Summary
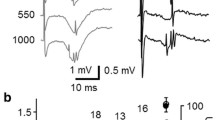
The nature of spontaneous unitary activity of rat neocortex was investigated during slow wave sleep and urethane anaesthesia. Neurones in layer IV and V locations fired in a burst-pause pattern at a low burst repetition rate (0.5–4 per second) during both stage 3/4 sleep and urethane anaesthesia. Occasionally an alternative mode of firing (spindle clusters), associated with focal spindle wave activity, was also found to occur in both states. Using dual microelectrode implants it was found that the onset times of bursts (but not spindle clusters), coincided in the same and opposing cortices, whether in functionally similar or disparate areas. The highest probability was that burst onsets occurred simultaneously (resolution =2.56 ms, interquartile range=40 ms). Spontaneous unitary activity was investigated in the thalamus for temporal correlation with spontaneous unitary activity in neocortex under urethane anaesthesia. Neurones of the anterior intralaminar group (aIL) consistently fired in a burst-pause pattern such that each aIL burst showed a strong tendency to precede a cortical burst. Unilateral electrical stimulation of the aIL nuclei evoked widespread bilateral entrainment of cortical bursts. In contrast stimulation of VP1, or cutaneous sites, evoked only short duration spike responses together with burst abolition in the appropriate restricted Sm1 area. Ionophoresis of NMDA (N-Methyl D-Aspartate) onto Sm1 neurones increased the probability of cortical burst responses to aIL stimulation in addition to decreasing the latency by 20–40 ms (n=11). lonophoresis of 2APV (2-amino 5-phosphono valeric acid) caused simultaneous abolition of spontaneous cortical bursts and bursts evoked by aIL stimulation. Short latency responses to cutaneous and VP1 stimulation were unaffected by ionophoresis of 2APV sufficient to cause burst elimination, suggesting that this pathway does not operate via a 2APV sensitive receptor mechanism. Anatomical features of the aIL nuclei and their overall cortical projection pattern are discussed in relationship to these findings. The activation of cortical NMDA/APV sensitive receptors by aIL afferents in the “spontaneous” generation of bursts in cortical cells is discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Akert K, Koella WP, Hess Jr R (1952) Sleep produced by electrical stimulation of the thalamus. Am J Physiol 168: 260–267
Andersen P, Andersson SA, Lomo T (1967) Some factors involved in the thalamic control of spontaneous barbiturate spindles. J Physiol 192: 257–282
Armstrong-James M, Fox K, Millar J (1980) A method for etching the tips of carbon fibre microelectrodes J Neurosci Meth 2: 431–432
Armstrong-James M, Fox K (1983) Effects of ionophoresed noradrenaline on the spontaneous activity of neurones in rat primary somatosensory cortex. J Physiol 335: 427–447
Armstrong-James M, Fox K (1984) Similarities in unitary cortical activity between SWS and light urethane anaesthesia in the rat. J Physiol 346: 55P
Armstrong-James M, Caan AW, Fox K (1985) Threshold effects of ionophoresed N-methyl D-aspartate (NMDA) and 2-amino 5-phosphono valeric acid (2APV) on the spontaneous activity of neocortical single neurones in the urethane anaesthetised rat. Exp Brain Res 60: 209–213
Baker MA (1971) Spontaneous and evoked activity of neurones in the somatosensory thalamus of the waking cat. J Physiol 217: 359–380
Bentivoglio M, Molinari M, Mineiackhi D, Macchi G (1983) Organisation of the cortical projections of the posterior complex and intralaminar nuclei of the thalamus as studied by means of retrograde tracers. In: A Macchi, Rustioni A, Spreafico R (eds) Somatosensory integration in the thalamus. Elsevier Science Publishers, Amsterdam
Calvet J, Fourment A, Thieffry M (1973) Electrical activity in neocortical projection and association areas during SWS. Brain Res 52: 173–187
Creutzfeldt OD, Jung R (1961) Neuronal discharges in the cat's motor cortex during sleep and arousal. In: Wolstenholme GEW, O'Connor M (ed) Ciba Foundation symposium on the nature of sleep. Churchill, London, pp 131–170
Creutzfeldt OD, Houchin J (1974) Neuronal basis of EEG waves. In: Creutzfeldt OD (eds) Handbook of electroencephalography and clinical neurophysiology, Vol 2, Part C. Elsevier, Amsterdam, pp 2C-5–2C-55
Flatman JA, Schwindt PC, Crill WE, Stafstrom CE (1983) Multiple actions of NMDA on cat neocortical neurones in vitro. Brain Res 266: 169–173
Glenn LL, Steriade M (1982) Discharge rate and excitability of cortically-projectiong neurons in the intralaminar nuclei during waking and sleep states. J Neurosci 2: 1387–1404
Herkenham M (1980) Laminar organisation of thalamic projections to the rat neocortex. Science 207: 533–535
Holmes O, Houchin J (1966) Units in the cerebral cortex of the anaesthetised rat and the correlations between their discharges. J Physiol 187: 651–671
Hubel DH (1959) Single unit activity in the striate cortex of unanaesthetised cats. J Physiol 147: 226–238
Jones BE, Harper ST, Halaris AF (1977) Effects of locus coeruleus lesions upon cerebral monoamine content, sleep wakefulness states and responses to amphetamines in the cat. Brain Res 124: 473–496
Jones EG, Leavitt RY (1974) Retrograde axonal transport and the demonstration of non-specific projections to the cerebral cortex and striatum from thalamic intralaminar nuclei in the rat, cat and monkey. J Comp Neurol 154: 349–378
Jones EG (1981) Functional subdivision and synaptic organisation of the mammalian thalamus. In: Porter R (eds) Neurophysiology IV. International review of physiology, Vol 25, Chap 5. University Park Press Baltimore
Jouvet M (1972) Neurophysiology and neurochemistry of sleep and wakefulness, In: Reviews of physiology, Vol 64. Springer, Berlin Heidelberg New York, pp 137–167
Konig JFR, Klippel RA (1963) The rat brain: a stereotaxic atlas of the forebrain and lower parts of the brain stem. Williams and Wilkins, Baltimore, MD
Krnjevic K, Phillis JW (1963) Acetyl choline sensitive cells in the cerebral cortex. J Physiol 166: 296–327
Macchi G, Bentivoglio M, D'Atena C, Rossini P, Tempesta E (1977) The cortical projections of the thalamic intralaminar nuclei restudied by means of the HRP retrograde axonal transport. Neurosci Lett 4: 121–126
Manzoni T, Barbaresi P, Bellardinelli E, Canialti R (1980) Callosal connections from the two body midlines. Brain Res 39: 1–9
McGinty PJ, Sterman MB (1968) Sleep suppression after basal forebrain lesions in the cat. Science 160: 1253–1255
Moore RY, Bloom FE (1979) Central catecholamine neuron systems: anatomy and physiology of the norepinephrine systems. Ann Rev Neurosci 2: 113–168
Moore RY, Halaris AE, Jones BE (1978) Seretonin systems of the midbrain raphé: ascending projections. J Comp Neurol 180: 417–438
Morison RS, Dempsey EW (1942) Production of rhythmically recurrent potentials after localised thalamic stimulation. Am J Physiol 135: 293–300
Moruzzi G (1963) Active processes in the brain stem during sleep. The Harvey lectures 58: 233–297. Academic Press, New York
Noda H, Adey WR (1970) Firing of neuron pairs in cat association cortex during sleep and wakefulness. J Neurophysiol 33: 672–684
Ottersen OP, Fisher BO, Storm-Mathisen J (1983) Retrograde transport of D(3H) aspartate in thalamocortical neurons. Neurosci Lett 42: 19–24
Paxinos G, Watson C (1982) The rat brain in stereotaxic coordinates. Academic Press, New York
Rechtschaffen A, Kales A (1976) A manual of standardised terminology, techniques and scoring system for sleep stages of human subjects. Brain Information Service, Brain Research Institute, University College of Los Angeles
Robinson TE, Vanderwolf CH, Pappas BA (1977) Are the dorsal noradrenergic bundle projections from the Locus Coeruleus important for neocortical or hippocampal activation? Brain Res 138: 75–98
Saper LB (1984) Organization of the cerebral cortical afferent systems of the rat. Part II. Magnocellular basal nucleus. J Comp Neurol 222: 313–432
Scheibel ME, Scheibel AB (1967) Structural organisation of non-specific nuclei and their projection toward the cortex. Brain Res 6: 60–94
Spehlmann R, Smathers CCJr (1974) The effects of acetyl choline and of synaptic stimulation on the sensorimotor cortex of cats: comparison of the neuronal responses to reticular and other stimuli. Brain Res 74: 243–253
Sterman MB, Clemente CD (1962) Forebrain inhibitory mechanisms: cortical synchronization induced by basal forebrain stimulation. Exp Neurol 6: 91–102
Streit P (1980) Selective retrograde labelling indicating the transmitter of neuronal pathways. J Comp Neurol 191: 429–463
Trulson ME, Jacobs BL (1979) Raphé unit activity in freely moving cats: correlation with level of behavioural arousal. Brain Res 163: 135–150
Walker AE (1938) The primate thalamus. University of Chicago Press, Chicago
Watkins JC, Evans RH (1981) Excitatory amino acids transmitters. Ann Rev Pharmacol Toxicol 21: 165–204
Wise SP, Jones EG (1976) The organisation and postnatal development of the commissural projection of the rat somatic sensory cortex. J Comp Neurol 168: 313–344
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fox, K., Armstrong-James, M. The role of the anterior intralaminar nuclei and N-methyl D-aspartate receptors in the generation of spontaneous bursts in rat neocortical neurones. Exp Brain Res 63, 505–518 (1986). https://doi.org/10.1007/BF00237474
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00237474