Summary
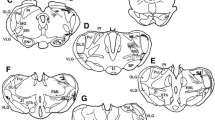
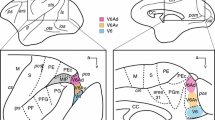
In 39 squirrel monkeys (Saimiri sciureus), the effects of various brain lesions on vocalizations elicited from the precallosal cingulate gyrus were tested. It was found that lesions abolishing the “cingular vocalization” completely can be traced from the stimulation site continuously down to the laryngeal motoneurons in the nucleus ambiguus. The pathway thus determined (Fig. 4) travels from the precallosal cingulate gyrus through the frontal white matter and enters the internal capsule from a dorsolateral position. The pathway then follows this structure in a medio-caudal direction down to the caudal diencephalon. Here, the effective lesions leave the corticospinal tract and ascend dorsally into the periaqueductal grey. The pathway follows this structure to its end where it sweeps lateral through the parabrachial area and then descends through the lateral pons and ventrolateral medulla to the nucleus ambiguus.
In nine of the animals, in addition, the effects of bilateral anterior cingular lesions on vocalizations elicited in other brain areas were tested. It was found that the only vocalization-eliciting area which becomes ineffective after destruction of the anterior cingulate gyrus is the postero-medial orbital cortex.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- a:
-
nucl. accumbens
- aa:
-
area anterior amygdalae
- ab:
-
nucl. basalis amygdalae
- ac:
-
nucl. centralis amygdalae
- al:
-
nucl. lateralis amygdalae
- am:
-
nucl. medialis amygdalae
- an:
-
nucl. anterior thalami
- aq:
-
griseum centralis
- bc:
-
brachium conjunctivum
- ca:
-
caudatum
- cb:
-
cerebellum
- cc:
-
corpus callosum
- cen:
-
nucl. centralis superior Bechterew
- ci:
-
capsula interna
- cin:
-
cingulum
- cl:
-
claustrum
- coa:
-
commissura anterior
- coli:
-
colliculus inferior
- cols:
-
colliculus superior
- cop:
-
commissura posterior
- cr:
-
corpus restiforme
- csp:
-
tractus corticospinalis
- db:
-
fasciculus diagonalis Brocae
- dbc:
-
decussatio brachii conjunctivi
- f:
-
fornix
- gc:
-
gyrus cinguli
- gl:
-
geniculatum laterale
- gm:
-
geniculatum mediale
- gp:
-
globus pallidus
- gr:
-
gyrus rectus
- gs:
-
gyrus subcallosus
- h:
-
area tegmentalis (Forel)
- ha:
-
habenula
- hi:
-
tractus habenulo-interpeduncularis
- hip:
-
hippocampus
- hya:
-
hypothalamus anterior
- hyv:
-
hypothalamus ventromedialis
- in:
-
nucl. interpeduncularis
- lap:
-
nucl. lateralis posterior thalami
- lem:
-
lemniscus medialis
- lm:
-
fasciculus longitudinalis medialis
- m:
-
nucl. mammillaris
- md:
-
nucl. medialis dorsalis thalami
- mt:
-
tractus mammillothalamicus
- nst:
-
nucl. striae terminalis
- nts:
-
nucl. solitarius
- oi:
-
oliva inferior
- ol:
-
fasciculus olfactorius (Zuckerkandl)
- os:
-
oliva superior
- p:
-
pedunculus cerebri
- pmc:
-
brachium pontis
- po:
-
griseum pontis
- pro:
-
area praeoptica
- pu:
-
nucl. pulvinaris
- put:
-
putamen
- re:
-
formatio reticularis mesencephali
- rep:
-
nucl. reticularis tegmenti pontis
- rl:
-
nucl. reticularis lateralis
- rub:
-
nucl. ruber
- s:
-
septum
- sm:
-
stria medullaris
- sn:
-
substantia nigra
- st:
-
stria terminalis
- sto:
-
stria olfactoria lateralis
- tec:
-
tractus tegmentalis centralis
- trz:
-
corpus trapezoideum
- va:
-
nucl. ventralis anterior thalami
- ves:
-
nucl. vestibularis
- vpl:
-
nucl. ventralis posterior lateralis th.
- vpm:
-
nucl. ventralis posterior medialis th.
- zi:
-
zona incerta
- II:
-
tractus opticus
- IIchde:
-
chiasma n. opticorum
- III:
-
nucl. n. oculomotorii and n. oculomotorius
- IV:
-
nucl. n. trochlearis
- VI:
-
n. abducens
- VII:
-
nucl. n. facialis and n. facialis
- VIII:
-
n. acusticus
- XII:
-
nucl. n. hypoglossi
References
Adametz, J., O'Leary, J.L.: Experimental mutism resulting from periaqueductal lesions in cats. Neurology (Minneap.) 9, 636–642 (1959)
Barris, R.W., Schuman, H.R.: Bilateral anterior cingulate gyrus lesions. Neurology (Minneap.) 3, 44–52 (1953)
Botez, M.J., Barbeau, A.: Role of subcortical structures and particularly of the thalamus in mechanisms of speech and language. Int. J. Neurol. 8, 300–320 (1971)
Brickner, R.M.: A human cortical area producing repetitive phenomena when stimulated. J. Neurophysiol. 3, 128–130 (1940)
Buge, A., Escourolle, R., Rancurel, G., Poisson, M.: Mutisme akinétique et ramollissement bicingulaire. Trois observations anatomo-cliniques. Rev. Neurol. (Paris) 131, 121–137 (1975)
De Molina, A.F., Hunsperger, R.W.: Organization of the subcortical system governing defence and flight reactions in the cat. J. Physiol. (Lond.) 160, 200–213 (1962)
Dua, S., MacLean, P.D.: Localization for penile erection in medial frontal lobe. Am. J. Physiol. 207, 1425–1434 (1964)
Hassler, R.: Spezifische und unspezifische Systeme des menschlichen Zwischenhirns. In: Bargmann W. and J.P. Schade J.P. (Eds.): Lectures on the Diencephalon. Progress in Brain Res., Vol. 5, pp. 1–32. Amsterdam: Elsevier 1964
Hughes, J.R., Mazurowski, J.A.: Studies on the supracallosal mesial cortex of unanesthetized, conscious mammals. II. Monkey. A. Movements elicited by electrical stimulation. Electroenceph. Clin. Neurophysiol. 14, 477–485 (1962)
Jürgens, U.: Projections from the cortical larynx area in the squirrel monkey. Exp. Brain Res. 25, 401–411 (1976a)
Jürgens, U.: Reinforcing concomitants of electrically-elicited vocalizations. Exp. Brain Res. 26, 203–214 (1976b)
Jürgens, U., Müller-Preuss, P.: Convergent projections of different limbic vocalization areas in the squirrel monkey. Exp. Brain Res. 29, 75–83 (1977)
Jürgens, U., Pratt, R.: Role of the periaqueductal grey in vocal expression of emotion. Brain Res. (in press)
Kaada, B.R.: Somato-motor, autonomic and electrocorticographic responses to electrical stimulation of “rhinencephalic” and other structures in primates, cat and dog. Acta Physiol. Scand. Suppl. 83, 1–285 (1951)
Kelly, A.H., Beaton, I.E., Magoun, H.W.: A midbrain mechanism for facio-vocal activity. J. Neurophysiol. 9, 181–189 (1946)
Müller-Preuss, P., Jürgens, U.: Projections from the “cingular” vocalization area in the squirrel monkey. Brain Res. 103, 29–43 (1976)
Penfield, W., Welch, K.: The supplementary motor area of the cerebral cortex. Arch. Neurol. Psychiat. (Chicago) 66, 289–317 (1951)
Randall, W.L.: The behavior of cats (Felis catus) with lesions in the caudal midbrain region. Behavior 23, 107–139 (1964)
Robinson, B.W.: Vocalization evoked from forebrain in Macaca mulatta. Physiol. Behav. 2, 245–254 (1967)
Rubens, A.B.: Aphasia with infarction in the territory of the anterior cerebral artery. Cortex 11, 239–250 (1975)
Showers, M.J.C., Crosby, E.C.: Somatic and visceral responses from the cingulate gyrus. Neurology (Minneap.) 8, 561–565 (1958)
Skultety, P.M.: The behavioral effects of destructive lesions of the periaqueductal grey matter in adult cats. J. Comp. Neurol. 110, 337–365 (1958)
Smith, W.K.: The functional significance of the rostral cingular cortex as revealed by its responses to electrical excitation. J. Neurophysiol. 8, 241–255 (1945)
Sutton, D., Larson, C., Lindeman, R.C.: Neocortical and limbic lesion effects on primate phonation. Brain Res. 71, 61–75 (1974)
Sutton, D., Samson, H.H., Larson, C.R.: Brain mechanisms in learned phonation of Macaca mulatta. In: Chivers, D.J. and Herbert, J. (Eds.): Recent Advances in Primatology, pp. 769–784. New York: Academic Press 1978
Talairach, J., Bancaud, J., Geier, S., Bordas-Ferrer, M., Bonis, A., Szikla, G., Rusu, M.: The cingulate gyrus and human behavior. Electroenceph. Clin. Neurophysiol. 34, 45–52 (1973)
Winter, P., Ploog, D., Latta, J.: Vocal repertoire of the squirrel monkey, its analysis and significance. Exp. Brain Res. 1, 359–384 (1966)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jürgens, U., Pratt, R. The cingular vocalization pathway in the squirrel monkey. Exp Brain Res 34, 499–510 (1979). https://doi.org/10.1007/BF00239145
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00239145