Abstract
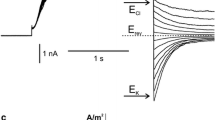
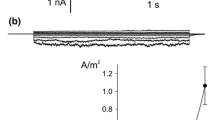
Single channel performance and deactivation currents have been analyzed in the presence of cation channel blockers to reveal pharmacological properties of the slow-activating (SV) cation-selective ion channel in the vacuolar membrane (tonoplast) isolated from suspension cells of Chenopodium rubrum L. At a holding potential of −100 mV, the SV channel showed half-maximal inhibition with 20mm tetraethylammonium (TEA), 7 μm 9amino-acridine, 6 μm (+)-tubocurarine, 300nm quinacrine, and 35 μm quinine, respectively. The SV channel is also blocked by charybdotoxin (20nm at −80 mV) but not by apamine. 9-Amino-acridine, (+)-tubocurarine and quinacrine act in a voltage-dependent fashion, binding to the open channel and to different sites along the transmembrane voltage profile according to Woodhull (J. Gen. Physiol. 61:687–708, 1973). No binding site could be specified for charybdotoxin, which binds to the closed channel, and for quinine. Except for quinine, all tested blockers were effective only if added to the cytoplasmic side of the tonoplast. A structural relationship between the SV channel and Maxi-K channels in animal systems is inferred.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adams, P.R., Feltz, A. 1980a. Quinacrine (mepacrine) action at frog end plate. J. Physiol. 306:261–281
Adams, P.R., Feltz, A. 1980b. End-plate channel opening and the kinetics of quinacrine (mepacrine) block. J. Physiol. 306: 283–306
Anderson, C.S., Mackinnon, R., Smith, C., Miller, C. 1988. Charybdotoxin block of single Ca2+-activated K+ channels effects of channel gating, voltage, and ionic strength. J. Gen. Physiol. 91:317–333
Armstrong, C.M. 1966. Time course of TEA+-induced anomalous rectification in squid giant axons. J. Gen. Physiol. 50:491–503
Barrett, J.N., Magleby, K.L., Pallotta, B.S. 1982. Properties of single calcium-activated potassium channels in cultered rat muscle J. Physiol. 331:211–230
Benham, D., Bolton, T.B. 1983. Patch-clamp studies of slow potential-sensitive potassium channels in longitudinal smooth muscle cells of rabbit jejunum. J. Physiol. 340:469–486
Benham C.D., Bolton, T.B., Lang, R.J., Takewaki, T. 1985. The mechanism of action of Ba2+ and TEA on single Ca2+activated K+-channels in arterial and intestinal smooth muscle cell membranes. Pfluegers Arch. 403:120–127
Bentrup, F.-W., Gogarten-Boekels, M., Hoffmann, B., Gogarten, J.P., Baumann, C. 1986. ATP-dependent acidification and tonoplast hyperpolarization in isolated vacuoles from green suspension cells of Chenopodium rubrum L. Proc. Natl. Acad. Sci. USA 83:2431–2433
Bleich, M., Schlatter, E., Greger, R. 1990. The luminal K+ channel of the thick ascending Henle's loop. Pfluegers Arch. 415:449–460
Castle, N.A., Haylett, D.G., Jenkinson, D.H. 1989. Toxins in the characterisation of potassium channels TINS 12, No. 2:59–65 cells. Biochim. Biophys. Acta 1029: 33—42
Chinn, K., Narahashi, T.1986. Stabilization of sodium channel states by deltamethrin in mouse neuroblastoma cells. J. Physiol. 380:191–207
Colombo, R., Cerrana, R., Lado, P., Peres, A. 1988. Voltagedependent channels permeable to K+ and Na+ in the membrane of Acer pseudoplatanus vacuoles. J. Membr. Biol. 103:227–236.
Cook, N.S. 1988. The pharmacology of potassium channels and their therapeutic potential. TIPS 9:21–28
Cook, N.S., Haylett, D.G. 1985. Effects of apamine, quinine, and neuromuscular blockers on calcium-activated potassium channels in guinea-pig hepatocytes J. Physiol. 358:373–394
Dreyer, F. 1990. Peptide toxins and potassium channels. Rev. Physiol. Biochem. Pharmacol. 115:93–136
Fernandez-Belda, F., Soler, F., Gomez-Fernandez, J.C. 1989. Quinacrine inhibits the calcium-induced calcium release in heavy sarcoplasmic reticulum vesicles. Biochim. Biophys. Acta 985:279–285
Geletyuk, V.I., Kazachenko, V.N. 1989. Single potential-dependent K+ channels and their oligomers in molluscan glial cells. Biochim. Biophys. Acta 981:343–350
Glavinovic, M.I., Trifaro, J.M. 1988. Quinine blockade of currents through Ca2+activated K+ channels in bovine chromaffine cells. J. Physiol. 399:139–152
Gögelein, H., Capek, K. 1990. Quinine inhibits chloride and nonselective cation channels in isolated rat distal colon cells. Biochim. Biophys. Acta 1027:191–198
Gögelein, H., Greger, R., Schlatter, E. 1987. Potassium channels in the basolateral membrane of the rectal gland of Squalus acanthias. Pfluegers Arch. 409:107–113
Gray, M.A., Argent, B.E. 1990. Non-selective cation channel on pancreatic duct cells. Biochim. Biophys. Acta 1029:33–42
Guggino, S.E., Guggino, W.B., Green, N., Sacktor, B. 1987. Blocking agents of Ca2+-activated K+ channels in cultured medullary thick ascending limb cells. Am. J. Physiol. 252:C128-C137.
Hedrich, R., Barbier-Brygoo, H., Felle, H., Flügge, U.I., Lüttge, U., Maathuis, F.J.M., Mar, S., Prins, H.B.A., Raschke, K., Schnabl, H., Schröder, J., Struve, I., Taiz, L., Ziegler, P. 1988. General mechanisms for solute transport across the tonoplast of plant vacuoles: A patch-clamp survey of ion channels and proton pumps. Botanica Acta 101:7–13
Hedrich, R., Flügge, U.I., Fernandez, J.M. 1986. Patch-clamp studies of ion transport in isolated plant vacuoles. FEBS Lett. 204:228–232
Hedrich, R., Kurkdjian, A. 1988. Characterization of an anionpermeable channel from sugar beet vacuoles: Effect of inhibitors. EMBO J. 7:3661–3666
Hedrich, R., Neher, E. 1987. Cytoplasmic calcium regulates voltage dependent ion channels in plant vacuoles. Nature 329:833–835
Hille, B. 1984. Ionic Channels of Excitable Membranes. Sinauer Associates, Sunderland, MA
Ketchum, K.A., Poole, R.J. 1991. Cytosolic calcium regulates a potassium current in corn (Zea mays). J. Membrane Biol. 119:277–288
Latorre, R. 1986. The large calcium-activated potassium channel. In: Ion Channel Reconstitution. C. Miller, editor, pp. 431–468. Plenum, New York
Laver, D. R. 1992. Divalent cation block and competition between divalent and monovalent cations in the large-conductance K+ channel from Chara australis. J. Gen. Physiol. 100:269–300.
Lucchesi, K., Ravindran, A., Young, H., Moczydlowski, E. 1989. Analysis of the blocking activity of charybdotoxin homologs and iodonated derivatives against Ca2+-activated K+ channels. J. Membrane Biol. 109:269–281
Ma, J., Coronado, R. 1988. Heterogeneity of conductance states in calcium channels of skeletal muscles. Biophys. J. 53:387–395
Matsuda, H. 1988. Open-state substructure of inwardly rectifying potassium channels revealed by magnesium block in guineapig heart cells. J. Physiol. 397:237–258
Moczydlowski, E., Lucchesi, K., Ravindran, A. 1988. An emerging pharmacology of peptide toxins targeted against potassium channels. J. Membrane Biol. 105:95–111
Rousseau, E., Smith, J.S., Meissner, G. 1987. Ryanodine modifies conductance and gating behavior of single Ca2+ release channels. Am. J. Physiol. 253:C364-C368
Sakmann, B., Trube, G. 1984. Conductance properties of single inwardly rectifying potassium channels in ventricular cells from guinea-pig heart. J. Physiol. 347:641–657
Schlichter, L.C. 1992. Nonselective cation channels in intact human T lymphocytes. Can. J. Physiol. Pharmacol. 70: 247–258
Schroeder J.I. 1989. Quantitative analysis of outward rectifying K+ channel currents in guard cell protoplasts from Vicia faba. J. Membrane Biol. 107:229–236
Smart, T.G. 1987. Single calcium-activated potassium channels recorded from cultured rat sympathetic neurones. J. Physiol. 389:337–360
Stockbridge, L.L., French, A.S., Paulman, S.F. 1991. Subconductance states in calcium-activated potassium channels from canine airway smooth muscle. Biochim. Biophys. Acta 1064:212–218
Takeda, K., Trautmann, A. 1984. A patch-clamp study of the partial agonist actions of tubocurarine on rat myotubes. J. Physiol. 349:353–374
Tester, M. 1988. Pharmacology of K+ channels in the plasmalemma of the green alga Chara corallina. J. Membrane Biol. 103:159–169
Tsai, M.C., Oliveira, A.C., Albuquerque, E., Eldefravi, M.E., Eldefravi, A.T. 1979. Mode of action of quinacrine on the acetylcholine receptor ionic channel complex. Mol. Pharmacol. 16:382–392
Tyerman, S.D., Terry, B.R., Findlay, G.P. 1992. Multiple conductances in the large K+ channel from Chara corallina shown by a transient analysis method. Biophys. J. 61: 736–749
van Duijn, B., Ypey, D.L., Libbenga, K.R. 1993. Whole-cell K+ currents across the plasma membrane of tobacco protoplasts from cell-suspension cultures. Plant Physiol. 101: 81–88
Vergara, C., Moczydlowski, E., Latorre, R. 1984. Conduction, blockade and gating in a Ca2+ activated, K+ channel incorporated into planar lipid bilayers. Biophys. J. 45:73–76
Villaroel, A., Alvarez, O., Oberhauser, A., Latorre, R. 1988. Probing a Ca2+-activated K+ channel with quaternary ammonium ions. Pfluegers Arch. 413:118–126
Weik, R., Lönnendonker, U., Neumcke, B. 1989. Low-conductance states of K+ channels in adult mouse skeletal muscle. Biochim. Biophys. Acta 983:127–134
Weiser, T., Bentrup, F.-W. 1990. (+)-Tubocurarine is a potent inhibitor of cation channels in the vacuolar membrane of Chenopodium rubrum L. FEBS Lett. 227:220–222
Weiser, T., Bentrup, F.-W. 1991. Charybdotoxin blocks cation channels in the vacuolar membrane of suspension cells of Chenopodium rubrum L. Biochim. Biophys. Acta 1066: 109–110
Weiser, T., Blum, W., Bentrup, F.-W. 1991. Calmodulin regulates the Ca2+-dependent slow-vacuolar ion channel in the tonoplast of Chenopodium rubrum suspension cells. Planta 185:440–442
Woodhull, A.M. 1973. Ionic blockage of sodium channels in nerve. J. Gen. Physiol. 61:687–708
Yamamoto, D., Yen, J.Z. 1984. Kinetics of 9-amino-acridine block of single Na channels. J. Gen. Physiol. 84:361–377
Author information
Authors and Affiliations
Additional information
We are grateful to Prof. F. Dreyer and Dr. J. Beise from the Pharmacology Department of the Justus-Liebig-Universität Giessen for continuous interest and helpful suggestions. This work was supported by a grant from the Deutsche Forschungsgemeinschaft (Be 466/21-5) and the Bundesminister für Forschung und Technologie, Bonn.
Rights and permissions
About this article
Cite this article
Weiser, T., Bentrup, FW. Pharmacology of the SV channel in the vacuolar membrane of chenopodium rubrum suspension cells. J. Membarin Biol. 136, 43–54 (1993). https://doi.org/10.1007/BF00241488
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00241488