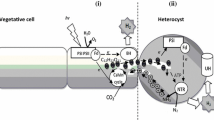
Abstract
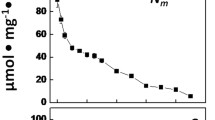
We have investigated the utilization of [14C]-fructose by whole filaments and isolated heterocysts of Anabaena variabilis ATCC 29413, a strain which is capable of fructose-dependent heterotrophic growth. The experimental conditions were chosen such that both transport and subsequent metabolism were studied. The apparent Km for fructose was 60 μmM, close to the results of previous studies. Rates of fructose utilization were the same in light and darkness. When photosynthetic CO2 fixation was possible, almost all the label appeared as cell-carbon. In darkness or in the presence of DCMU appreciable amounts of label were released as CO2. Isolated heterocysts with high rates of endogenous metabolism were not capable of utilizing added fructose at significant rates. The effects of oxygen concentration on the metabolism of added fructose in darkness showed that uptake was saturated at low pO2 values. Increasing the pO2 values lead to an increase in the ratio between the lable released as CO2 and that recovred as cell-carbon. These results suggest that fructose is taken up only by the vegetative cells but carbon derived from added fructose can be released as CO2 as a result of respiration in the heterocysts. Fructose utilization was inhibited by uncouplers. The greatest inhibition was found when both (delta) (psi) and (delta) pH were abolished. High concentrations of erythrose inhibited fructose utilization. None of the other potential analogs tested had any effect.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Beauclerk AAD, Smith AJ (1978) Transport of d-glucose and 3-O-methyl-d-glucose in the cyanobacteria Aphanocapsa 6714 and Nostoc strain Mac. Eur J Biochem 82: 187–197
Burris RH (1974) Methodology. In: Quisped A (ed) The biology of nitrogen fixation. North Holland, Amsterdam, pp 9–13
Doolittle WF (1979) The cyanobacterial genome, its expression, and the control of that expression. Adv Microb Physiol 20:2–102
Flores E, Schmetterer G (1986) Interaction of fructose with the glucose permease of the cyanobacterium Synechocystis sp. strain PCC 6803. J Bacteriol 166:693–696
Flores E, Wolk CP (1985) Indentification of facultatively heterotrophic, N2−fixing cyanobacteria able to receive plasmid vectors from Escherichia coli by conjugation. J Bacteriol 162:1339–1341
Fogg GE (1949) Growth and heterocyst production in Anabaena cylindria lemm. II. In relation to carbon and nitrogen metabolism. Ann Bot 13:299–302
Hawkesford MJ, Reed RH, Rowell P, Stewart WDP (1981) Nitrogenase activity and membrane electrogenesis in the cyanobacterium Anabaena variabilis Kürtz. Eur J Biochem 115:519–523
Haury JF, Spiller H (1981) Fructose uptake and influence on growth and nitrogen fixation by Anabaena variabilis. J Bacteriol 147:227–235
Hoare DS, Ingram LO, Thurston EL, Walkup R (1971) Dark heterotrophic growth of an endophytic blue-green alga. Arch Microbiol 78:310–321
Jensen BB (1983) Energy requirement for diazotrophic growth of the cyanobacterium Anabaena variabilis determined from growth yields in the dark. J Gen Microbiol 129:2633–2640
Jensen BB, Cox RP (1983) Effect of oxygen concentration on dark nitrogen fixation and respiration in cyanobacterium. Arch Microbiol 135:287–292
Jensen BB, Cox RP, Burris RH (1986) Isolation of cyanobacterial heterocysts with high sustained dinitrogen-fixation capacity supported by endogenous reductants. Arch Microbiol 145: 241–247
Lang NJ, Krupp JM, Koller AL (1987) Morphological and ultrastructural changes in vegetative cells and heterocysts of Anabaena variabilis grown with fructose. J Bacteriol 169:920–923
Lockau W (1984) Properties and intracellular localization of a cyanobacterial (Ca2+, Mg2+)-ATPase. In: Subesma C (ed) Advances in photosynthesis research, vol II. Martinus Nijhoff/dr W. Junk Publishers, Amsterdam, pp 603–606
McKinney G (1941) Absorption of light by chlorophyll solutions. J Biol Chem 150:315–322
Pelroy RA, Rippka R, Stanier RY, (1972) The metabolism of glucose by unicellular blue-green algae. Arch Microbiol 87:303–322
Privalle LS, Burris RH (1984) d-Erythrose supports nitrogenase activity in isolated Anabaena sp strain 7120 heterocysts. J Bacteriol 157:350–356
Raboy B, Padan E (1978) Active transport of glucose and α-methylglucoside in the cyanobacterium Plectonema boryanum J Biol Chem 253:3287–3291
Raboy B, Padan E, Shilo M (1976) Heterotrophic capacities of Plectonema boryanum. Arch Microbiol 110:77–85
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stanier RY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
Schmetter G, Flores E (1988) Uptake of fructose by the cyanobacterium Nostoc sp. ATCC 29150. Biochim Biophys Acta 942:33–37
Smith AJ (1982) Modes of cyanobacterial carbon metabolism. In: Carr NG, Whitton BA (ed) The biology of cyanobacteria. Blackwell Scientific, Oxford, pp 47–52
Smith AJ (1983) Modes of cyanobacterial carbon metabolism. Ann Microbiol (Inst Pasteur) 134B:93–113
Watanabe A, Yamamoto Y (1967) Heterotrophic nitrogen fixation by the blue-green alga Anabaenopsis circularis. Nature (Lond) 214:738
Wolk CP (1982) Heterocysts. In: Carr NG, Whitton BW (eds) The biology of cyanobacteria. Blackwell, Oxford, pp 359–386
Wolk CP, Schaffer PW (1976) Heterotrophic micro-and macrocultures of a nitrogen-fixing cyanobacterium. Arch Microbiol 110:145–147
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jensen, B.B. Fructose utilization by the cyanobacterium Anabaena variabilis studied using whole filaments and isolated heterocysts. Arch. Microbiol. 154, 92–98 (1990). https://doi.org/10.1007/BF00249184
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00249184