganisms adsorbed on activated carbon. Appl Microbiol Biotechnol 21:32–36
Eklund D (1985) The effect of sorbic acid and esters of phydroxybenzoic acid on the proton motive force in Escherichia coli membrane vesicles. J Gen Microbiol 131:73–76
Ferry JG, Wolfe RS (1976) Anaerobic degradation of benzoate to methane by a microbial consortium. Arch Microbiol 107:33–40
Fina LR, Bridges RL, Coblentz TH, Roberts FF (1978) The anaerobic decomposition of benzoic acid during methane fermentation. III. The fate of carbon four and the identification of propanoic acid. Arch Microbiol 118:169–172
Healy JB, Young LY (1978) Catechol and phenol degradation by a methanogenic population of bacteria. Appl Environ Microbiol 78:216–218
Kristjansson JK, Schönheit P, Thauer RK (1982) Different K s-values for hydrogen of methanogenic and sulfate reducing bacteria: An explanation for the apparent inhibition of methanogenesis by sulfate. Arch Microbiol 131:278–282
Mountfort DO, Bryant MP (1982) Isolation and characterization of an anaerobic syntrophic benzoate-degrading bacterium from sewage sludge. Arch Microbiol 133:249–256
Mountfort DO, Brulla WJ, Krumholz LR, Bryant MP (1984) Syntrophus buswelli gen. nov. sp. nov.: A benzoate catabolizer from methanogenic ecosystems. Int J Syst Bact 34:216–217
Oshima T (1965) On the anaerobic metabolism of aromatic compounds in the presence of nitrate by soil microorganisms. Zeitschrift f. Allg. Mikrobiologie 5:386–394
Schink B, Pfennig N (1982) Fermentation of Trihydroxybenzenes by Pelobacter acidigallici gen. nov. sp. nov., a new strictly anaerobic non-sporforming bacterium. Arch Microbiol 133:195–201
Stanier RY (1950) Problems of bacterial oxidative metabolism. Bact Rev 14:179–191
Szewzyk M, Szewzyk R, Schink B (1985) Methanogenic degradation of hydroquinone and catechol via reductive dehydroxylation to phenol. FEMS Microbiol Ecol 31:79–87
Taylor BF, Campbell WL, Chinoy I (1970) Anaerobic degradation of the benzene nucleus by a facultatively anaerobic microorganism. J Bact 102:430–437
Taylor BF, Heeb MJ (1972) The anaerobic degradation of aromatic compounds by a denitrifying bacterium. Arch Microbiol 83:165–171
Tarvin D, Buswell AM (1934) The methane fermentation of organic acids and carbohydrates. J Am Chem Soc 56:1751–1755
Thauer RK, Jungermann K, Decker K (1977) Energy conservation in chemotrophic anaerobic bacteria. Bact Rev 41:100–180
Tschech A, Schink B (1985) Fermentative degradation of resorcinol and resorcylic acids. Arch Microbiol 143:52–59
Wagner R (1914) Über Benzolbakterien. Zschr Gärungsphysiol 4:289–319
Widdel F (1980) Anaerober Abbau von Fettsäuren und Benzoesäure durch neu isolierte Arten sulfatreduzierender Bakterien. Dissertation, Georg-August-Universität Göttingen, Göttingen
Williams RJ, Evans WC (1973) Anaerobic metabolism of aromatic substrates by certain microorganisms. Biochem Soc Transactions1:186–187
Williams RJ, Evans WC (1975) The metabolism of benzoate by Moraxella sp. through anaerobic nitrate respiration. Biochem J 148:1–10
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Knoll, G., Winter, J. Anaerobic degradation of phenol in sewage sludge. Appl Microbiol Biotechnol 25, 384–391 (1987). https://doi.org/10.1007/BF00252552
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00252552