Summary
-
1.
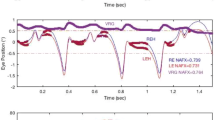
Optokinetic nystagmus (OKN), optokinetic after-nystagmus (OKAN), vestibular nystagmus and visual-vestibular interactions were studied in monkeys after surgical ablation of the flocculus and paraflocculus. After bilateral flocculectomy the initial rapid rise in slow phase eye velocity of horizontal and vertical OKN was severely attenuated, and maximum velocities fell to the preoperative saturation level of OKAN. There is generally little or no upward OKAN in the normal monkey, and upward OKN was lost after bilateral lesions. Unilateral flocculectomy affected the rapid rise in horizontal velocity to both sides.
-
2.
Consistent with the absence of a rapid response to steps of surround velocity, animals were unable to follow acceleration of the visual field with eye accelerations faster than about 3–57 °/s2.
-
3.
The slow rise in OKN slow phase velocity to a steady state level was prolonged after operation. However, rates of rise were approximately equal for the same initial retinal slips before and after operation. The similarity in the time course of OKN when adjusted for initial retinal slip, and in the gain, saturation level and time course of OKAN before and after flocculectomy indicates that the lesions had not significantly altered the coupling of the visual system to the velocity storage integrator or its associated time constant.
-
4.
When animals were rotated in a subjectstationary visual surround after flocculectomy, they could not suppress the initial jump in eye velocity at the onset of the step. Despite this, they could readily suppress the subsequent nystagmus. The time constant of decline in the conflict situations was almost as short as in the normal monkey and was in the range of the peripheral vestibular time constant. This suggests that although the animals were unable to suppress rapid changes in eye velocity due to activation of direct vestibulo-oculomotor pathways, they had retained their ability to discharge activity from the velocity storage mechanism. Consistent with this, animals had no difficulty in suppressing OKAN after flocculectomy.
-
5.
Visual-vestibular interactions utilizing the velocity storage mechanism were normal after flocculectomy, as was nystagmus induced by rotation about a vertical axis or about axes tilted from the vertical. Also unaffected were the discharge of nystagmus caused by tilting the head out of the plane of the response and visual suppression of nystagmus induced by off-vertical axis rotation. The flocculus does not appear to play an important role in mediating these responses.
-
6.
The data before and after flocculectomy were simulated by a model which is homeomorphic to that presented previously. The model has direct pathways from the vestibular and visual systems. The visual and vestibular systems couple to a common velocity storage integrator. There is also a dump mechanism which shortens the time constant of the integrator when eye velocity exceeds surround velocity. An important element in the model is a nonlinearity that couples the visual system to the integrator. Approximate closed form relationships were established between the parameters of the nonlinearity and the dynamics of OKN and OKAN. The nonlinear element is assumed to receive a central representation of retinal slip as its input. Its output drives the velocity storage integrator. The model predicts the normal responses, and by removal of the direct pathway it simulates the data after flocculectomy. The nonlinearity explains why the storage integrator charges more slowly for larger initial retinal slips both in the normal animal and after flocculectomy. The model also predicts that surround velocity would be followed better during deceleration than acceleration. This is a result of activation of the dump mechanism which shortens the time constant of the velocity storage integrator, effectively discharging it. Activation of the dump mechanism is independent of the flocculus.
-
7.
Although the flocculus is closely linked to the vestibular system, its function in the monkey appears closely related to production of rapid changes in eye velocity from the visual system, either during slow phases of nystagmus or during ocular pursuit (Zee et al. 1981). It does not appear to cancel the horizontal VOR at the level of the vestibular nuclei, nor does it directly affect the dynamics of vestibular nystagmus, OKAN or the velocity storage mechanism.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baloh RW, Konrad HR, Honrubia V (1975) Vestibulo-ocular function in patients with cerebellar atrophy. Neurology (NY) 25: 160–168
Barlow HB, Hill RM, Levick WR (1964) Retinal ganglion cells responding selectively to direction and speed of image motion in the rabbit. J Physiol (Lond) 173: 377–407
Bond HW, Ho P (1970) Solid miniature silver-silver chloride electrodes for chronic implantation. Electroencephalogr Clin Neurophysiol 28: 206–208
Buizza A, Schmid R (1982) Visual-vestibular interaction in the control of eye movement: Mathematical modelling and computer simulation. Biol Cybern 43: 209–223
Collewijn H (1969) Optokinetic eye movements in the rabbit: Input-output relations. Vision Res 9: 117–132
Collewijn H (1972) An analog model of the rabbits. Brain Res 36: 71–88
Collewijn H (1975) Direction-selective units in the rabbit's nucleus of the optic tract. Brain Res 100: 489–508
Cohen B, Matsuo V, Raphan T (1977) Quantitative analysis of the velocity characteristics of optokinetic nystagmus and optokinetic after-nystagmus. J Physiol (Lond) 270: 321–344
Cohen B, Henn V, Raphan T, Dennett D (1981) Velocity storage, nystagmus, and visual-vestibular interactions in humans. In: Cohen B (ed) Vestibular and oculomotor physiology. Ann NY Acad Sci 374: 421–433
Demer JL (1981) The variable gain element of the vestibulo-ocular reflex is common to the optokinetic system of the cat. Brain Res 229: 1–13
Dichgans J, von Reutern GM, Rommelt U (1978) Impaired suppression of vestibular nystagmus by fixation in cerebellar and non-cerebellar patients. Arch Psychiatr Nervenkr 266: 183–199
Eccles JC, Ito M, Szentágothai J (1967) The cerebellum as a neuronal machine. Springer, Berlin Heidelberg New York
Goldberg JM, Fernandez C (1971) Physiology of peripheral neurons innervating semicircular canals of the squirrel monkey. I. Resting discharge and response to constant angular accelerations. J Neurophysiol 34: 635–660
Guedry FE (1965) Orientation of the rotation axis relative to gravity: Its influence on nystagmus and the sensation of rotation. Acta Otolaryngol (Stockh) 60: 30–48
Hassul M, Daniels PD, Kimm J (1976) Effects of flocculectomy on the vestibulo-ocular reflex in the chinchilla. Brain Res 118: 339–343
Henn V, Cohen B, Young LR (1980) Visual-vestibular interaction in motion perception and the generation of nystagmus. Neurosci Res Program Bull 18: 459–651
Hoffman KP (1981) Neuronal responses related to optokinetic nystagmus in the cat's nucleus of the optic tract. In: Fuchs A, Becker W (eds) Progress in oculomotor research. Elsevier/ North Holland, Amsterdam, pp 443–454
Hoffman KP, Schoppmann A (1981) A quantitative analysis of the direction-specific response of neurons in the cat's nucleus of the optic tract. Exp Brain Res 42: 146–157
Ito M, Jastreboff PJ, Miyashita T (1982) Specific effects of unilateral lesions in the flocculus upon eye movements in albino rabbits. Exp Brain Res 45: 233–242
Koenig E, Dichgans J (1981) Aftereffects of vestibular and optokinetic stimulation and their interaction. In: Cohen B (ed) Vestibular and oculomotor physiology. Ann NY Acad Sci 374: 434–445
Keller EL, Precht W (1979) Visual-vestibular responses in vestibular nuclear neurons in intact and cerebellectomized alert cats. Neuroscience 4: 1599–1613
Keller EL, Crandall WF (1981) Neural activity in the nucleus reticularis tegmenti pontis in the monkey related to eye movements and visual stimulation. In: Cohen B (ed) Vestibular and oculomotor physiology. Ann NY Acad Sci 374: 249–261
Lisberger SG, Fuchs AF (1978) Role of primate flocculus during rapid behavioral modification of vestibulo-ocular reflex. I. Purkinje cell activity during visually guided horizontal smooth-pursuit eye movements and passive head rotation. J Neurophysiol 41: 764–777
Marr D (1969) A theory of cerebellar cortex. J Physiol (Lond) 202: 437–470
Matsuo V, Cohen B, Raphan T, de Jong V, Henn V (1979) Asymmetric velocity storage for upward and downward nystagmus. Brain Res 176: 159–164
Miles FA, Fuller JH (1975) Visual tracking and the primate flocculus. Science 189: 1000–1002
Miles FA, Eighmy BB (1980) Long term adaptive changes in primate vestibuloocular reflex. I. Behavioral observations. J Neurophysiol 43: 1406–1425
Miles FA, Braitman DJ, Dow BM (1980) Long term adaptive changes in primate vestibulo-ocular reflex. III. Electrophysiological observations in flocculus of adapted monkeys. J Neurophysiol 43: 1477–1493
Noda H (1981) Visual mossy fiber inputs to the flocculus of the monkey. In: Cohen B (ed) Vestibular and oculomotor physiology. Ann NY Acad Sci 374: 465–475
Noda H, Warabi T (1982) Eye position signals in the flocculus of the monkey during smooth-pursuit eye movements. J Physiol (Lond) 324: 187–202
Oyster CW, Takahashi E, Collewijn H (1972) Direction-selective retinal ganglion cells and control of optokinetic nystagmus in the rabbit. Vision Res 12: 183–193
Precht W (1981) Visual-vestibular interaction in vestibular neurons: Functional pathway organization. In: Cohen B (ed) Vestibular and oculomotor physiology. Ann NY Acad Sci 374: 230–248
Raphan T, Cohen B, Matsuo V (1977) A velocity-storage mechanism responsible for optokinetic nystagmus (OKN), optokinetic after-nystagmus (OKAN) and vestibular nystagmus. In: Baker R, Berthoz A (eds) Control of gaze by brain stem neurons. Elsevier/North Holland, Amsterdam, pp 37–47
Raphan T, Matsuo V, Cohen B (1979) Velocity storage in the vestibulo-ocular reflex arc (VOR). Exp Brain Res 35: 229–248
Raphan T, Cohen B (1981) The role of integration in oculomotor control. In: Zuber B (ed) Models of oculomotor behavior and control. CRC Press Inc, West Palm Beach, Fla, pp 91–109
Raphan T, Cohen B, Henn V (1981) Effects of gravity on rotatory nystagmus in monkeys. In: Cohen B (ed) Vestibular and oculomotor physiology. Ann NY Acad Sci 374: 44–55
Robinson DA (1977) Vestibular and optokinetic symbiosis: An example of explaining by modelling. In: Baker R, Berthoz A (eds) Control of gaze by brain stem neurons. Elsevier/North Holland, Amsterdam, pp 49–58
Robinson DA (1980) In: Henn V, Cohen B, Young LR (eds) Visual-vestibular interaction in motion perception and the generation of nystagmus. Neuroscience Res Program Bull 18: 582–589
Simpson JI, Alley KE (1974) Visual climbing fiber input to rabbit vestibulo-cerebellum: A source of direction-specific information. Brain Res 82: 302–308
Simpson JI, Soodak RE, Hess R (1979) The accessory optic system and its relation to the vestibulo cerebellum. Prog Brain Res 50: 715–724
Skavenski AA, Robinson DA (1973) Role of abducens motoneurons in the vestibulo-ocular reflex. J Neurophysiol 36: 724–738
Takemori S, Cohen B (1974) Loss of suppression of vestibular nystagmus after flocculus lesion. Brain Res 72: 213–224
Waespe W, Henn V (1977a) Neuronal activity in the vestibular nuclei of the alert monkey during vestibular and optokinetic stimulation. Exp Brain Res 27: 523–538
Waespe W, Henn V (1977b) Vestibular nuclei activity during optokinetic after nystagmus (OKAN) in the alert monkey. Exp Brain Res 30: 323–330
Waespe W, Henn V (1978) Conflicting visual vestibular stimulation and vestibular nucleus activity in alert monkeys. Exp Brain Res 33: 203–211
Waespe W, Henn V (1979) Motion information in the vestibular nuclei of alert monkeys: Visual and vestibular vs. optomotor output. Prog Brain Res 50: 683–693
Waespe W, Henn V (1981) Visual-vestibular interaction in the flocculus of the alert monkey. II. Purkinje cell activity. Exp. Brain Res 43: 349–360
Waespe W, Cohen B (1983) Effects of flocculectomy on unit activity in the vestibular nuclei during visual-vestibular interactions. Exp Brain Res (in press)
Waespe W, Büttner U, Henn V (1981) Visual-vestibular interaction in the flocculus of the alert monkey. I. Input activity. Exp Brain Res 43: 337–348
Walberg F, Bowsher D, Brodal A (1958) The termination of primary vestibular fibers in the vestibular nuclei of the cat. An experimental study with silver methods. J Comp Neurol 110: 391–419
Young LR (1971) Pursuit eye tracking movements. In: Bach-y-Rita P, Collins CC, Hyde JE (eds) The control of eye movements. Academic Press, New York, pp 429–443
Young LR, Henn V (1975) Nystagmus induced by pitch and yaw rotation of monkeys. Fortschr Zool 23: 235–246
Zee DS, Yamazaki A, Butler PH, Gucer G (1981) Effects of ablation of flocculus and paraflocculus on eye movements in primate. J Neurophysiol 46: 878–899
Author information
Authors and Affiliations
Additional information
Supported by NIH Research Grant NS-00294, Core Center Grant EY 02296, and a grant from the Young Mens's Philanthropic League
Walter Waespe had a Fogarty Fellowship (FO TW 02768) and a grant from the Swiss National Foundation
Theodore Raphan was supported by a National Eye Institute Academic Investigator Award (EY 00157)
Rights and permissions
About this article
Cite this article
Waespe, W., Cohen, B. & Raphan, T. Role of the flocculus and paraflocculus in optokinetic nystagmus and visual-vestibular interactions: Effects of lesions. Exp Brain Res 50, 9–33 (1983). https://doi.org/10.1007/BF00238229
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00238229