Abstract
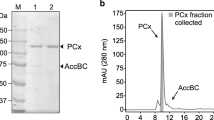
The enzyme catalysing the first step in the anaerobic degradation pathway of phenylacetate was purified from a denitrifying Pseudomonas strain KB 740. It catalyses the reaction phenylacetate+CoA+ATP → phenylacetyl-CoA+AMP+PPi and requires Mg2+. Phenylacetate-CoA ligase (AMP forming) was found in cells grown anaerobically with phenylacetate and nitrate. Maximal specific enzyme activity was 0.048 μmol min-1 x mg-1 protein in the mid-exponential growth phase. After 640-fold purification with 18% yield, a specific activity of 24.4 μmol min-1 mg-1 protein was achieved. The enzyme is a single polypeptide with Mr of 52 ±2 kDa. The purified enzyme shows high specificity towards the aromatic inducer substrate phenylacetate and uses ATP preferentially; Mn2+ can substitute for Mg2+. The apparent K m values for phenylacetate, CoA, and ATP are 60, 150, and 290 μM, respectively. The soluble enzyme has an optimum pH of 8.5, is insensitive to oxygen, but is rather labile and requires the presence of glycerol and/or phenylacetate for stabilization. The N-terminal amino acid sequence showed no homology to other reported CoA-ligases. The expression of the enzye was studied by immunodetection. It is present in cells grown anaerobically with phenylacetate, but not with mandelate, phenylglyoxylate, benzoate; small amounts were detected in cells grown aerobically with phenylacetate.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Altenschmidt U, Fuchs G (1992) Novel aerobic 2-aminobenzoate metabolism. Purification and characterization of 2-aminobenzoate-CoA ligase, localisation of the gene on a 8-kbp plasmid, and cloning and sequencing of the gene from a denitrifying Pseudomonas sp. Eur J Biochem 205: 721–727
Altenschmidt U, Oswald B, Fuchs G (1991) Purification and characterization of benzoate-CoA ligase and 2-aminobenzoate-CoA ligases from a denitrifying Pseudomonas sp. J Bacteriol 173: 5494–5501
Bak F, Widdel F (1986) Anaerobic degradation of phenol and phenol derivatives by Desulfobacterium phenolicum sp. nov. Arch Microbiol 146: 177–180
Balba MT, Evans WC (1979) The methanogenic fermentation of omega-phenylalkane carboxylic acids. Biochem Soc Trans 7: 403–405
Biegert T, Altenschmidt U, Eckerskorn C, Fuchs G (1993) Enzymes of anaerobic metabolism of phenolic compounds. 4-Hydroxybenzoate-CoA ligase from a denitrifying Pseudomonas species. Eur J Biochem 213: 555–561
Braun K, Gibson DT (1984) Anaerobic degradation of 2-aminobenzoate (anthranilic acid) by denitrifying bacteria. Appl Environ Microbiol 48: 102–107
Chang KH, Liang PH, Beck W, Scholten JD, Dunaway-Mariano D (1992) Isolation and characterization of the 3 polypeptide components of 4-chlorobenzoate dehalogenase from Pseudomonas sp. strain CBS-3. Biochemistry 31: 5605–5610
Dagley S (1986) Biochemistry of aromatic hydrocarbon degradation in Pseudomonas. In: Sokatch IR (ed) The bacteria, vol 10. Academic Press, New York, pp 527–555
Dangel W, Brackmann R, Lack A, Mohamed M, Koch J, Oswald B, Seyfried B, Tschech A, Fuchs G (1991) Differential expression of enzyme activities initiating anoxic metabolism of various aromatic compounds via benzoyl-CoA. Arch Microbiol 155: 256–262
D'Ari L, Barker HA (1985) p-Cresol formation by cell-free extracts of Clostridium difficile. Arch Microbiol 143: 311–312
Dawson RMC, Elliott DC, Elliott WH, Tones KM (1986) Data for biochemical research, 3rd edn. Oxford Science Publications, Oxford, pp 316–397
Decker K (1959) Die aktivierte Essigsäure. Enke, Stuttgart
Eckerskorn C, Mewes W, Goretzki H, Lottspeich F (1988) A new siliconized-glass fiber as support for protein-chemical analysis of electroblotted proteins. Eur J Biochem 176: 509–519
Elder DJE, Morgan P, Kelly DJ (1992) Anaerobic degradation of trans-cinnamate and ω-phenylalkane carboxylic acids by the photosynthetic bacterium Rhodopseudomonas palustris: evidence for a β-oxidation mechanism. Arch Microbiol 157: 148–154
Evans WC, Fuchs G (1988) Anaerobic degradation of aromatic compounds. Annu Rev Microbiol 42: 289–317
Fogg GC, Gibson J (1990) 4-Hydroxybenzoate-coenzyme A ligase from Rhodopseudomonas palustris. Abstr Annu Meet Am Soc Microbiol, p 242
Geissler JF, Harwood CS, Gibson J (1988) Purification and properties of benzoate-coenzyme A ligase, a Rhodopseudomonas palustris enzyme involved in the anaerobic degradation of benzoate. J Bacteriol 170: 1709–1714
Gianfreda L, Searfi MR (1991) Enzyme stabilization: state of the art. Mol Cell Biochem 100: 97–128
Gibson DT, Subramanian V (1984) Microbial degradation of aromatic hydrocarbons. In: Gibson DT (ed) Microbial degradation of organic compounds. Marcel Dekker, New York, pp 181–252
Gibson KJ, Gibson J (1992) Potential early intermediates in anaerobic benzoate degradation by Rhodopseudomonas palustris. Appl Environm Microbiol 58: 696–698
Grbic-Galic D (1985) Fermentative and oxidative transformation of ferulate by a facultatively anaerobic bacterium isolated from sewage sludge. Appl Environ Microbiol 50: 1052–1057
Gross GG (1981) Phenolic acids. In: Stumpf PK, Conn EE (eds) The biochemistry of plants, vol 7. Academic Press, New York, pp 301–315
Gross GG, Mansell RH, Zenk MH (1975) Hydroxycinnamate: CoA ligase from lignifying tissue of higher plants. Some properties and taxonomic distribution. Biochem Physiol Pflanz 168: 41–51
Hancock K, Tsang VCW (1983) India ink staining of proteins on nitrocellulose paper. Anal Biochem 133: 157–162
Harwood CS, Gibson J (1986) Uptake of benzoate by Rhodopseudomonas palustris grown anaerobically in light. J Bacteriol 165: 504–509
Healy JB, Young LY, Reinhard M (1980) Methanogenic decomposition of ferulic acid, a model lignin derivative. Appl Environ Microbiol 39: 436–444
Heising S, Brune A, Schink B (1991) Anaerobic degradation of 3-hydroxybenzoate by a newly isolated nitrate-reducing bacterium. FEMS Microbiol Lett 84: 267–272
Hutber GN, Ribbons DW (1983) Involvement of coenzyme A esters in metabolism of benzoate and cyclohexanecarboxylate by Rhodopseudomonas palustris. J Gen Microbiol 129: 2413–2420
Kim M-K, Harwood CS (1991) Regulation of benzoate-CoA ligase in Rhodopseudomonas palustris. FEMS Microbiol Lett 83: 199–204
Kim YS, Chae HZ (1991) Purification and properties of malonyl-CoA synthetase from Rhizobium japonicum. Biochem J 273: 511–516
Knobloch K-H, Hahlbrock K (1977) 4-Coumarate: CoA ligase from cell suspension cultures of Petroselinum hortense Hoffm. Partial purification, substrate specificity, and further properties. Arch Biochem Biophys 184: 237–248
Koch J, Eisenreich W, Bacher A, Fuchs G (1993) Products of enzymatic reduction of benzoyl-CoA, a key reaction in anaerobic aromatic metabolism. Eur J Biochem 211: 649–661
Koch J, Fuchs G (1992) Enzymatic reduction of benzoyl-CoA to alicyclic compounds, a key reaction in anaerobic aromatic metabolism. Eur J Biochem: 195–202
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Lindl T, Kreuzaler F, Hahlbrock K (1973) Synthesis of p-coumaroyl coenzyme A with a partially purified p-coumarate-CoA ligase from cell suspension cultures of soybean (Glycine max). Biochim Biophys Acta 302: 457–464
Maniatis T, Fritsch EF, Sambrock J (ed) (1982) Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Markwell MAK, Haas SM, Bieber LL, Tolbert NE (1978) A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem 87: 206–210
Martinez-Blanco H, Reglero A, Rodriguez-Aparicio LB, Luengo JM (1990) Purification and biochemical characterization of phenylacetyl-CoA ligase from Pseudomonas putida. A specific enzyme for the catabolism of phenylacetic acid. J Biol Chem 265: 7084–7090
Mohamed M, Seyfried B, Tschech A, Fuchs G (1993) Anaerobic oxidation of phenylacetate and 4-hydroxyphenylacetate to benzoyl-CoA and CO2 in denitrifying Pseudomonas species: evidence for an α-oxidation mechanism. Arch Microbiol 159: 570–580
Morrissey JH (1981) Silverstain for proteins in polyacrylamide gels: a modified procedure with enhanced uniform sensitivity. Anal Biochem 117: 307–310
Nilsson BO, Svolander PC, Larsson A (1987) Immunization of mice and rabbits by intrasplenic deposition of nanogram quantities of protein attached to sepharose beads or nitrocellulose paper strips. J Immunol. Methods 99: 67–75
Nozawa T, Maruyama Y (1988) Denitrification by a soil bacterium with phthalate and other aromatic compounds as substrates. J Bacteriol 170: 2501–2505
Person GG, Wall JD, Emerich DW (1990) Purification and properties of acetyl-CoA synthetase from Bradyrhizobium japonicum bacteroids. Biochem J 267: 179–183
Rhodes MJC, Wooltorton LSC (1975) The p-coumaroyl CoA ligase of potato tubers. Phytochemistry 14: 2161–2164
Rudolphi A, Tschech A, Fuchs G (1991) Anaerobic degradation of cresols by denitrifying bacteria. Arch Microbiol 155: 238–248
Schnell S, Schink B (1991) Anaerobic aniline degradation via reductive deamination of 4-aminobenzoyl-CoA in Desulfobacterium anilini. Arch Microbiol 155: 183–190
Sembiring T, Winter J (1989) Anaerobic degradation of phenylacetic acid by mixed and pure cultures. Appl Microbiol Biotechnol 31: 84–88
Seyfried B, Tschech A, Fuchs G (1991) Anaerobic degradation of phenylacetate and 4-hydroxyphenylacetate by denitrifying bacteria. Arch Microbiol 155: 249–255
Spoelstra SF (1978) Degradation of tyrosine in anaerobically stored piggery wastes and in pig feces. Appl Environ Microbiol 36: 631–638
Subba-Rao PV, Francis NLM, Metcalfe DD (1983) An avidinbiotin micro ELISA for rapid measurement of total and allergenspecific human IgE. J Immunol Methods 57: 71–85
Utkin IB, Yakimov MM, Matveea LN, Kozlyak EI, Rogozhin IS, Solomon ZG, Bezborodov AM (1991) Degradation of styrene and ethyl benzene by Pseudomonas species Y2. FEMS Microbiol Lett 77: 237–242
Walton E, Butt VS (1971) The demonstration of cinnamoyl-CoA synthesis activity in leaf extracts. Phytochemistry 10: 295–304
Ward LA, Johnson KA, Robinson JM, Yokoyama MT (1987) Isolation from swine feces of a bacterium which decarboxylates p-hydroxyphenylacetic acid to 4-methylphenol (p-cresol). Appl Environ Microbiol 53: 189–192
Widdel F, Pfennig N (1984) Dissimilatory sulfate-or sulfur-reducing bacteria. In: Krieg NR, Holt IG (eds) Bergey's manual of systematic bacteriology, vol 1, 9th edn. Williams & Wilkins, Baltimore London, pp 663–679
Widdel F, Kohring GW, Mayer F (1983) Studies on dissimilatory sulfate-reducing bacteria that decompose fatty acids. III. Characterization of the filamentous gliding Desulfonema limicola gen. nov. sp. nov., and Desulfonema magnum sp. nov. Arch Microbiol 134: 286–294
Yokoyama MT, Carlson JR (1981) Production of skatole and paracresol by a rumen Lactobacillus sp. Appl Environ Microbiol 41: 71–76
Zenk MH (1971) Metabolism of prearomatic and aromatic compounds in plants. In: Wagner H, Hörhammer L (eds) Pharmacognosy and phytochemistry. Springer, Berlin Heidelberg New York, pp 314–346
Zenk MH, Ulbrich B, Brusse J, Stöckigt J (1980) Procedure for the enzymatic synthesis and isolation of cinnamoyl-CoA thioesters using a bacterial system. Anal Biochem 101: 182–187
Zhao Y, Kung SD, Dube SK (1990) Nucleotide sequence of rice 4-coumarate: CoA ligase gene, 4-CL. 1. Nucleic Acids Res 18: 6144
Ziegler K, Buder R, Winter J, Fuchs G (1989) Activation of aromatic acids and aerobic 2-aminobenzoate metabolism in a denitrifying Pseudomonas strain. Arch Microbiol 151: 171–176
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mohamed, M.ES., Fuchs, G. Purification and characterization of phenylacetate-coenzyme A ligase from a denitrifying Pseudomonas sp., an enzyme involved in the anaerobic degradation of phenylacetate. Arch. Microbiol. 159, 554–562 (1993). https://doi.org/10.1007/BF00249035
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00249035